Scalable Treprostinil Manufacturing: A Technical Breakthrough in Prostacyclin Derivative Synthesis
The pharmaceutical landscape for pulmonary arterial hypertension treatments relies heavily on the consistent supply of high-purity prostacyclin derivatives, specifically Treprostinil, the active ingredient in Remodulin®. Patent CN103274926A discloses a transformative methodology that addresses the longstanding bottlenecks in the commercial synthesis of this complex molecule. By shifting away from traditional chromatographic purification towards a robust salt-formation strategy, this intellectual property offers a pathway to significantly enhanced process efficiency and product quality. For R&D directors and procurement specialists alike, understanding the nuances of this improved process is critical for securing a reliable supply chain. The core innovation lies in the strategic conversion of the intermediate carboxylic acid into a diethanolamine salt, which serves as a pivotal purification handle, allowing for the removal of trace impurities that are notoriously difficult to eliminate in the free acid form.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Treprostinil and related prostacyclin analogs has been plagued by reliance on column chromatography for the purification of key intermediates, particularly the nitrile precursor. This traditional approach presents severe disadvantages when transitioning from laboratory scale to commercial manufacturing. Column chromatography is inherently batch-limited, requiring massive volumes of flammable organic solvents such as ethyl acetate and hexanes, which escalates both operational costs and environmental waste disposal burdens. Furthermore, the use of silica gel introduces the risk of product loss through irreversible adsorption and potential contamination with silicate particulates. For a high-value API intermediate, the low throughput and high labor intensity of chromatographic separation create a fragile supply chain that is susceptible to delays and quality variability. The viscosity of the free acid intermediates often complicates handling, making large-scale processing inefficient and prone to inconsistencies in yield and purity profiles.
The Novel Approach
The methodology outlined in the patent data fundamentally reengineers the downstream processing by introducing a crystallization-driven purification protocol. Instead of struggling with viscous oils, the process converts the crude Treprostinil acid into a crystalline diethanolamine salt. This transformation is not merely a formulation change but a strategic purification step. The salt formation allows for the exploitation of solubility differences between the desired product and impurities generated during the upstream alkylation and hydrolysis steps. By seeding the solution with specific polymorphs (Polymorph B) and controlling the cooling profile, manufacturers can induce the precipitation of highly pure crystals while leaving impurities in the mother liquor. This approach drastically simplifies the operation, replacing complex chromatography columns with standard filtration and drying equipment, thereby enhancing the robustness and reproducibility of the manufacturing process suitable for multi-kilogram production.
This strategic pivot enables the production of Treprostinil with purity levels consistently exceeding 99.8% as determined by HPLC area under the curve (AUC) analysis. The elimination of chromatography also means a substantial reduction in the overall solvent footprint, aligning the manufacturing process with modern green chemistry principles and regulatory expectations for environmental compliance. For supply chain heads, this translates to a more predictable lead time and reduced dependency on specialized purification consumables like silica gel.
Mechanistic Insights into Alkylation and Salt-Mediated Purification
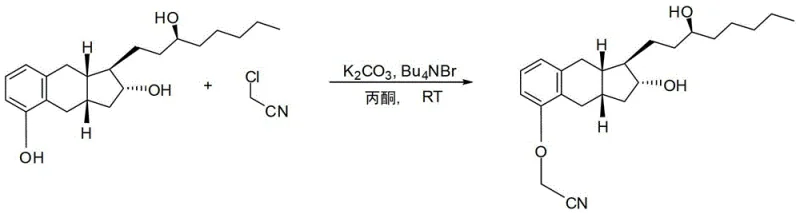
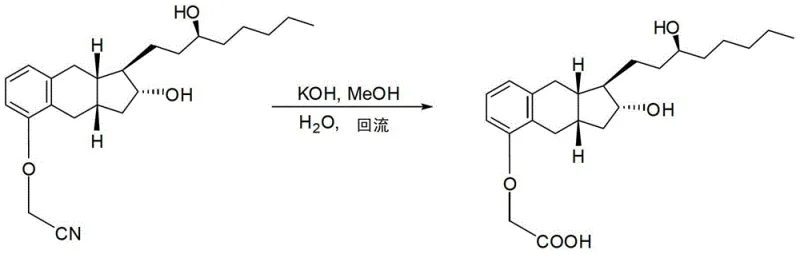
The chemical foundation of this improved process begins with the O-alkylation of the benzoindene triol core. In this critical step, the phenolic hydroxyl group is selectively alkylated using chloroacetonitrile in the presence of potassium carbonate and a phase transfer catalyst, tetrabutylammonium bromide. The reaction is conducted in acetone at ambient temperatures ranging from 23°C to 25°C, which minimizes thermal degradation of the sensitive tricyclic core. The use of a phase transfer catalyst ensures efficient interaction between the inorganic base and the organic substrate, driving the reaction to completion over a period of 16 to 72 hours. Following alkylation, the nitrile group undergoes base-catalyzed hydrolysis using potassium hydroxide in a methanol-water mixture under reflux conditions at approximately 72°C. This step converts the nitrile functionality into the requisite carboxylic acid side chain, completing the construction of the Treprostinil backbone.
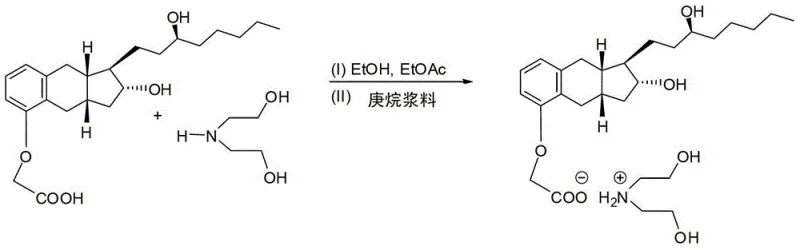
However, the true mechanistic brilliance lies in the subsequent salt formation. Upon acidification of the hydrolysis mixture, the crude Treprostinil acid is treated with diethanolamine in a solvent system comprising ethanol and ethyl acetate. The stoichiometry is carefully controlled to form a 1:1 salt. The introduction of seed crystals of Treprostinil diethanolamine salt Polymorph B initiates controlled nucleation. As the suspension is cooled from roughly 60°C down to 20°C over an extended period, the crystal lattice selectively incorporates the Treprostinil-diethanolamine complex while excluding structurally similar impurities. This crystallization mechanism acts as a highly effective filter at the molecular level. The resulting solid can be isolated via filtration and washed with cold solvents to further enhance purity. Finally, the pure salt is converted back to the free acid form through simple acidification with dilute hydrochloric acid in a biphasic system, yielding the final API intermediate with exceptional quality attributes.
How to Synthesize Treprostinil Efficiently
Implementing this synthesis route requires precise control over reaction parameters and crystallization kinetics to maximize yield and purity. The process is designed to be telescoped where possible, minimizing the isolation of unstable intermediates. Detailed standard operating procedures regarding reagent grades, addition rates, and specific temperature ramps are essential for successful technology transfer. The following guide outlines the critical operational phases derived from the patent examples, serving as a framework for process engineers to establish robust manufacturing protocols.
- Alkylate benzoindene triol with chloroacetonitrile using potassium carbonate and tetrabutylammonium bromide in acetone at room temperature.
- Hydrolyze the resulting nitrile intermediate using potassium hydroxide in methanol and water under reflux conditions to form the carboxylic acid.
- Convert the crude acid into a diethanolamine salt in ethanol/ethyl acetate, seed with polymorph B, and crystallize to remove impurities before final acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this salt-based purification technology offers compelling economic and logistical benefits that extend beyond simple yield improvements. The primary value driver is the drastic simplification of the purification train. By removing the requirement for large-scale column chromatography, the process eliminates a major bottleneck that typically limits batch size and extends cycle times. This structural change in the manufacturing workflow directly correlates to increased production capacity without the need for significant capital expenditure on new equipment. Furthermore, the reduction in solvent usage—specifically the avoidance of large volumes of eluents required for chromatography—leads to substantial cost savings in raw material procurement and waste treatment. The ability to store the intermediate as a stable salt also provides flexibility in production scheduling, allowing manufacturers to decouple the synthesis of the core structure from the final isolation steps.
- Cost Reduction in Manufacturing: The elimination of silica gel chromatography removes a significant variable cost associated with consumables and solvent recovery. In traditional processes, the volume of solvent required for column loading, elution, and column regeneration is immense. By switching to a crystallization-based purification, the solvent demand is reduced to levels manageable by standard distillation and recovery units. Additionally, the labor hours required to pack columns, monitor fractions, and pool cuts are completely eradicated. This operational efficiency translates into a lower cost of goods sold (COGS), making the final Treprostinil intermediate more price-competitive in the global market. The higher purity achieved also reduces the risk of batch rejection due to specification failures, further protecting profit margins.
- Enhanced Supply Chain Reliability: Supply continuity is often threatened by the complexity of purification steps. Chromatography is prone to channeling, pressure fluctuations, and inconsistent separation performance, which can lead to batch failures or extended processing times. The crystallization method described in the patent is inherently more robust and scalable. Crystallizers and filters are standard unit operations found in almost any multipurpose chemical plant, reducing the dependency on specialized facilities. Moreover, the stability of the diethanolamine salt intermediate allows for inventory buffering. Manufacturers can produce the salt in large batches and hold it in storage, releasing it for final conversion to the free acid based on demand. This decoupling enhances the agility of the supply chain, ensuring reliable delivery schedules for downstream API manufacturers.
- Scalability and Environmental Compliance: Scaling a chromatographic process from grams to kilograms often involves non-linear challenges regarding flow dynamics and bed height. In contrast, crystallization scales linearly; the physics of nucleation and growth remain consistent regardless of vessel size, provided mixing and cooling rates are maintained. This makes the technology ideal for commercial scale-up of complex pharmaceutical intermediates. From an environmental perspective, the reduction in hazardous waste generation is significant. Less solvent waste means a lower carbon footprint and reduced regulatory burden regarding VOC emissions and hazardous waste disposal. This alignment with green chemistry initiatives not only improves corporate sustainability metrics but also mitigates the risk of regulatory shutdowns or fines associated with environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this improved Treprostinil synthesis. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the salt-mediated purification strategy.
Q: How does this new process improve purity compared to traditional methods?
A: The process eliminates the need for column chromatography purification of the nitrile intermediate. Instead, it utilizes a diethanolamine salt crystallization step which effectively removes impurities carried over from alkylation and hydrolysis, achieving purity levels exceeding 99.8% by HPLC.
Q: What are the specific reaction conditions for the alkylation step?
A: The alkylation of benzoindene triol is conducted at room temperature (23±2°C) using acetone as the solvent, potassium carbonate as the base, and tetrabutylammonium bromide as a phase transfer catalyst. The reaction typically proceeds for 16 to 72 hours.
Q: Why is the diethanolamine salt formation critical for scalability?
A: Forming the diethanolamine salt allows for the isolation of a stable solid intermediate that can be stored and purified via recrystallization. This avoids the handling of viscous oils typical of the free acid form and significantly reduces solvent consumption compared to chromatographic purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Treprostinil Supplier
The technical advancements detailed in patent CN103274926A underscore the complexity and sophistication required to manufacture high-purity prostacyclin derivatives efficiently. At NINGBO INNO PHARMCHEM, we possess the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production necessary to bring such innovative processes to life. Our facility is equipped with state-of-the-art reactors and purification systems capable of handling the specific solvent systems and temperature controls required for this salt-formation methodology. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch of Treprostinil intermediate meets the exacting standards required for pharmaceutical applications. Our commitment to quality assurance ensures that the theoretical benefits of this patent are realized in every gram of product we deliver.
We invite global partners to leverage our technical expertise to optimize their supply chains. By collaborating with us, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage you to contact our technical procurement team to request specific COA data and route feasibility assessments. Whether you require clinical trial materials or commercial-scale API intermediates, our team is ready to demonstrate how our advanced manufacturing capabilities can secure your supply of this critical cardiovascular therapeutic.
