Advanced Copper-Catalyzed Synthesis of 2-Aminobenzimidazole Derivatives for Commercial Scale-Up
Advanced Copper-Catalyzed Synthesis of 2-Aminobenzimidazole Derivatives for Commercial Scale-Up
The landscape of heterocyclic chemistry is constantly evolving, driven by the demand for more efficient and versatile synthetic routes for bioactive scaffolds. A pivotal advancement in this domain is detailed in patent CN102060779B, which discloses a highly effective method for preparing 2-aminobenzimidazole derivatives. This technology leverages a copper-catalyzed coupling reaction between o-haloaniline compounds and carbodiimides in the presence of a basic compound. For R&D directors and procurement specialists in the pharmaceutical and agrochemical sectors, this represents a significant opportunity to access complex intermediates with improved cost-efficiency and supply chain reliability. The method is not merely a laboratory curiosity but a robust protocol capable of handling diverse substituents, thereby expanding the chemical space available for drug discovery and process development.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminobenzimidazoles has been plagued by significant operational and economic hurdles that limit their widespread industrial adoption. Traditional approaches, such as nucleophilic substitution reactions (SNAr) involving 2-chlorobenzimidazole and amines, typically necessitate extreme reaction conditions, including high temperatures and elevated pressures, which escalate energy consumption and safety risks. Moreover, the requisite starting materials for these pathways are often scarce or prohibitively expensive to source commercially, creating a bottleneck in raw material availability. Alternative strategies, such as the polymerization of o-phenylenediamine with cyanamide under microwave irradiation, while environmentally friendlier, suffer from a lack of versatility, being restricted to the preparation of only specific types of aminoimidazoles. Furthermore, routes utilizing o-fluoronitrobenzene involve lengthy multi-step sequences with reductive cyclization, resulting in cumulatively lower overall yields and increased waste generation, making them less attractive for cost-sensitive manufacturing environments.
The Novel Approach
In stark contrast to these legacy methods, the technology outlined in CN102060779B introduces a streamlined, one-pot cyclization strategy that fundamentally reshapes the production economics of these valuable heterocycles. By employing readily available o-haloaniline compounds and reacting them directly with carbodiimides under copper catalysis, this method bypasses the need for pre-functionalized benzimidazole cores or harsh nitro-reduction steps. The reaction operates under relatively mild thermal conditions, typically between 90°C and 110°C, utilizing common solvents like N-methylpyrrolidone (NMP). This approach not only simplifies the operational workflow but also dramatically broadens the scope of accessible chemical structures, allowing for the introduction of various alkyl, alkenyl, alkoxy, and halogen substituents that were previously difficult to incorporate. The result is a scientifically reasonable and highly adaptable synthetic platform that delivers high synthesis yields and products that are exceptionally easy to purify.
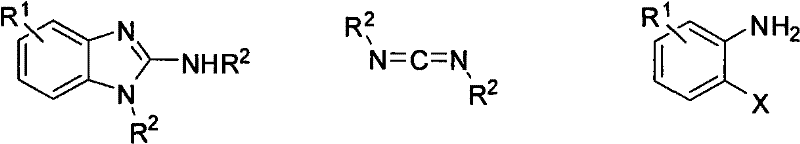
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological breakthrough lies in the intricate interplay between the copper catalyst and the basic promoter, which facilitates the formation of the benzimidazole ring through a concerted mechanism. The reaction initiates with the activation of the o-haloaniline substrate by the copper species, likely forming an organocopper intermediate that enhances the nucleophilicity of the amine nitrogen. Simultaneously, the strong base, sodium tert-butoxide (tBuONa), plays a critical role in deprotonating the amine and activating the carbodiimide electrophile. This dual activation lowers the energy barrier for the initial C-N bond formation, leading to an amidine-like intermediate. Subsequent intramolecular cyclization occurs efficiently, driven by the thermodynamic stability of the aromatic benzimidazole system. The choice of copper halides such as CuI, CuBr, or CuCl is crucial, as these catalysts provide the optimal balance of reactivity and stability, ensuring that the catalytic cycle turns over effectively without premature deactivation or the formation of inactive copper aggregates.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or high-energy thermal processes. The specificity of the copper-catalyzed pathway minimizes side reactions such as homocoupling of the aniline or hydrolysis of the carbodiimide, which are common pitfalls in less controlled environments. The use of anhydrous and oxygen-free NMP solvent further suppresses oxidative degradation of the catalyst and substrates, ensuring a cleaner reaction profile. Consequently, the crude reaction mixtures contain fewer byproducts, which simplifies the downstream purification burden. The structural integrity of sensitive functional groups, such as fluorine or bromine substituents on the aromatic ring, is preserved throughout the process, allowing for the synthesis of highly functionalized intermediates required for modern medicinal chemistry programs without the need for extensive protecting group strategies.
How to Synthesize 2-Aminobenzimidazole Derivatives Efficiently
Implementing this synthesis route in a production setting requires strict adherence to the optimized parameters defined in the patent to ensure consistent quality and yield. The process begins with the rigorous preparation of the solvent system, where NMP must be distilled over molecular sieves under high-purity nitrogen to eliminate moisture and oxygen, which are detrimental to the copper catalyst's performance. Reactants are charged in specific molar ratios, typically with a slight excess of the o-haloaniline and a stoichiometric amount of the base relative to the carbodiimide. The reaction is then sealed and heated to the preferred temperature of 110°C for a duration ranging from 16 to 72 hours, depending on the electronic nature of the substituents. Upon completion, the workup involves a straightforward aqueous quench followed by extraction with ethyl acetate, washing with saturated brine, and drying. The detailed standardized synthesis steps for specific derivatives are provided in the guide below.
- Prepare anhydrous and oxygen-free NMP solvent and mix o-haloaniline, copper catalyst (CuI/CuBr/CuCl), sodium tert-butoxide, and carbodiimide in a reactor.
- Seal the reactor and heat the mixture to 90°C-110°C, maintaining the temperature for 16 to 72 hours to ensure complete cyclization.
- Quench the reaction with water, extract with ethyl acetate, wash with brine, dry over magnesium sulfate, and purify via silica gel column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this copper-catalyzed methodology offers tangible strategic benefits that extend beyond mere technical feasibility. The primary advantage lies in the drastic simplification of the raw material portfolio; by utilizing commodity o-haloanilines and commercially available carbodiimides, manufacturers can mitigate the risks associated with sourcing specialized or proprietary starting materials. This shift significantly reduces the complexity of the supply chain, enhancing reliability and reducing lead times for high-purity pharmaceutical intermediates. Furthermore, the elimination of precious metal catalysts like palladium, which are often required in alternative cross-coupling routes, results in substantial cost savings and removes the regulatory burden of heavy metal residue testing in the final API. The robustness of the reaction conditions also implies a higher tolerance for scale-up, ensuring that batch-to-batch consistency can be maintained from pilot plant to commercial production volumes.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the replacement of expensive noble metal catalysts with abundant and inexpensive copper salts, which directly lowers the bill of materials for every kilogram produced. Additionally, the high selectivity of the reaction minimizes the formation of difficult-to-remove impurities, thereby reducing the consumption of silica gel and solvents during the purification phase. This efficiency translates into lower waste disposal costs and a reduced environmental footprint, aligning with modern green chemistry mandates. By avoiding the multi-step sequences associated with nitro-reduction routes, the overall process mass intensity is significantly improved, leading to a more lean and cost-effective manufacturing operation that maximizes asset utilization.
- Enhanced Supply Chain Reliability: The reliance on widely available building blocks such as substituted bromoanilines and dicyclohexylcarbodiimide ensures that production schedules are not held hostage by the availability of exotic reagents. These starting materials are produced by multiple global suppliers, fostering a competitive market that stabilizes pricing and guarantees continuity of supply. The simplified reaction protocol also reduces the dependency on specialized equipment capable of withstanding high pressure or extreme temperatures, allowing for production to be flexibly allocated across standard stainless steel reactors. This operational flexibility is crucial for maintaining uninterrupted supply to downstream customers, particularly in the event of market fluctuations or logistical disruptions affecting specific regions.
- Scalability and Environmental Compliance: Scaling this chemistry from gram to ton scale is facilitated by the homogeneous nature of the reaction and the absence of hazardous reagents like cyanamide or strong mineral acids. The use of NMP, while requiring careful handling, is a well-understood solvent in the industry with established recovery and recycling protocols, minimizing volatile organic compound (VOC) emissions. The high yields reported in the patent examples indicate that the process is atom-economical, generating less chemical waste per unit of product. This inherent efficiency simplifies the permitting process for new manufacturing lines and ensures compliance with increasingly stringent environmental regulations, positioning the technology as a sustainable choice for long-term commercial partnerships.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology, derived directly from the patent specifications and experimental data. Understanding these details is essential for evaluating the feasibility of integrating this route into existing production workflows. The answers reflect the specific advantages of the copper-catalyzed system over traditional methods, focusing on yield optimization, substrate scope, and purification efficiency. This information serves as a foundational resource for technical teams assessing the potential for process intensification and cost reduction in their intermediate manufacturing strategies.
Q: What are the limitations of conventional SNAr reactions for benzimidazole synthesis?
A: Conventional SNAr reactions typically require harsh conditions such as high temperature and high pressure. Furthermore, the starting materials like 2-chlorobenzimidazole are often difficult to obtain, which significantly hinders their practical application in large-scale manufacturing.
Q: Why is the copper-catalyzed method superior for diverse substituent introduction?
A: This novel approach allows for the synthesis of 2-aminobenzimidazole derivatives with various substituents that cannot be easily accessed by other methods. It utilizes readily available o-haloanilines and carbodiimides, offering a scientifically reasonable pathway with high synthesis yields.
Q: How does this process impact product purification and final purity?
A: The products obtained through this method are characterized by being easy to purify. Standard workup procedures involving extraction and column chromatography consistently yield products with purity greater than 99%, ensuring high quality for downstream pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzimidazole Derivatives Supplier
The technological potential of copper-catalyzed heterocycle synthesis is immense, yet realizing its full value requires a partner with deep process engineering expertise and a commitment to quality excellence. NINGBO INNO PHARMCHEM stands at the forefront of this capability, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art rigorous QC labs that enforce stringent purity specifications, ensuring that every batch of 2-aminobenzimidazole derivatives meets the exacting standards required by global pharmaceutical and agrochemical clients. We understand that the transition from lab-scale innovation to industrial reality demands more than just a recipe; it requires a holistic approach to process safety, impurity profiling, and regulatory compliance that we deliver consistently.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis route can be tailored to your specific project needs. By requesting a Customized Cost-Saving Analysis, you can gain clear visibility into the economic benefits of switching to this copper-catalyzed method for your supply chain. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your decision-making process. Let us collaborate to optimize your intermediate sourcing strategy, ensuring a reliable, cost-effective, and high-quality supply of critical building blocks for your next generation of therapeutic and agricultural products.
