Advanced Manufacturing of Benidipine Hydrochloride: A Scalable Route for Global Pharma Supply Chains
The global pharmaceutical landscape is continuously demanding more efficient and safer pathways for producing critical cardiovascular medications, particularly calcium channel blockers like Benidipine Hydrochloride. As hypertension prevalence rises globally, the need for robust supply chains delivering high-purity active pharmaceutical ingredients (APIs) has never been more urgent. A groundbreaking patent, CN112679416A, filed in April 2021, introduces a transformative preparation method that addresses long-standing inefficiencies in Benidipine synthesis. This novel approach leverages a streamlined sequence of Knoevenagel condensation, Michael addition, and selective esterification to achieve exceptional product quality. By eliminating the need for column chromatography and utilizing mild reaction conditions, this technology offers a compelling solution for manufacturers seeking to optimize their production lines. The reported HPLC purity exceeds 99.5%, with overall yields surpassing 68%, setting a new benchmark for industrial feasibility in the production of this vital antihypertensive agent.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Benidipine Hydrochloride has been plagued by significant operational hurdles and safety concerns that hinder large-scale adoption. Traditional routes, such as those described in patent CN103641774B, rely on the formation of mixed anhydrides using chlorophosphate under alkaline conditions, followed by reflux with N-benzyl-3-hydroxypiperidine. This legacy methodology suffers from inherently low yields, typically ranging between 40.1% and 49.2%, primarily due to complicated post-treatment procedures and difficult crystallization steps that require extended processing times. Furthermore, alternative pathways documented in EP 0063365 often necessitate the use of unstable starting materials, such as N-benzyl-3-piperidyl-3-aminocrotonate, which carry inherent polymerization explosion risks. These safety hazards not only increase operational liability but also complicate regulatory compliance and environmental management, making such routes less attractive for modern, safety-conscious manufacturing facilities aiming for consistent output.
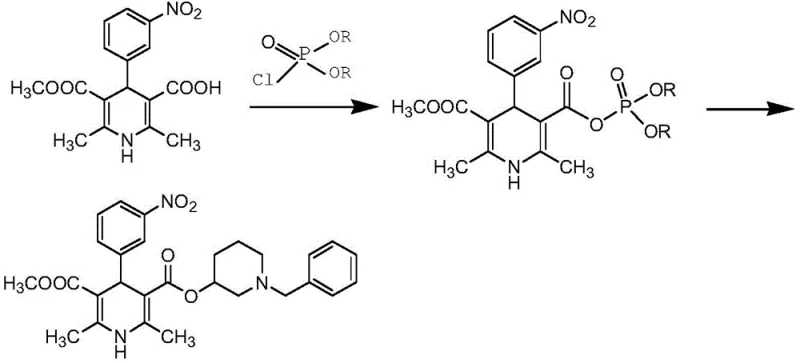
The Novel Approach
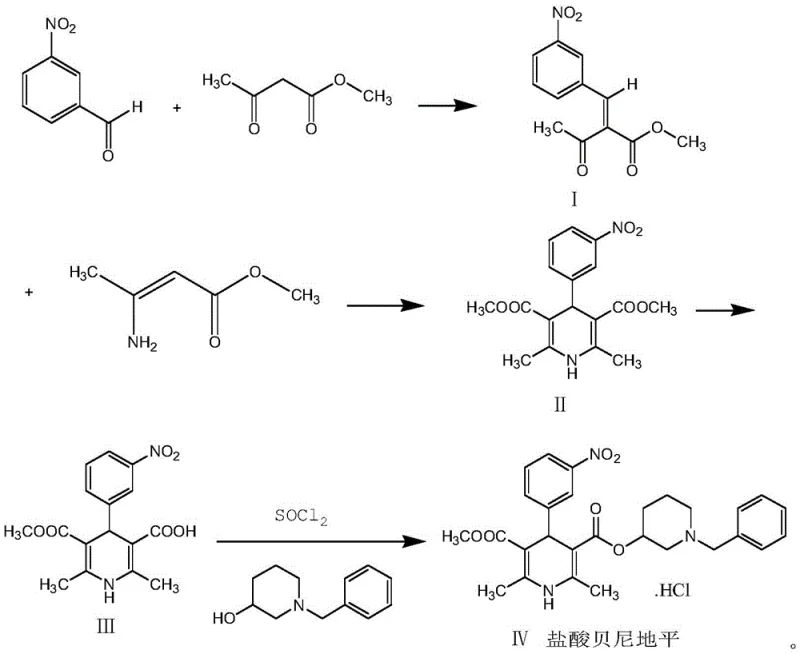
In stark contrast to these cumbersome legacy processes, the innovative strategy outlined in CN112679416A presents a radically simplified and safer synthetic trajectory. This method initiates with a Knoevenagel condensation between readily available 3-nitrobenzaldehyde and methyl acetoacetate, catalyzed effectively by concentrated sulfuric acid or acetic acid-piperidine. The resulting intermediate undergoes a seamless Michael addition and cyclization with beta-amino methyl crotonate in common solvents like absolute ethanol, avoiding the need for exotic or hazardous reagents. The subsequent steps involve selective hydrolysis and conversion to the acid chloride using thionyl chloride, followed by direct coupling with 1-benzyl-3-hydroxypiperidine. This logical progression minimizes side reactions and eliminates the dangerous polymerization risks associated with older methods. The result is a robust process that delivers high-purity product suitable for direct pharmaceutical application without the burden of extensive chromatographic purification.
Mechanistic Insights into the Modified Hantzsch Pyridine Synthesis
The core of this technological advancement lies in the precise control of the Hantzsch pyridine synthesis mechanism, specifically tailored to maximize regioselectivity and minimize impurity formation. The initial Knoevenagel condensation creates an activated olefinic intermediate, 2-(3-nitrobenzylidene)-methyl acetoacetate, which serves as a highly reactive electrophile for the subsequent Michael addition. When reacted with beta-amino methyl crotonate, the system undergoes a concerted cyclization where the amine nitrogen attacks the carbonyl carbon while the enamine double bond attacks the activated alkene. This cascade reaction is meticulously managed by controlling the temperature between 40-50°C in anhydrous alcohol solvents, ensuring that the dihydropyridine ring closes efficiently to form the dimethyl ester intermediate. The choice of solvent is critical here; absolute ethanol or methanol facilitates the necessary proton transfers while maintaining the solubility of the intermediates, preventing premature precipitation that could trap impurities within the crystal lattice.
Following the construction of the dihydropyridine core, the process employs a sophisticated selective hydrolysis strategy to differentiate between the two ester groups at the 3 and 5 positions. By utilizing sodium hydroxide in a methanol-water system at moderate temperatures (40-60°C), the method achieves selective mono-hydrolysis to generate the 3-carboxylic acid derivative while preserving the 5-methyl ester. This selectivity is paramount for the final coupling step, where the free acid is activated using thionyl chloride to form an acid chloride in situ. This activated species then reacts rapidly with 1-benzyl-3-hydroxypiperidine to form the final ester linkage. The entire sequence is designed to avoid harsh conditions that could oxidize the sensitive dihydropyridine ring to the corresponding pyridine, a common degradation pathway that compromises potency. The final salification with hydrochloric acid ensures the formation of the stable hydrochloride salt, ready for isolation via simple crystallization.
How to Synthesize Benidipine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to reaction parameters, particularly temperature control during the exothermic condensation and chlorination steps. The process begins with the preparation of the benzylidene intermediate, followed by the cyclization to form the dihydropyridine scaffold. Operators must ensure strict moisture control during the cyclization phase to prevent hydrolysis of the starting esters before ring closure. Following the formation of the dimethyl ester, the selective hydrolysis step demands precise pH monitoring to ensure only one ester group is cleaved. The final coupling involves handling thionyl chloride, requiring appropriate gas scrubbing systems to manage HCl and SO2 off-gassing. For a comprehensive, step-by-step technical guide including specific molar ratios, stirring speeds, and workup procedures, please refer to the standardized protocol below.
- Perform Knoevenagel condensation between 3-nitrobenzaldehyde and methyl acetoacetate using concentrated sulfuric acid catalyst to form the benzylidene intermediate.
- Execute Michael addition and cyclization with beta-amino methyl crotonate in absolute ethanol to construct the dihydropyridine core structure.
- Conduct selective hydrolysis of the dimethyl ester using sodium hydroxide, followed by chlorination with thionyl chloride and coupling with 1-benzyl-3-hydroxypiperidine.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this novel synthesis route offers profound strategic advantages that extend far beyond simple chemical efficiency. The elimination of column chromatography, a notoriously expensive and time-consuming purification technique, translates directly into substantial cost reductions in manufacturing overhead. By relying on simple crystallization and filtration for purification, facilities can drastically reduce solvent consumption and waste disposal costs, aligning with increasingly stringent environmental regulations. Furthermore, the use of stable, commodity-grade starting materials like 3-nitrobenzaldehyde and methyl acetoacetate mitigates supply chain volatility. Unlike specialized enamines that may have limited suppliers and long lead times, these bulk chemicals are widely produced, ensuring a reliable [pharmaceutical intermediates] supplier base that can support continuous production schedules without the risk of raw material shortages.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the removal of complex purification stages and the utilization of high-yield reactions that maximize raw material conversion. By avoiding the use of expensive coupling reagents like chlorophosphate and eliminating the need for silica gel chromatography, the overall cost of goods sold (COGS) is significantly lowered. The simplified workup procedure reduces labor hours and equipment occupancy time, allowing for higher throughput in existing reactor trains. Additionally, the high purity achieved (>99.5%) minimizes the need for reprocessing or recycling off-spec batches, further enhancing the economic viability of the operation for any entity focused on cost reduction in [pharmaceutical intermediates] manufacturing.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the reliance on non-hazardous, universally available feedstocks that do not require special storage or transport permits. The avoidance of explosion-prone intermediates removes a major bottleneck in logistics and warehousing, facilitating smoother movement of materials across international borders. This stability allows for more accurate demand forecasting and inventory planning, reducing the need for excessive safety stock. Consequently, lead times for high-purity [pharmaceutical intermediates] can be reduced, enabling faster response to market fluctuations and ensuring that downstream API manufacturers receive their materials on schedule without disruption.
- Scalability and Environmental Compliance: From a scale-up perspective, the mild reaction conditions (mostly below 60°C) and the use of common solvents like ethanol and dichloromethane make this process highly adaptable to large-scale reactors. The absence of extreme temperatures or pressures reduces the engineering complexity required for commercial scale-up of complex [pharmaceutical intermediates]. Moreover, the process generates less hazardous waste compared to traditional routes, simplifying effluent treatment and lowering the environmental footprint. This alignment with green chemistry principles not only reduces compliance costs but also enhances the corporate sustainability profile, a key metric for modern pharmaceutical partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Benidipine Hydrochloride synthesis method. These insights are derived directly from the experimental data and claims presented in the patent literature, providing a factual basis for decision-making. Understanding these nuances is essential for R&D teams evaluating technology transfer and for procurement officers assessing vendor capabilities. The answers reflect the specific advantages of the Knoevenagel-Hantzsch cascade approach over legacy anhydride methods.
Q: What is the purity level achievable with this new Benidipine synthesis route?
A: The patented process achieves an HPLC purity of over 99.5% without the need for complex column chromatography separation, significantly simplifying the purification workflow compared to traditional methods.
Q: Does this manufacturing process involve hazardous explosive reagents?
A: No, unlike prior art methods that require raw materials with polymerization explosion risks, this route utilizes stable, commercially available starting materials like 3-nitrobenzaldehyde and methyl acetoacetate, ensuring a safer industrial environment.
Q: How does the yield of this method compare to conventional mixed anhydride routes?
A: This optimized protocol delivers a total yield exceeding 68%, which represents a substantial improvement over conventional mixed anhydride methods that typically struggle to achieve yields above 50% due to complex post-treatment requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benidipine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of advanced pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this novel synthesis are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify that every batch meets the >99.5% HPLC purity standard. Our infrastructure is designed to handle the specific solvent systems and reaction conditions required for this route, guaranteeing a consistent supply of high-quality Benidipine Hydrochloride for your formulation needs.
We invite global partners to collaborate with us to leverage this cutting-edge technology for their supply chains. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis that quantifies the potential efficiencies of switching to this manufacturing route. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your volume requirements. Let us help you secure a competitive advantage through superior chemistry and reliable delivery.
