Scalable Manufacturing of High-Purity Treprostinil via Salt-Mediated Purification
Introduction to Patented Treprostinil Synthesis
The pharmaceutical landscape for pulmonary arterial hypertension treatments relies heavily on the consistent supply of high-quality prostacyclin derivatives, specifically Treprostinil, the active ingredient in Remodulin®. A pivotal advancement in this domain is detailed in Chinese Patent CN103274926A, which discloses an improved process for preparing Treprostinil and its intermediates. This technology represents a significant departure from traditional synthesis routes that often rely on cumbersome purification techniques. By introducing a novel salt-mediated purification strategy, the process addresses critical bottlenecks in manufacturing efficiency and product quality. For R&D directors and procurement specialists, understanding this methodology is essential for securing a reliable pharmaceutical intermediate supplier capable of meeting stringent regulatory standards. The core innovation lies in the conversion of the crude acid into a stable diethanolamine salt, which facilitates high-purity isolation without the need for column chromatography.
This technical insight report analyzes the mechanistic advantages and commercial implications of this patented route. The method not only enhances the purity profile of the final Active Pharmaceutical Ingredient (API) but also streamlines the supply chain by reducing dependency on complex separation technologies. As the demand for cardiovascular therapies grows, the ability to scale such processes from kilogram to multi-ton levels becomes a decisive factor for market competitiveness. The following sections provide a deep dive into the chemical transformations, comparing conventional limitations with this novel approach, and outlining the tangible benefits for global supply chains.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of complex prostacyclin analogues like Treprostinil has been plagued by purification challenges that hinder commercial viability. Traditional routes often necessitate the use of silica gel column chromatography to isolate intermediates and the final product. While effective on a laboratory scale, column chromatography is notoriously difficult to scale up for industrial production due to high solvent consumption, significant waste generation, and the risk of product degradation on silica. Furthermore, the reliance on chromatographic purification introduces variability in batch-to-batch consistency and increases the potential for metal contamination from stationary phases. These factors collectively drive up the cost of goods sold (COGS) and extend lead times, creating vulnerabilities in the commercial scale-up of complex pharmaceutical intermediates. The environmental footprint associated with disposing of large volumes of chromatographic eluents also poses compliance challenges under increasingly strict environmental regulations.
The Novel Approach
The improved process described in the patent circumvents these issues by leveraging the physicochemical properties of Treprostinil salts. Instead of chromatography, the method employs a strategic salt formation step using diethanolamine. This transformation converts the oily or semi-solid crude acid into a crystalline salt that can be purified through recrystallization. This shift from chromatography to crystallization is a game-changer for cost reduction in API intermediate manufacturing. The process involves alkylating a benzoindene triol with chloroacetonitrile, followed by hydrolysis to the acid, and finally, salt formation. The crystalline nature of the diethanolamine salt allows for the efficient exclusion of impurities into the mother liquor. Subsequent acidification regenerates the free acid with exceptional purity. This approach not only simplifies the operational workflow but also enhances the robustness of the manufacturing process, making it highly attractive for large-scale production.
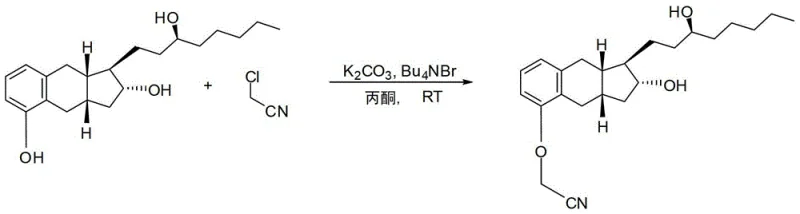
Mechanistic Insights into Salt-Mediated Purification
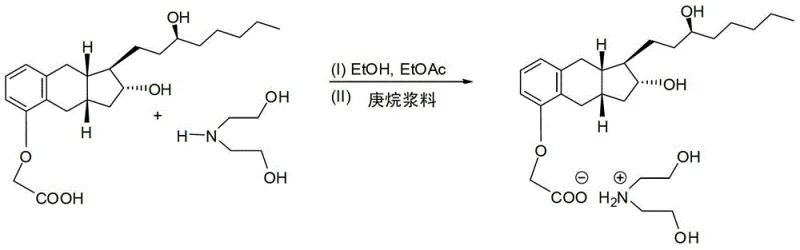
The chemical elegance of this process lies in the specific interaction between the carboxylic acid moiety of Treprostinil and the amine functionality of diethanolamine. Upon reacting the crude Treprostinil acid with diethanolamine in a solvent system comprising ethanol and ethyl acetate, a 1:1 salt is formed. This salt exhibits distinct solubility characteristics compared to the free acid and associated impurities. The patent specifies heating the mixture to 60°C to 75°C to ensure complete dissolution, followed by controlled cooling and seeding with polymorph B crystals. This seeding step is critical for controlling the crystal lattice formation, ensuring that the desired polymorph precipitates out while impurities remain in solution. The use of a phase transfer catalyst like tetrabutylammonium bromide in the initial alkylation step further ensures high conversion rates, minimizing the formation of side products that could complicate downstream purification.
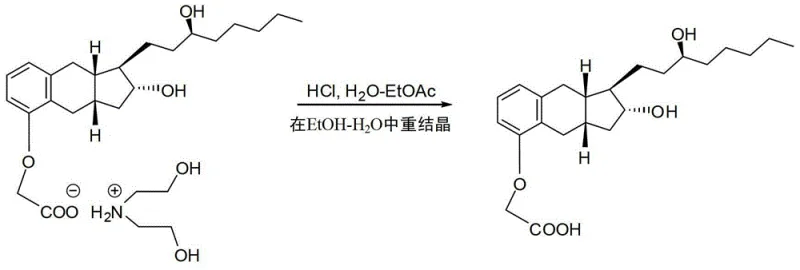
Impurity control is inherently built into the crystallization dynamics. During the hydrolysis of the nitrile intermediate, various byproducts may form, including unreacted starting materials or partially hydrolyzed species. However, the subsequent salt formation acts as a 'chemical filter.' The crystal lattice of the treprostinil diethanolamine salt is highly selective, rejecting molecules that do not fit its structural geometry. This phenomenon is evidenced by the patent data showing purity levels reaching 99.8% by HPLC after just one crystallization cycle. Moreover, the stability of the salt form allows for storage and transport without degradation, providing flexibility in the supply chain. The final regeneration of the free acid via acidification with dilute HCl is a straightforward aqueous workup that avoids harsh conditions, preserving the integrity of the sensitive prostacyclin core structure.
How to Synthesize Treprostinil Efficiently
The synthesis of Treprostinil via this improved route is a testament to process chemistry optimization. It begins with the alkylation of benzoindene triol using chloroacetonitrile in the presence of potassium carbonate and a catalytic amount of tetrabutylammonium bromide in acetone. This reaction proceeds at ambient temperature (23±2°C) over 16 to 72 hours, yielding the nitrile intermediate which is used directly without isolation. The second stage involves the hydrolysis of this nitrile using potassium hydroxide in a methanol-water mixture under reflux conditions (approx. 72°C) for about 5 hours. The resulting crude acid is then subjected to the critical salt formation step with diethanolamine. Detailed standardized operating procedures for each reaction stage, including exact stoichiometry and temperature profiles, are essential for reproducibility.
- Alkylate benzoindene triol with chloroacetonitrile using K2CO3 and Bu4NBr in acetone at room temperature to form the nitrile intermediate.
- Hydrolyze the nitrile intermediate using KOH in methanol/water under reflux conditions to generate crude Treprostinil acid.
- React the crude acid with diethanolamine in ethanol/ethyl acetate to form a crystalline salt, then regenerate the pure free acid via acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from chromatographic purification to salt-mediated crystallization offers profound economic and logistical benefits. The elimination of silica gel columns removes a major bottleneck in production throughput. Chromatography is not only slow but also requires significant infrastructure investment in terms of columns, pumps, and solvent recovery systems. By replacing this with simple filtration and crystallization vessels, the capital expenditure (CAPEX) for manufacturing facilities is drastically reduced. Furthermore, the operational expenditure (OPEX) is lowered due to the decreased consumption of high-purity solvents typically required for gradient elution in chromatography. This efficiency translates directly into a more competitive pricing structure for the final high-purity pharmaceutical intermediate.
- Cost Reduction in Manufacturing: The primary driver for cost savings in this process is the removal of column chromatography. Silica gel is a consumable cost that adds up quickly at scale, and the solvents required to flush columns are substantial. By utilizing crystallization, the process relies on cheaper solvents like ethyl acetate and heptane, which are easier to recover and recycle. Additionally, the labor hours associated with packing columns and monitoring fractions are eliminated. The patent explicitly notes that this method saves a significant amount of solvent and labor in intermediate purification. This lean manufacturing approach ensures that the cost per kilogram of Treprostinil is optimized, allowing for better margin management in a competitive generic drug market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the robustness of the intermediates. The treprostinil diethanolamine salt is stable and can be stored at room temperature, acting as a buffer inventory if downstream processing faces delays. Unlike the free acid, which may be prone to oxidation or degradation, the salt form provides a stable checkpoint in the manufacturing timeline. This stability reduces the risk of batch failures due to material degradation during storage or transit. Moreover, the reagents used—such as chloroacetonitrile, potassium carbonate, and diethanolamine—are commodity chemicals with reliable global availability, minimizing the risk of raw material shortages that could disrupt production schedules.
- Scalability and Environmental Compliance: Scaling a chromatographic process from grams to tons is non-linear and fraught with engineering challenges. In contrast, crystallization scales linearly; a 50-L reactor operates on the same principles as a 5000-L reactor. The patent demonstrates successful execution on a 5 kg scale, proving the feasibility of this route for commercial production. From an environmental perspective, the reduction in solvent waste aligns with Green Chemistry principles. Less waste means lower disposal costs and a smaller carbon footprint, which is increasingly important for pharmaceutical companies aiming to meet sustainability goals. The process generates less hazardous waste, simplifying compliance with environmental regulations and reducing the administrative burden of waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis route. Understanding these details is crucial for technical teams evaluating the feasibility of adopting this process for their own manufacturing lines or for procurement teams assessing supplier capabilities. The answers are derived directly from the experimental data and claims presented in the patent documentation, ensuring accuracy and relevance to real-world production scenarios.
Q: Why is the diethanolamine salt formation critical in this process?
A: The formation of the treprostinil diethanolamine salt allows for purification via crystallization rather than column chromatography. This step effectively removes impurities carried over from the alkylation and hydrolysis steps, resulting in a final product purity exceeding 99.8% while significantly reducing solvent usage and processing time.
Q: What are the purity specifications achievable with this method?
A: According to the patent data, this improved process consistently yields Treprostinil with a purity of at least 99.0%, with specific batches demonstrating 99.8% purity by HPLC (AUC). The method eliminates the need for silica gel chromatography, which often introduces variability and metal contaminants.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process is designed for scalability. The patent describes successful implementation on a 5 kg scale, utilizing standard reactors (50-L to 72-L) and avoiding specialized chromatography equipment. The use of common solvents like acetone, ethyl acetate, and ethanol further supports easy scale-up for industrial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Treprostinil Supplier
The technical sophistication required to execute this salt-mediated purification process demands a partner with deep expertise in process development and scale-up. NINGBO INNO PHARMCHEM stands at the forefront of this capability, offering extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facilities are equipped with state-of-the-art reactors and filtration systems capable of handling the specific thermal and solvent requirements of the Treprostinil synthesis, such as the precise temperature control needed for polymorph seeding. We maintain stringent purity specifications and operate rigorous QC labs to ensure that every batch meets the >99.8% purity benchmark established by the patent, guaranteeing safety and efficacy for the final drug product.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced manufacturing technology. By partnering with NINGBO INNO PHARMCHEM, you gain access to a Customized Cost-Saving Analysis tailored to your specific volume requirements. Our technical procurement team is ready to provide specific COA data and route feasibility assessments to demonstrate how this improved process can optimize your supply chain. Contact us today to discuss how we can support your clinical and commercial needs with high-quality, cost-effective Treprostinil intermediates.
