Advanced Purification Technology for Vildagliptin: Enhancing Yield and Purity for Global Pharmaceutical Supply Chains
Advanced Purification Technology for Vildagliptin: Enhancing Yield and Purity for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust manufacturing processes that balance high purity with economic efficiency, particularly for critical antidiabetic agents like Vildagliptin. Patent CN113527168B introduces a transformative purification methodology that addresses the persistent challenge of removing disubstituted impurities without compromising overall yield. This innovation is pivotal for manufacturers aiming to secure a reliable API intermediate supplier status in the competitive diabetes treatment market. The core of this technology lies in a sophisticated acid-base extraction protocol that replaces laborious and yield-depleting recrystallization sequences. By leveraging precise pH control and selective solvent partitioning, the process ensures that the final active pharmaceutical ingredient meets stringent regulatory standards while maximizing material throughput. This report analyzes the technical merits and commercial implications of this patented approach for global procurement and R&D strategies.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
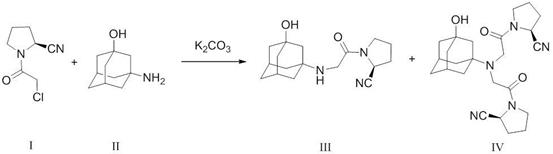
Historically, the synthesis of Vildagliptin involves the condensation of (2S)-N-chloroacetyl-2-cyano-pyrrolidine with 3-amino-1-adamantanol, a reaction that inevitably generates a structurally similar byproduct known as the Vildagliptin disubstituted compound. Conventional purification strategies rely heavily on repeated recrystallization to reduce the levels of this specific impurity to acceptable limits, typically below 0.15 percent. However, this approach is inherently inefficient, as each crystallization cycle results in significant product loss trapped within the mother liquor, driving down the overall process yield. Furthermore, the solubility characteristics of the disubstituted impurity often mimic the target molecule, making separation difficult and requiring excessive solvent consumption and extended processing times. These operational bottlenecks not only inflate manufacturing costs but also introduce variability in batch-to-batch consistency, posing risks for supply chain reliability and regulatory compliance in large-scale production environments.
The Novel Approach
In stark contrast to traditional solid-state purification, the method disclosed in patent CN113527168B employs a liquid-liquid extraction strategy that fundamentally alters the separation mechanism. Instead of relying on differential solubility in a single solvent system, this novel approach exploits the acid-base properties of the Vildagliptin molecule to achieve selective partitioning between aqueous and organic phases. The process begins by dissolving the crude product in an acidic aqueous solution, where the target amine functionality is protonated to form a water-soluble salt, while the neutral disubstituted impurity remains preferentially soluble in the organic extraction solvent. This single operational shift allows for the bulk removal of the problematic impurity in the early stages of workup, eliminating the need for multiple recrystallization steps. Consequently, the process achieves a remarkable purification efficiency, controlling the disubstituted impurity to below 0.1 percent while maintaining a process yield exceeding 90 percent, thereby offering a superior alternative for cost reduction in pharmaceutical intermediates manufacturing.
Mechanistic Insights into Acid-Base Extraction Purification
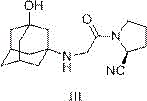
The efficacy of this purification method is rooted in the precise manipulation of ionization states through pH control, a critical parameter for R&D directors focused on impurity profiling. During the initial extraction phase, the aqueous environment is maintained at a pH range of 3 to 5 using acids such as citric acid, hydrochloric acid, or potassium dihydrogen phosphate. At this acidity level, the secondary amine group within the Vildagliptin structure (Formula III) becomes protonated, rendering the molecule highly polar and water-soluble as a cationic species. Conversely, the disubstituted impurity (Formula IV), which lacks the same basic character or possesses different steric and electronic properties, does not protonate to the same extent and retains its lipophilicity. When an immiscible organic solvent such as ethyl acetate or dichloromethane is introduced, the neutral impurity partitions strongly into the organic layer, effectively stripping it from the aqueous phase containing the desired product salt. This mechanism ensures that the impurity is physically separated from the product stream before the final isolation step, providing a robust barrier against contamination that is far more effective than simple washing procedures.
Following the removal of impurities, the recovery of the pure product relies on a second pH adjustment to reverse the ionization state. The aqueous phase, now depleted of the disubstituted byproduct, is treated with an alkaline reagent such as sodium bicarbonate or sodium carbonate to raise the pH to a range of 7 to 9. This basification deprotonates the Vildagliptin salt, regenerating the free base form of the molecule which is significantly less soluble in water and highly soluble in organic solvents. A subsequent extraction with an organic solvent transfers the purified Vildagliptin from the aqueous phase into the organic phase, leaving behind inorganic salts and water-soluble residues. The careful control of the upper pH limit is essential, as excessively high alkalinity can lead to degradation of the nitrile group or other sensitive functionalities, while insufficient basification would result in poor extraction efficiency. This two-stage pH swing extraction creates a highly selective purification window that maximizes both purity and recovery, demonstrating a deep understanding of physical organic chemistry applied to process development.
How to Synthesize Vildagliptin Efficiently
The implementation of this purification protocol requires strict adherence to the defined stoichiometric ratios and phase separation techniques to ensure reproducibility at scale. The patent outlines a clear sequence where the crude material is first subjected to acidic washes to remove lipophilic contaminants, followed by basification and organic extraction to isolate the pure active ingredient. This workflow is designed to be compatible with standard chemical engineering equipment, facilitating easy technology transfer from laboratory to pilot and commercial scales. The detailed standardized synthesis steps see the guide below for specific operational parameters regarding solvent volumes and mixing times.
- Dissolve crude Vildagliptin in an acidic aqueous solution (pH 3-5) and extract with an organic solvent like ethyl acetate or dichloromethane to remove disubstituted impurities into the organic phase.
- Collect the aqueous phase containing the Vildagliptin salt and adjust the pH to 7-9 using an alkaline reagent such as sodium bicarbonate or sodium carbonate.
- Extract the basified aqueous phase with an organic solvent to recover the pure Vildagliptin, followed by drying and concentration to obtain the final solid product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this purification technology translates directly into enhanced operational efficiency and reduced total cost of ownership. By eliminating the need for repeated recrystallization, the process significantly reduces solvent consumption and energy usage associated with heating and cooling cycles, leading to substantial cost savings in utility and waste disposal. The simplification of the workflow also shortens the overall production cycle time, allowing for faster turnover of batches and improved responsiveness to market demand fluctuations. Furthermore, the high yield retention means that less raw material is required to produce the same amount of finished goods, optimizing the utilization of expensive starting materials and intermediates. These factors collectively contribute to a more resilient and cost-effective supply chain for high-purity pharmaceutical intermediates.
- Cost Reduction in Manufacturing: The elimination of multiple recrystallization steps removes a major cost driver associated with solvent purchase, recovery, and disposal, while the high yield minimizes raw material waste. This streamlined approach reduces the number of unit operations required, lowering labor costs and equipment occupancy time, which ultimately drives down the manufacturing cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: A simpler process with fewer critical steps reduces the risk of batch failures and deviations, ensuring a more consistent and reliable supply of material. The robustness of the extraction method against variations in crude quality provides a buffer against upstream synthesis fluctuations, guaranteeing steady delivery schedules for downstream formulation partners and reducing the need for safety stock inventory.
- Scalability and Environmental Compliance: Liquid-liquid extraction is a well-established unit operation that scales linearly from laboratory to industrial reactors, minimizing the technical risks associated with commercial scale-up of complex pharmaceutical intermediates. Additionally, the potential to use greener solvents like ethyl acetate or isopropyl acetate instead of chlorinated solvents aligns with modern environmental regulations and sustainability goals, reducing the ecological footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this purification method, derived directly from the patent specifications and experimental data. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this process into existing manufacturing lines. The answers provide clarity on impurity control mechanisms, solvent flexibility, and yield expectations based on the documented examples.
Q: How does this purification method control disubstituted impurities in Vildagliptin?
A: The method utilizes a specific pH range (3-5) during the initial acidic dissolution. Under these conditions, the target Vildagliptin forms a water-soluble salt and remains in the aqueous phase, while the lipophilic disubstituted impurity partitions into the organic extraction solvent, effectively separating them without recrystallization.
Q: What is the expected yield improvement compared to traditional recrystallization?
A: Traditional methods often suffer from yield loss due to multiple recrystallization steps required to lower impurity levels. This patented extraction process maintains the product in solution during purification, achieving yields consistently above 90% while reducing impurity content to below 0.1%.
Q: Which solvents are compatible with this purification process?
A: The process demonstrates flexibility with various organic solvents. Effective extraction can be achieved using chlorinated alkanes such as dichloromethane or chloroform, as well as alkyl acetates including ethyl acetate, methyl acetate, isopropyl acetate, and butyl acetate, allowing for optimization based on cost and availability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Vildagliptin Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient purification technologies in delivering high-quality pharmaceutical intermediates to the global market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the acid-base extraction method for Vildagliptin are seamlessly translated into industrial reality. We maintain stringent purity specifications and operate rigorous QC labs to verify that every batch meets the <0.1% impurity threshold and >90% yield targets outlined in the latest patents. Our commitment to technical excellence ensures that our clients receive materials that are not only chemically pure but also produced with a focus on sustainability and cost-efficiency.
We invite potential partners to engage with our technical procurement team to discuss how this advanced purification strategy can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain insights into the specific economic benefits of switching to this streamlined process for your Vildagliptin requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project needs, ensuring a successful and profitable collaboration in the competitive landscape of antidiabetic drug manufacturing.
