Advanced Manufacturing of Olatinib Maleate: A Safer, High-Yield Synthetic Route for Veterinary Pharmaceuticals
The global veterinary pharmaceutical market is witnessing unprecedented demand for effective treatments for canine atopic dermatitis, with Olatinib Maleate standing out as a critical active pharmaceutical ingredient (API). As supply chains tighten and regulatory scrutiny on manufacturing processes intensifies, the industry requires robust, scalable, and safe synthetic methodologies. Patent CN111909156A introduces a groundbreaking preparation method that addresses the longstanding inefficiencies of prior art. This innovation leverages a streamlined "one-pot" strategy that integrates reduction, condensation, and esterification, fundamentally altering the economic and safety landscape of Olatinib production. By replacing hazardous oxidants and expensive organic solvents with safer, cost-effective alternatives, this technology offers a compelling value proposition for reliable veterinary drug supplier networks seeking to optimize their manufacturing portfolios.
The significance of this patent extends beyond mere chemical novelty; it represents a paradigm shift towards greener chemistry in fine chemical intermediates manufacturing. Traditional routes have been plagued by complex work-up procedures, low yields, and the use of dangerous reagents like red aluminum and hydrogen peroxide. In contrast, the disclosed method utilizes sodium dihydrobis(2-methoxyethoxy)aluminate for reduction and employs mild sulfites for sulfonation. This approach not only mitigates safety risks associated with exothermic reactions and explosive intermediates but also drastically simplifies the downstream processing. For procurement managers and supply chain heads, this translates to a more resilient supply source with reduced dependency on volatile raw material markets and specialized hazardous waste disposal services.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Olatinib Maleate has been hindered by several critical bottlenecks that compromise both safety and cost-efficiency. Early routes, such as those described in WO2010/020905, relied heavily on the use of hydrogen peroxide and formic acid for oxidation steps, introducing significant explosion hazards that require specialized containment infrastructure. Furthermore, these legacy processes often necessitated the use of tetrahydrofuran (THF) as a solvent for methylamination, which not only drives up raw material costs due to the high price of THF but also creates severe environmental challenges regarding wastewater treatment. The presence of THF in effluent streams demands complex recovery systems, adding to the operational expenditure (OPEX) and carbon footprint of the facility.
Another major drawback of conventional pathways is the generation of difficult-to-separate isomers. For instance, certain palladium-catalyzed hydrogenation routes produce a mixture of cis and trans isomers, where approximately 30% of the undesired cis-form cannot be easily isomerized or separated. This results in a substantial loss of raw material utilization and generates large volumes of solid waste, directly impacting the overall atom economy. Additionally, the requirement for ultra-low temperature reactions (below -30°C) using n-butyllithium in alternative routes imposes heavy energy loads on the manufacturing plant and limits the feasibility of large-scale commercial production due to the difficulty in maintaining such cryogenic conditions safely and consistently.
The Novel Approach
The innovative methodology presented in CN111909156A effectively dismantles these barriers through a cleverly designed multi-step cascade. The core of this advancement lies in the integration of reduction, condensation, and esterification into a single pot, eliminating the need for intermediate isolation and purification. This "telescoped" approach significantly reduces the number of unit operations, thereby cutting down on solvent usage, labor hours, and equipment occupancy time. By employing methylsulfonyl chloride directly in the condensation mixture, the process achieves high conversion rates without the need for the cumbersome thioacetate substitution and subsequent oxidation steps found in older patents.
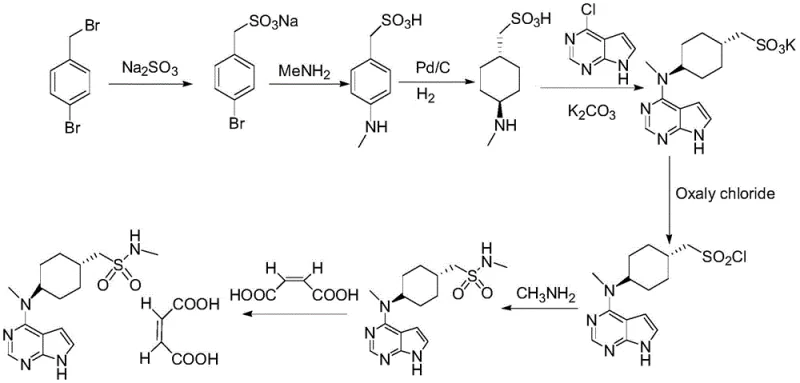
Furthermore, the novel route substitutes the hazardous hydrogen peroxide oxidation with a mild sulfonation using ammonium or lithium sulfite. This change not only enhances process safety by removing explosive reagents but also improves the yield of the sulfonic acid intermediate to over 86%. The replacement of methylamine-THF solution with a simple aqueous methylamine solution is another masterstroke of process engineering. It eliminates the need for THF entirely from this step, reducing raw material costs and simplifying wastewater treatment to basic aqueous handling. Finally, the salification step utilizes 95% ethanol instead of the traditional 1-butanol/water mixture, avoiding the energy-intensive vacuum distillation of butanol and streamlining the final crystallization process.
Mechanistic Insights into One-Pot Reduction-Condensation-Esterification
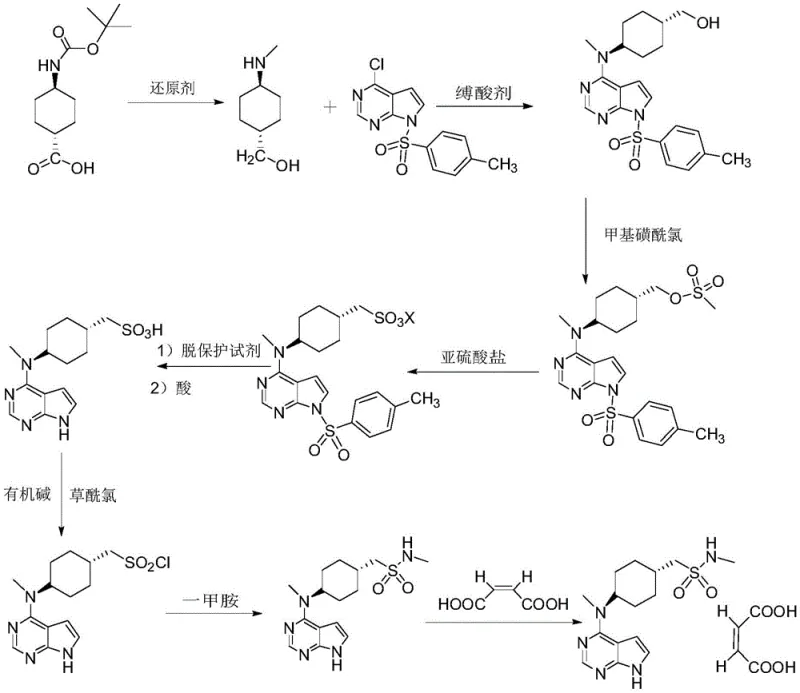
The mechanistic elegance of this synthesis begins with the reduction of trans-4-[(tert-butoxycarbonyl)amino]cyclohexanecarboxylic acid using sodium dihydrobis(2-methoxyethoxy)aluminate in toluene. Unlike lithium aluminum hydride, this reagent offers better solubility and controllability in organic solvents, allowing for a smooth reduction to the corresponding alcohol without over-reduction or side reactions. Following the reduction, the reaction mixture is directly subjected to condensation with 4-chloro-7-tosyl-7H-pyrrolo[2,3-d]pyrimidine in the presence of a base and a catalytic amount of potassium iodide. The iodide ion acts as a nucleophilic catalyst, facilitating the displacement of the chloride on the pyrimidine ring by the newly formed amine, driving the reaction to completion at moderate temperatures (65-82°C).
Crucially, the subsequent esterification with methylsulfonyl chloride occurs in the same vessel without isolating the intermediate amine. This prevents the exposure of the sensitive amine to air or moisture, which could lead to degradation or the formation of Impurity A (the sulfonic acid derivative formed by premature hydrolysis). The precise control of temperature during the dropwise addition of methylsulfonyl chloride (20-25°C) ensures that the sulfinate ester is formed selectively. The use of acetonitrile as a crystallization solvent in the work-up further purifies the product, yielding a white solid with purity exceeding 99.3%. This high purity at an early stage is vital for preventing the carry-over of impurities into the final API, ensuring that Impurity B (ethyl sulfonamide analog) and Impurity C (dimer) remain well below the 0.1% threshold.
How to Synthesize Olatinib Maleate Efficiently
The implementation of this synthesis route requires careful attention to stoichiometry and temperature control to maximize the benefits of the one-pot design. The process is divided into four distinct operational phases: the initial reduction-condensation-esterification sequence, the sulfonation-deprotection phase, the chlorination-methylamination sequence, and the final salification. Each phase is optimized to minimize solvent exchange and maximize throughput. For R&D teams looking to replicate or scale this process, the key lies in the precise quenching of the reduction step and the selection of the appropriate sulfite salt for the sulfonation step to avoid ester hydrolysis.
- Perform a one-pot reduction, condensation, and esterification using sodium dihydrobis(2-methoxyethoxy)aluminate and methylsulfonyl chloride to form the key sulfinate intermediate.
- Execute sulfonation and deprotection using ammonium or lithium sulfite in an alcohol-water system to generate the methanesulfonic acid derivative.
- Conduct chlorination with oxalyl chloride followed by methylamination using aqueous methylamine to form the sulfonamide backbone.
- Complete the synthesis by salifying the free base with maleic acid in ethanol to obtain the final Olatinib Maleate product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers transformative advantages that go beyond simple yield improvements. The most immediate impact is seen in the drastic reduction of raw material costs and waste disposal fees. By eliminating the use of tetrahydrofuran (THF) and replacing it with water and ethanol, the process removes the need for expensive solvent recovery systems and reduces the hazard classification of the waste stream. This shift significantly lowers the environmental compliance burden and allows for more flexible siting of manufacturing facilities, as the process no longer requires specialized infrastructure for handling explosive peroxides or cryogenic reagents.
- Cost Reduction in Manufacturing: The elimination of expensive reagents such as red aluminum and THF-based methylamine solutions directly lowers the Bill of Materials (BOM) cost. Furthermore, the "one-pot" nature of the initial steps reduces the number of filtration and drying operations, which translates to substantial savings in energy consumption and labor hours. The avoidance of vacuum distillation for butanol removal in the final step further contributes to lower utility costs, making the overall production economics highly favorable compared to legacy routes.
- Enhanced Supply Chain Reliability: The reliance on commodity chemicals like ammonium sulfite, aqueous methylamine, and ethanol ensures a stable and secure supply of raw materials. Unlike specialized reagents that may face supply disruptions, these inputs are widely available globally. The simplified workflow also reduces the lead time for batch production, allowing manufacturers to respond more agilely to market demand fluctuations. The high yield (>65%) ensures that less starting material is required to produce the same amount of API, buffering the supply chain against upstream price volatility.
- Scalability and Environmental Compliance: The mild reaction conditions (0-110°C) and the absence of high-pressure hydrogenation steps make this process inherently safer and easier to scale from pilot plant to multi-ton commercial production. The reduction in hazardous waste generation aligns perfectly with modern Green Chemistry principles and increasingly strict environmental regulations. This compliance advantage minimizes the risk of production shutdowns due to environmental violations and enhances the corporate sustainability profile of the manufacturing entity.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on how this method outperforms traditional approaches in terms of safety, purity, and operational efficiency.
Q: How does this new synthesis route improve safety compared to conventional methods?
A: Conventional routes often utilize explosive hydrogen peroxide for oxidation or dangerous n-butyllithium at ultra-low temperatures. This patented method replaces these hazardous reagents with milder sulfites and avoids cryogenic conditions, significantly enhancing operational safety.
Q: What are the primary cost-saving drivers in this manufacturing process?
A: The process eliminates the need for expensive tetrahydrofuran (THF) solvents by using aqueous methylamine and ethanol. Additionally, the 'one-pot' strategy reduces isolation steps, lowering labor, energy, and solvent consumption costs substantially.
Q: What is the expected purity and yield profile for commercial scale-up?
A: The optimized route achieves a total yield exceeding 65% with product purity greater than 99%. Critical impurities (A, B, and C) are tightly controlled to below 0.4%, 0.1%, and 0.1% respectively, meeting stringent regulatory standards.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Olatinib Maleate Supplier
At NINGBO INNO PHARMCHEM, we understand that the transition from laboratory innovation to commercial reality requires a partner with deep technical expertise and robust manufacturing capabilities. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in practice. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to guarantee that every batch of Olatinib Maleate meets the highest international standards for veterinary pharmaceuticals.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing process can deliver superior value, reliability, and quality for your veterinary drug portfolio.
