Advanced Copper-Catalyzed Synthesis of 2-Aminobenzimidazole Derivatives for Commercial Scale-up
The pharmaceutical and agrochemical industries continuously demand efficient, scalable, and cost-effective routes for synthesizing heterocyclic scaffolds, among which the benzimidazole core stands out due to its profound biological activity. Patent CN102060779A introduces a scientifically robust methodology for the preparation of 2-aminobenzimidazole derivatives, addressing critical bottlenecks in existing synthetic pathways. This innovation leverages a copper-catalyzed cyclization strategy that reacts o-haloaniline compounds with carbodiimides in the presence of a basic compound. For R&D directors and procurement managers seeking a reliable pharmaceutical intermediate supplier, this technology represents a significant leap forward in process chemistry. By utilizing abundant copper catalysts instead of precious metals and operating under relatively mild thermal conditions, the method not only enhances reaction yields but also drastically simplifies the downstream purification workflow, ensuring a consistent supply of high-purity intermediates essential for modern drug development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 2-aminobenzimidazoles has been plagued by significant operational challenges that hinder large-scale manufacturing and cost efficiency. Traditional approaches often rely on nucleophilic aromatic substitution (SNAr) reactions between 2-chlorobenzimidazoles and amines, which typically necessitate extreme conditions such as high temperatures and elevated pressures to drive the reaction to completion. Furthermore, while palladium-catalyzed variants have been developed to mitigate some of these issues, they introduce new complexities, including the high cost of noble metal catalysts and the stringent requirement for removing trace metal residues from the final active pharmaceutical ingredients (APIs). Alternative methods involving the polymerization of o-phenylenediamine with cyanamide or multi-step reductive cyclizations from o-fluoronitrobenzenes often suffer from limited substrate scope, poor atom economy, and lengthy reaction sequences that result in lower overall yields. These inefficiencies create substantial barriers for supply chain heads who require predictable lead times and cost reduction in fine chemical manufacturing.
The Novel Approach
The methodology disclosed in CN102060779A fundamentally reshapes the synthetic landscape by employing a direct coupling of o-haloanilines and carbodiimides mediated by a copper catalyst. This novel approach eliminates the need for pre-functionalized benzimidazole starting materials or expensive palladium systems. Instead, it utilizes readily available o-haloanilines and stable carbodiimides, reacting them in a polar aprotic solvent like N-methylpyrrolidone (NMP) with sodium tert-butoxide as the base. The reaction proceeds efficiently at temperatures between 90°C and 110°C, which are easily achievable in standard stainless steel reactors without the need for specialized high-pressure equipment. This shift not only reduces capital expenditure on reactor infrastructure but also enhances safety profiles by avoiding hazardous high-pressure operations. The versatility of this system allows for the introduction of diverse substituents, enabling the synthesis of complex derivatives that were previously difficult or impossible to access through conventional routes, thereby expanding the chemical space available for medicinal chemists.
Mechanistic Insights into Copper-Catalyzed Cyclization
The core of this technological advancement lies in the intricate interplay between the copper catalyst, the base, and the substrates. The reaction initiates with the activation of the o-haloaniline by the copper species, likely forming an organocopper intermediate through oxidative addition or a similar coordination mechanism. Simultaneously, the strong base, sodium tert-butoxide, deprotonates the amine or activates the carbodiimide, facilitating the nucleophilic attack necessary for ring closure. The use of copper halides such as CuI, CuBr, or CuCl is critical, as these salts provide the optimal balance of Lewis acidity and redox potential to drive the cyclization without promoting unwanted side reactions. The molar ratio of reactants is precisely tuned, with a preferred stoichiometry of 1.0 part carbodiimide to 1.0 part o-haloaniline, 0.1 part copper catalyst, and 2.0 parts base. This specific ratio ensures that the catalytic cycle turns over efficiently while maintaining a basic environment that suppresses the formation of acidic byproducts. The solvent choice of anhydrous NMP is equally vital, as it solubilizes the ionic intermediates and stabilizes the transition states, leading to the high isolated yields observed across various substrate examples.
From an impurity control perspective, this mechanism offers distinct advantages over traditional acid-catalyzed condensations. The mild basic conditions prevent the degradation of sensitive functional groups such as esters or nitriles that might be present on the aromatic ring. Furthermore, the selectivity of the copper catalyst minimizes the formation of homocoupling byproducts or over-alkylated species that often complicate the purification of benzimidazole derivatives. The reaction pathway is designed to favor the formation of the five-membered imidazole ring directly, reducing the occurrence of linear oligomers or polymeric tars. This inherent selectivity translates to a cleaner crude reaction profile, which significantly reduces the burden on downstream purification units. For quality assurance teams, this means that achieving purity specifications greater than 99% is more attainable with fewer crystallization steps, ensuring that the final material meets the rigorous standards required for clinical trial applications and commercial API production.
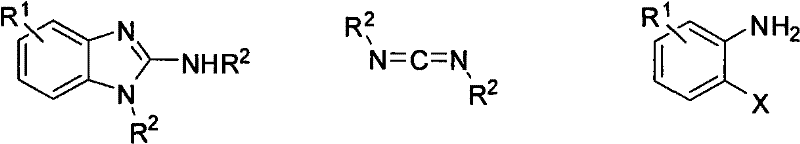
How to Synthesize 2-Aminobenzimidazole Derivatives Efficiently
Implementing this synthesis route requires careful attention to reaction conditions and reagent quality to maximize yield and reproducibility. The process begins with the rigorous drying of the solvent, NMP, which must be treated to remove moisture and oxygen, typically by refluxing over molecular sieves under a nitrogen atmosphere. This step is crucial because the presence of water can deactivate the copper catalyst or hydrolyze the carbodiimide reagent, leading to incomplete conversion. Once the anhydrous environment is established, the reagents are charged into the reactor in a specific sequence to ensure proper mixing and heat dissipation. The reaction is then heated to the optimal temperature of 110°C and maintained for approximately 24 hours, although the time can vary between 16 to 72 hours depending on the steric hindrance of the substituents. Following the reaction, a standardized workup procedure involving aqueous quenching and organic extraction is employed to isolate the product.
- Prepare the reaction mixture by combining o-haloaniline, carbodiimide, copper catalyst (CuI/CuBr/CuCl), and sodium tert-butoxide in anhydrous NMP solvent under inert atmosphere.
- Heat the sealed reactor to a temperature range of 90°C to 110°C and maintain stirring for a duration of 16 to 72 hours to ensure complete cyclization.
- Quench the reaction with water, extract the organic phase with ethyl acetate, wash with brine, dry over anhydrous magnesium sulfate, and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain leaders, the adoption of this copper-catalyzed technology offers transformative benefits that extend far beyond simple yield improvements. The primary advantage lies in the substantial cost savings achieved by replacing expensive palladium catalysts with inexpensive and abundant copper salts. This substitution not only lowers the direct material cost per kilogram but also eliminates the complex and costly heavy metal scavenging steps that are mandatory when using noble metals, thereby streamlining the entire manufacturing workflow. Additionally, the use of commercially available starting materials like o-haloanilines and carbodiimides ensures a robust and resilient supply chain, reducing the risk of raw material shortages that can plague more exotic synthetic routes. The mild reaction conditions further contribute to operational efficiency by allowing the use of standard glass-lined or stainless steel reactors, avoiding the need for specialized high-pressure vessels that require frequent certification and maintenance.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the elimination of precious metal catalysts and the simplification of the purification train. By avoiding palladium, manufacturers save significantly on catalyst procurement costs and the associated expenses of metal removal resins or activated carbon treatments. The high selectivity of the reaction reduces the generation of waste byproducts, leading to lower disposal costs and improved overall atom economy. Furthermore, the ability to achieve high purity (>99%) directly after column chromatography or crystallization minimizes the need for repetitive recrystallization cycles, saving both time and solvent consumption. These cumulative efficiencies result in a drastically simplified cost structure that enhances the competitiveness of the final pharmaceutical intermediate in the global market.
- Enhanced Supply Chain Reliability: Supply chain continuity is bolstered by the reliance on commodity chemicals that are widely produced and stocked by multiple global vendors. Unlike specialized reagents that may have long lead times or single-source dependencies, o-haloanilines and copper halides are staple items in the fine chemical industry. This diversity of supply sources mitigates the risk of production stoppages due to raw material unavailability. Moreover, the robustness of the reaction conditions means that the process is less sensitive to minor fluctuations in utility supplies or environmental conditions, ensuring consistent batch-to-batch quality. For supply chain heads, this reliability translates into predictable delivery schedules and the ability to scale production volumes rapidly in response to market demand without compromising product integrity.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial production is straightforward due to the absence of hazardous high-pressure steps and the use of standard solvents. The reaction exotherm is manageable, and the workup procedure utilizes common extraction and distillation techniques that are easily replicated in large-scale plants. From an environmental perspective, the process aligns well with green chemistry principles by reducing the use of toxic heavy metals and minimizing waste generation. The solvents used, such as NMP and ethyl acetate, can be effectively recovered and recycled through distillation, further reducing the environmental footprint. This compliance with stringent environmental regulations facilitates smoother regulatory approvals and enhances the sustainability profile of the manufacturing site, which is increasingly important for corporate social responsibility goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and beneficial effects described in the patent documentation, providing clarity on the practical aspects of the method. Understanding these details is crucial for technical teams evaluating the feasibility of integrating this route into their existing production capabilities. The answers highlight the versatility, efficiency, and robustness of the copper-catalyzed system, reinforcing its value proposition for high-volume manufacturing.
Q: What are the advantages of this copper-catalyzed method over traditional SNAr reactions?
A: Unlike traditional SNAr reactions that often require harsh high-temperature and high-pressure conditions or expensive palladium catalysts, this copper-catalyzed method operates at moderate temperatures (90°C-110°C) using abundant copper salts. It offers broader substrate tolerance for various substituents and simplifies the purification process, leading to higher isolated yields.
Q: Can this synthesis method accommodate diverse substituent groups on the benzene ring?
A: Yes, the patent demonstrates excellent functional group tolerance. The method successfully synthesizes derivatives with alkyl, alkenyl, alkoxy, and halogen substituents (fluoro, chloro, bromo) at various positions on the phenyl ring, making it highly versatile for generating diverse chemical libraries for drug discovery.
Q: Is the purification process complex for these derivatives?
A: The purification is straightforward and scalable. The protocol utilizes standard workup procedures involving aqueous quenching, ethyl acetate extraction, and silica gel column chromatography. The products are obtained as high-purity solids (>99% purity) without the need for complex recrystallization or specialized separation techniques.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 2-Aminobenzimidazole Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that high-quality intermediates play in the success of your drug development programs. As a premier CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project transitions smoothly from benchtop discovery to full-scale manufacturing. Our facilities are equipped with state-of-the-art rigorous QC labs capable of meeting stringent purity specifications, guaranteeing that every batch of 2-aminobenzimidazole derivative we deliver adheres to the highest international standards. We understand the nuances of copper-catalyzed chemistries and have optimized our processes to maximize yield while minimizing impurities, providing you with a reliable source of material that accelerates your timeline to market.
We invite you to collaborate with us to leverage this advanced synthetic technology for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements, demonstrating how this route can optimize your budget without sacrificing quality. Please contact us today to request specific COA data and route feasibility assessments. By partnering with NINGBO INNO PHARMCHEM, you gain access to not just a chemical supplier, but a strategic ally committed to driving efficiency and innovation in your supply chain.
