Advanced Manufacturing of Benidipine Hydrochloride: A Safer, Scalable Route for Global Pharma Supply Chains
Advanced Manufacturing of Benidipine Hydrochloride: A Safer, Scalable Route for Global Pharma Supply Chains
The global demand for high-quality cardiovascular therapeutics continues to surge, placing immense pressure on the supply chains of critical active pharmaceutical ingredients (APIs) and their precursors. Among these, Benidipine Hydrochloride stands out as a potent dihydropyridine calcium antagonist, renowned for its long duration of action and high affinity for Ca2+ channels. However, the historical manufacturing landscape for this compound has been fraught with safety hazards and process inefficiencies. The groundbreaking methodology detailed in patent CN103641774A introduces a paradigm shift by utilizing a mixed acid anhydride intermediate strategy. This approach not only circumvents the use of explosive reagents like diketene but also eliminates the need for energy-intensive cryogenic reactions typically associated with acid chloride formations. For procurement leaders and R&D directors seeking a reliable pharmaceutical intermediate supplier, understanding this technological leap is crucial for securing a stable, cost-effective, and compliant supply of this vital hypertension medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Benidipine Hydrochloride has relied on pathways that pose significant operational risks and yield inconsistencies. Early methods, such as those disclosed in EP0063365, often involved the condensation of 3-nitrobenzaldehyde with unstable intermediates, resulting in prolonged reaction times and suboptimal yields. More critically, certain conventional routes necessitate the use of diketene, a raw material known for its tendency to polymerize explosively at room temperature, creating severe safety liabilities for manufacturing facilities. Furthermore, alternative strategies involving the conversion of carboxylic acids to acid chlorides using thionyl chloride require rigorous low-temperature control, often down to -20°C. These cryogenic conditions are not only energy-prohibitive on a commercial scale but also increase the risk of raw material decomposition and side reactions if temperature fluctuations occur, leading to difficult purification processes and inconsistent batch quality.
The Novel Approach
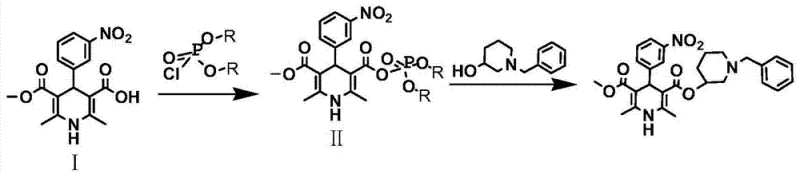
In stark contrast to these legacy techniques, the innovative process defined in CN103641774A leverages a mixed acid anhydride mechanism to achieve esterification under remarkably mild conditions. By reacting the dihydropyridine carboxylic acid derivative with a chlorophosphate ester in the presence of an organic base, the synthesis generates a highly reactive yet stable mixed anhydride intermediate at ambient temperatures ranging from 20°C to 30°C. This intermediate subsequently reacts with N-benzyl-3-hydroxypiperidine to form the target molecule without the need for hazardous reagents or extreme thermal management. This methodological pivot effectively resolves the safety concerns associated with diketene and the operational complexities of cryogenic acid chloride synthesis, offering a streamlined pathway that is inherently safer, easier to purify, and far more conducive to large-scale industrial production.
Mechanistic Insights into Mixed Acid Anhydride Esterification
The core of this technological advancement lies in the precise generation and utilization of the mixed acid anhydride species. The reaction initiates when 2,6-dimethyl-4-(m-nitrophenyl)-1,4-dihydropyridine-5-carboxylic acid methyl ester-3-carboxylic acid is treated with a chlorophosphate reagent, such as diethyl chlorophosphate, under alkaline conditions provided by triethylamine or pyridine. This activation step converts the relatively unreactive carboxylic acid into a potent electrophile capable of undergoing nucleophilic attack by the hydroxyl group of the piperidine derivative. Unlike acid chlorides, which are prone to hydrolysis and require strict moisture exclusion and low temperatures, the mixed anhydride formed here exhibits sufficient stability to be generated and consumed in situ at 20-30°C. This thermal tolerance significantly reduces the kinetic barriers to reaction while minimizing the formation of thermal degradation by-products that often plague high-temperature or highly exothermic processes.
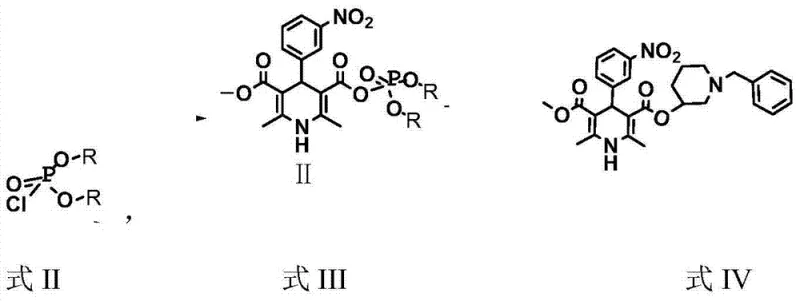
Furthermore, the impurity profile of the final product is meticulously controlled through this mechanism. The mild reaction environment prevents the decomposition of the sensitive dihydropyridine ring, a common issue in harsher synthetic routes. Following the coupling reaction, the process incorporates a robust purification sequence involving activated carbon decolorization at 40-50°C, followed by a multi-stage aqueous wash. This includes treatment with sodium hydroxide to remove acidic impurities, water to neutralize salts, and hydrochloric acid to convert the free base into the stable hydrochloride salt. The final recrystallization from an ethanol-acetone solvent system ensures the removal of any residual organic impurities, consistently delivering a product with HPLC purity exceeding 99.5%. This level of control over the chemical landscape ensures that the final API intermediate meets the rigorous specifications required for downstream pharmaceutical formulation.
How to Synthesize Benidipine Hydrochloride Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and thermal profiles to maximize yield and safety. The process begins with the activation of the dihydropyridine acid in a suitable organic solvent such as dichloromethane or ethyl acetate. Once the mixed anhydride is confirmed via TLC monitoring, the N-benzyl-3-hydroxypiperidine is introduced, and the mixture is allowed to reflux gently. The simplicity of the workup procedure, which avoids complex chromatographic separations in favor of crystallization, makes this route particularly attractive for commercial manufacturing. Below is the structured guide for executing this high-efficiency synthesis protocol.
- React 2,6-dimethyl-4-(m-nitrophenyl)-1,4-dihydropyridine-5-carboxylic acid methyl ester-3-carboxylic acid with chlorophosphate and organic base at 20-30°C to form the mixed anhydride.
- Add N-benzyl-3-hydroxypiperidine to the reaction mixture, stir at ambient temperature, and then reflux for 4-6 hours to complete the esterification.
- Purify the crude product through activated carbon decolorization, sequential aqueous washing, and final recrystallization using an ethanol-acetone solvent system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this mixed anhydride-based synthesis offers profound strategic benefits beyond mere chemical elegance. The elimination of cryogenic requirements translates directly into substantial reductions in energy consumption and capital expenditure on specialized cooling infrastructure. Moreover, the removal of explosive raw materials like diketene from the supply chain drastically lowers insurance premiums and regulatory compliance burdens associated with handling hazardous substances. This process stability ensures that production schedules are not disrupted by safety incidents or the logistical challenges of sourcing dangerous reagents, thereby enhancing the overall reliability of the supply chain for this critical cardiovascular intermediate.
- Cost Reduction in Manufacturing: The operational expenditure (OPEX) for this process is significantly optimized by the absence of energy-intensive cooling systems. Traditional acid chloride routes often require maintaining reactors at -20°C for extended periods, consuming vast amounts of electricity and refrigerant capacity. By operating at ambient temperatures (20-30°C), this method utilizes standard heating and stirring equipment, reducing utility costs. Additionally, the high selectivity of the mixed anhydride reaction minimizes the formation of by-products, which reduces the volume of solvents and reagents needed for purification, further driving down the cost of goods sold (COGS) without compromising quality.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the volatility of hazardous raw material markets. Diketene, used in older methods, is subject to strict transport regulations and limited supplier availability due to its instability. By substituting this with stable chlorophosphates and readily available hydroxypiperidine derivatives, the supply chain becomes far more resilient. The robustness of the reaction conditions also means that production is less susceptible to delays caused by equipment maintenance or environmental constraints, ensuring a steady flow of high-purity intermediates to downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling chemical processes often amplifies safety risks, but this methodology is inherently designed for expansion. The mild exothermic profile allows for safe operation in large-volume reactors without the risk of thermal runaway. From an environmental perspective, the process generates less hazardous waste compared to thionyl chloride routes, which produce corrosive sulfur dioxide gas. The use of recyclable solvents like dichloromethane and the ability to recover unreacted materials through simple washing steps align with modern green chemistry principles, facilitating easier permitting and long-term sustainability for manufacturing sites.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Benidipine Hydrochloride using this advanced mixed anhydride protocol. These insights are derived directly from the experimental data and process descriptions found in the underlying patent literature, providing a transparent view of the technology's capabilities and limitations for potential partners.
Q: Why is the mixed anhydride method superior to the acid chloride route for Benidipine?
A: The mixed anhydride method operates at mild temperatures (20-30°C), eliminating the need for hazardous cryogenic conditions (-20°C) and violent reagents like thionyl chloride, thereby significantly improving operational safety and scalability.
Q: What purity levels can be achieved with this synthesis protocol?
A: Through optimized recrystallization using ethanol and acetone, this process consistently achieves HPLC purity levels exceeding 99.5%, meeting stringent pharmaceutical quality standards.
Q: Does this process avoid the use of explosive raw materials?
A: Yes, unlike conventional Hantzsch variants that may utilize dangerous diketene, this patented route relies on stable chlorophosphates and hydroxypiperidine derivatives, removing significant explosion risks from the manufacturing floor.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Benidipine Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of complex pharmaceutical intermediates requires more than just a patent; it demands deep process engineering expertise and a commitment to quality. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the mixed anhydride route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced HPLC and spectroscopic tools to guarantee that every batch of Benidipine Hydrochloride meets the exacting standards of the global pharmaceutical industry.
We invite you to collaborate with us to optimize your supply chain for cardiovascular therapeutics. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can drive value and security for your organization.
