Advanced Gold-Catalyzed Synthesis of Spiro Compounds for Commercial Pharmaceutical Manufacturing
Advanced Gold-Catalyzed Synthesis of Spiro Compounds for Commercial Pharmaceutical Manufacturing
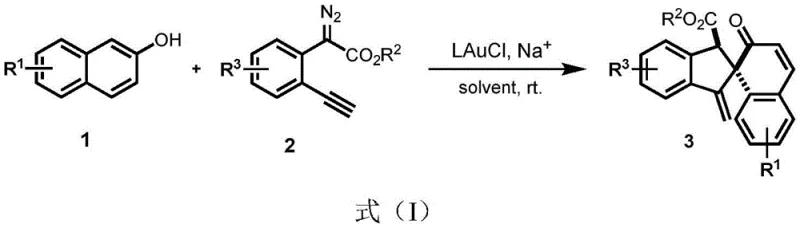
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex molecular architectures, particularly those containing quaternary carbon centers which are prevalent in bioactive natural products and drug candidates. Patent CN111732508B introduces a groundbreaking synthetic methodology that addresses these challenges by utilizing a gold-catalyzed tandem reaction to build spirocyclic skeletons directly from 2-naphthol derivatives. This innovation represents a significant leap forward in synthetic efficiency, enabling the direct functionalization of C-H bonds coupled with dearomatization in a single operational step. By leveraging the unique reactivity of gold carbenes, this process bypasses the need for pre-functionalized starting materials or harsh reaction conditions, thereby offering a streamlined approach that aligns perfectly with the principles of green chemistry and modern process intensification strategies required by today's demanding supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditionally, the construction of spirocyclic frameworks has been a formidable challenge in organic synthesis, often requiring multi-step sequences that involve tedious protection and deprotection strategies, stoichiometric amounts of oxidants, or the use of toxic heavy metal reagents. Conventional routes frequently suffer from poor atom economy and low overall yields due to the accumulation of losses at each synthetic step, which drastically increases the cost of goods sold for the final active pharmaceutical ingredient. Furthermore, many existing methods rely on harsh acidic or basic conditions that are incompatible with sensitive functional groups, limiting the structural diversity that can be accessed and complicating the purification process. These inefficiencies create significant bottlenecks for procurement teams who struggle with volatile pricing and supply chain disruptions caused by complex, low-yielding manufacturing processes that are difficult to scale reliably.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN111732508B utilizes a sophisticated gold-catalyzed cascade reaction that merges C-H bond functionalization and dearomatization into a single, elegant transformation. This novel approach employs readily available 2-naphthol and ortho-alkynyl diazo compounds as starting materials, reacting them under mild room temperature conditions in common solvents like dichloromethane. The use of a gold catalyst allows for the generation of reactive gold carbene intermediates that selectively insert into the aromatic C-H bond, followed immediately by an intramolecular cyclization to form the spiro center. This one-pot strategy not only reduces the number of unit operations but also eliminates the need for external directing groups, resulting in a cleaner reaction profile and significantly simplified downstream processing.
Mechanistic Insights into Gold-Catalyzed C-H Functionalization and Dearomatization
The mechanistic pathway of this transformation is a testament to the power of homogeneous gold catalysis in enabling complex bond constructions with high precision. The reaction initiates with the activation of the diazo compound by the gold catalyst to form a reactive gold carbene intermediate, which then undergoes electrophilic addition with the nucleophilic 2-naphthol substrate. This step generates an ionic intermediate that, following protonolysis of the gold species, is rapidly captured by the tethered alkyne moiety to form a new gold complex. Subsequent activation of the phenolic hydroxyl group by the counter-anion facilitates a 5-exo-dig cyclization, leading to the formation of an exocyclic alkenyl gold complex. The cycle concludes with a final protonolysis step that releases the desired spiro compound and regenerates the active gold catalyst, ensuring high turnover numbers and catalytic efficiency throughout the process.
From an impurity control perspective, this mechanism offers distinct advantages due to its high chemoselectivity and the mild nature of the reaction conditions. Because the transformation proceeds through a well-defined catalytic cycle at room temperature, there is minimal risk of thermal decomposition or non-specific side reactions that often plague high-temperature processes. The specificity of the gold carbene insertion ensures that functionalization occurs exclusively at the desired position on the naphthol ring, minimizing the formation of regioisomers that are difficult to separate. This inherent selectivity translates directly into a cleaner crude reaction mixture, reducing the burden on purification teams and ensuring that the final product meets stringent purity specifications required for pharmaceutical applications without extensive recrystallization or chromatographic efforts.
How to Synthesize Spiro Compounds Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to catalyst preparation and addition rates to maximize yield and reproducibility. The process begins with the activation of the gold catalyst system in a dry solvent environment, followed by the controlled addition of the diazo reagent to manage the exotherm and concentration of the reactive carbene species. Detailed standard operating procedures regarding stoichiometry, solvent quality, and workup protocols are essential for transferring this chemistry from bench scale to commercial production. For a comprehensive breakdown of the specific experimental parameters and purification techniques validated in the patent examples, please refer to the standardized synthesis guide below.
- Prepare the catalyst system by mixing gold catalyst and sodium salt in dry dichloromethane at room temperature.
- Add 2-naphthol substrate and slowly dropwise add the diazo compound solution while stirring.
- Monitor reaction by TLC, quench with water, extract, and purify via column chromatography to isolate the spiro product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this gold-catalyzed methodology presents a compelling value proposition centered around cost optimization and supply reliability. By consolidating multiple synthetic steps into a single pot reaction, manufacturers can significantly reduce the consumption of solvents, reagents, and energy, leading to a drastic simplification of the production workflow. The elimination of intermediate isolation steps not only saves time but also reduces the physical footprint required for manufacturing, allowing for higher throughput in existing facilities. Furthermore, the use of commercially abundant starting materials like 2-naphthol mitigates the risk of raw material shortages, ensuring a stable and continuous supply of critical intermediates for downstream drug synthesis.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis directly correlates to substantial cost savings by removing the need for expensive protecting group chemistry and multiple purification stages. Since the reaction operates at room temperature, there is no requirement for energy-intensive heating or cooling infrastructure, which lowers utility costs and reduces the carbon footprint of the manufacturing process. Additionally, the high atom economy of the transformation means that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs and maximizing the return on investment for every kilogram of input material purchased.
- Enhanced Supply Chain Reliability: Relying on a robust synthetic route with broad substrate scope allows for greater flexibility in sourcing raw materials, as various substituted naphthols and diazo compounds can be utilized without re-optimizing the entire process. This flexibility acts as a buffer against market volatility, ensuring that production schedules can be maintained even if specific grades of reagents face temporary availability issues. The simplicity of the reaction setup also means that technology transfer to contract manufacturing organizations is faster and less prone to failure, securing the long-term continuity of supply for critical pharmaceutical programs.
- Scalability and Environmental Compliance: The mild reaction conditions and absence of toxic heavy metal waste streams make this process inherently safer and easier to scale from gram to ton quantities. Regulatory compliance is simplified as the process avoids the use of restricted reagents, facilitating smoother approval pathways for new drug applications. The reduced generation of chemical waste aligns with increasingly strict environmental regulations, protecting the company from potential fines and reputational damage while promoting a sustainable manufacturing image that is highly valued by global partners.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this spiro compound synthesis technology. These answers are derived directly from the experimental data and technical specifications outlined in the patent documentation to provide clarity on feasibility and performance. Understanding these details is crucial for R&D teams evaluating this route for inclusion in their development pipelines and for procurement specialists assessing the long-term viability of the supply chain.
Q: What are the key advantages of this gold-catalyzed method over traditional spiro compound synthesis?
A: This method achieves tandem C-H functionalization and dearomatization in a single pot without directing groups, significantly simplifying the synthetic route and improving atom economy compared to multi-step traditional methods.
Q: Is this synthesis method scalable for industrial production of pharmaceutical intermediates?
A: Yes, the reaction operates under mild room temperature conditions with commercially available reagents like 2-naphthol, making it highly suitable for scale-up and continuous supply chain integration.
Q: What is the substrate scope for this spiro compound synthesis technique?
A: The method demonstrates broad substrate tolerance, accommodating various substituents on the naphthol ring including halogens, alkyl groups, and esters, allowing for diverse library generation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Spiro Compound Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN111732508B for accelerating drug discovery and development. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory chemistry can be successfully translated into robust industrial processes. Our state-of-the-art facilities are equipped with rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of spiro intermediate we deliver meets the highest standards of quality and consistency required by the global pharmaceutical industry.
We invite you to collaborate with us to leverage this cutting-edge synthesis technology for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements and timeline constraints. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our expertise in gold-catalyzed transformations can drive efficiency and value for your organization.
