Advanced Synthesis of Olatinib Maleate: Technical Breakthroughs for Commercial Scale-up
Introduction to Advanced Olatinib Maleate Manufacturing
The global demand for effective veterinary pharmaceuticals, particularly for managing allergic dermatitis and pruritus in canines, has placed Olatinib Maleate at the forefront of the animal health market. As a potent JAK1 inhibitor, its commercial viability relies heavily on the efficiency and safety of its synthetic production. Patent CN111909156B introduces a transformative preparation method that addresses the critical bottlenecks of traditional synthesis. This technical disclosure outlines a robust pathway utilizing trans-4-[(tert-butoxycarbonyl)amino]cyclohexanecarboxylic acid and 4-chloro-7-tosyl-7H-pyrrolo[2,3-d]pyrimidine as key starting materials. By integrating reduction, condensation, and esterification into a streamlined sequence, the invention achieves a total yield exceeding 65 percent and product purity surpassing 99 percent. For industry stakeholders, this represents a pivotal shift towards greener, more cost-effective pharmaceutical intermediate manufacturing, ensuring a stable supply chain for this high-value active ingredient.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior art synthesis routes for Olatinib Maleate are plagued by significant operational hazards and inefficiencies that hinder large-scale production. For instance, earlier patents describe routes requiring explosive hydrogen peroxide for oxidation steps, introducing severe safety risks in an industrial setting. Furthermore, traditional methods often rely on methylamine tetrahydrofuran solutions, which are not only expensive but generate difficult-to-treat wastewater containing tetrahydrofuran. The isolation of intermediates in these legacy processes is equally problematic; specific steps require crystallization from ethyl acetate and hexane under nitrogen storage, creating complex handling requirements. Additionally, the use of strong bases like sodium sulfite in sulfonation steps frequently leads to unwanted hydrolysis of methanesulfonate esters, resulting in low intermediate purity and necessitating cumbersome purification protocols that erode overall process economics.
The Novel Approach
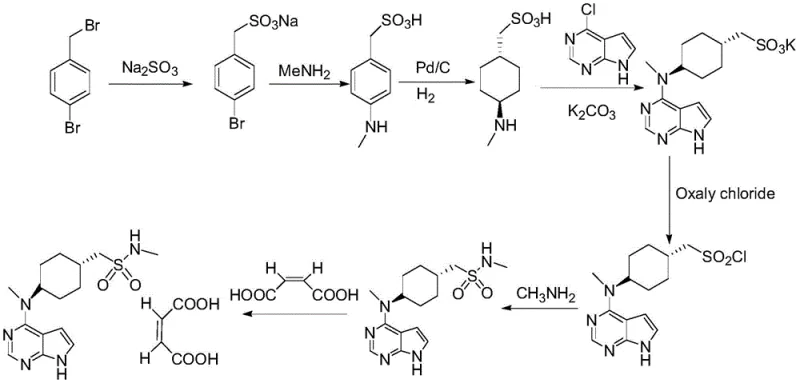
The methodology disclosed in CN111909156B fundamentally reengineers the synthetic landscape by adopting a "one-pot" strategy for the initial reduction, condensation, and esterification phases. This integration drastically reduces post-treatment frequency and eliminates the need for isolating unstable intermediates between these critical transformations. A key innovation lies in the substitution of sodium sulfite with ammonium or lithium sulfite during sulfonation; these weaker bases effectively suppress ester hydrolysis, thereby boosting yields to over 86 percent for this specific step. Moreover, the replacement of organic solvents with aqueous methylamine for the amination step and 95 percent ethanol for salification simplifies solvent recovery and waste management. This holistic optimization ensures that the production of high-purity Olatinib Maleate is not only chemically superior but also operationally streamlined for domestic and international industrial standards.
Mechanistic Insights into Optimized Sulfonation and Deprotection
The chemical elegance of this new process is most evident in the sulfonation and deprotection stages, where precise control over reaction conditions dictates final product quality. In conventional pathways, the use of strongly basic sulfonating agents often triggers the hydrolysis of the sensitive methanesulfonate ester linkage, leading to the formation of alcohol byproducts and reducing the overall mass balance. The patented approach mitigates this by employing ammonium sulfite or lithium sulfite, which provide the necessary nucleophilicity for sulfonation while maintaining a pH environment that preserves the ester functionality. Following sulfonation, the deprotection of the tosyl group is executed under controlled conditions using reagents such as tetrabutylammonium fluoride or alkali metal hydroxides. The subsequent pH adjustment to neutrality using acids like phosphoric or formic acid ensures that the free base is obtained without inducing racemization or degradation, a critical factor for maintaining the biological efficacy of the final veterinary drug substance.
Furthermore, the chlorination and methylamination sequence demonstrates a sophisticated understanding of functional group tolerance. The conversion of the methanesulfonic acid intermediate to the corresponding sulfonyl chloride using oxalyl chloride is performed at low temperatures (0-10 ℃) to prevent thermal decomposition. The immediate subsequent reaction with aqueous methylamine capitalizes on the high reactivity of the sulfonyl chloride, driving the formation of the sulfonamide bond with high conversion rates. This tandem operation avoids the isolation of the potentially unstable sulfonyl chloride, minimizing exposure to moisture and reducing the risk of side reactions. Such mechanistic refinements are essential for a reliable veterinary drug intermediate supplier aiming to deliver consistent batch-to-batch quality in a commercial scale-up of complex pharmaceutical intermediates.
How to Synthesize Olatinib Maleate Efficiently
The synthesis of Olatinib Maleate via this optimized route involves four distinct operational phases that maximize atom economy and minimize waste generation. The process begins with the concurrent reduction of the carboxylic acid starting material and its condensation with the pyrrolopyrimidine core, followed immediately by esterification to lock in the structural integrity. Subsequent steps involve the strategic installation of the sulfonamide moiety through sulfonation and deprotection, followed by activation and amination. The final stage involves salt formation to stabilize the active molecule. While the chemical principles are robust, successful execution requires precise control over temperature, stoichiometry, and addition rates as detailed in the patent examples. For a comprehensive understanding of the specific operational parameters required to replicate these results in a GMP environment, please refer to the standardized synthesis guide below.
- Perform a one-pot reduction, condensation, and esterification using trans-4-[(tert-butoxycarbonyl)amino]cyclohexanecarboxylic acid and 4-chloro-7-tosyl-7H-pyrrolo[2,3-d]pyrimidine.
- Execute sulfonation and deprotection using ammonium or lithium sulfite to minimize hydrolysis and improve yield.
- Conduct chlorination with oxalyl chloride followed by methylamination using aqueous methylamine to replace hazardous THF solutions.
- Finalize the process via salification with maleic acid in 95% ethanol to obtain the pure Olatinib Maleate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this novel synthesis route offers tangible strategic benefits that extend beyond simple chemical yield. The elimination of hazardous reagents like hydrogen peroxide and n-butyllithium significantly lowers the barrier for regulatory approval and reduces the insurance and safety infrastructure costs associated with manufacturing. By replacing expensive tetrahydrofuran-based reagents with aqueous alternatives, the process inherently lowers raw material expenditure and simplifies wastewater treatment protocols, leading to substantial cost savings in environmental compliance. The reduction in unit operations—specifically the consolidation of reduction, condensation, and esterification into a single vessel—directly translates to reduced labor hours, lower energy consumption for heating and cooling cycles, and decreased equipment occupancy time. These efficiencies collectively enhance the cost reduction in pharmaceutical manufacturing, making the final API more competitive in the global marketplace.
- Cost Reduction in Manufacturing: The transition from organic solvent-based methylamine solutions to aqueous methylamine eliminates the need for costly solvent recovery systems dedicated to tetrahydrofuran. Additionally, the use of 95 percent ethanol for salification replaces the complex 1-butanol-water azeotropic distillation required in older methods. This simplification removes the energy-intensive vacuum distillation step, directly lowering utility costs and reducing the carbon footprint of the production facility without compromising product quality.
- Enhanced Supply Chain Reliability: By utilizing widely available and stable reagents such as ammonium sulfite and oxalyl chloride, the process reduces dependency on specialized or hazardous raw materials that are prone to supply disruptions. The robustness of the one-pot methodology minimizes the risk of batch failures due to intermediate handling errors, ensuring a more predictable production schedule. This reliability is crucial for reducing lead time for high-purity veterinary drug intermediates, allowing manufacturers to respond swiftly to market demand fluctuations.
- Scalability and Environmental Compliance: The avoidance of cryogenic conditions (below -30 ℃) and high-pressure hydrogenation steps found in alternative routes makes this process inherently easier to scale from pilot plant to multi-ton production. The reduction in solvent diversity and the elimination of heavy metal catalysts simplify the waste stream, facilitating easier treatment and disposal. This alignment with green chemistry principles ensures long-term sustainability and compliance with increasingly stringent environmental regulations governing pharmaceutical intermediate production.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Olatinib Maleate using the methodology described in patent CN111909156B. These insights are derived directly from the experimental data and comparative analysis provided in the intellectual property documentation. Understanding these nuances is vital for partners evaluating the feasibility of integrating this technology into their existing manufacturing portfolios.
Q: How does the new synthesis route improve safety compared to conventional methods?
A: The novel method eliminates the use of explosive hydrogen peroxide and hazardous n-butyllithium reagents found in prior art. It replaces methylamine tetrahydrofuran solutions with safer aqueous methylamine, significantly reducing fire risks and toxic wastewater generation.
Q: What are the purity and yield specifications achievable with this process?
A: According to patent CN111909156B, the optimized route achieves a total yield exceeding 65% with product purity greater than 99%. Critical impurities (A, B, and C) are strictly controlled to levels below 0.4%, 0.1%, and 0.1% respectively.
Q: Why is the switch to ammonium sulfite significant for manufacturing?
A: Replacing sodium sulfite with weakly basic ammonium or lithium sulfite prevents the hydrolysis of the methanesulfonate intermediate under basic conditions. This chemical adjustment drastically reduces side reactions, leading to higher intermediate purity and simplified downstream processing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olatinib Maleate Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this optimized synthesis are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped to analyze complex impurity profiles, guaranteeing that every batch of Olatinib Maleate meets the highest international standards for veterinary applications. Our commitment to quality assurance ensures that the high-purity Olatinib Maleate we deliver is consistent, safe, and effective for end-use formulations.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this advanced synthetic technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing processes can drive value and efficiency in your supply chain.
