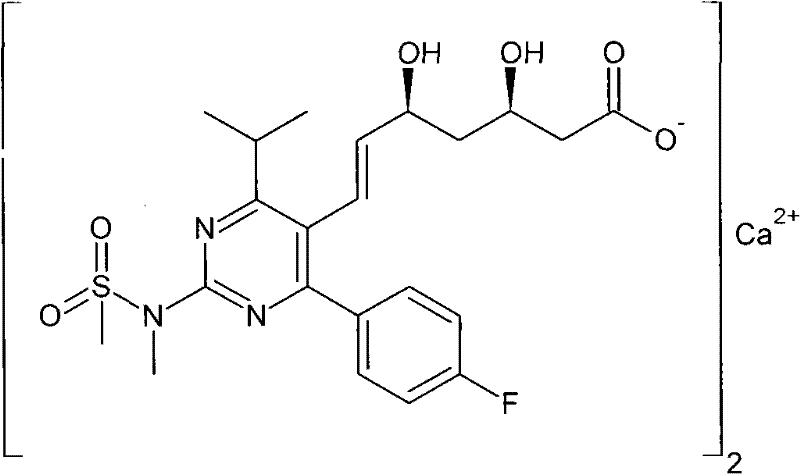
Scalable Synthesis of Rosuvastatin Calcium Intermediate via Optimized Grignard and Wittig Pathways
Scalable Synthesis of Rosuvastatin Calcium Intermediate via Optimized Grignard and Wittig Pathways
The pharmaceutical landscape for cardiovascular therapeutics continues to demand robust, cost-effective supply chains for high-value active pharmaceutical ingredients (APIs) and their precursors. Patent CN101735272A discloses a highly efficient seven-step synthetic methodology for preparing a critical rosuvastatin calcium intermediate, specifically compounds of general formula I where R is a C1-C10 alkyl group and Y represents a hydroxyl protecting group. This technical breakthrough addresses significant bottlenecks in the production of HMG-CoA reductase inhibitors by utilizing readily available starting materials such as vinyl chloride and (R)-epichlorohydrin. The disclosed route offers a compelling alternative to traditional methods by integrating a copper-catalyzed Grignard coupling, nucleophilic cyanide substitution, and a terminal Wittig olefination to construct the complex side chain with high stereochemical fidelity. For R&D directors and procurement specialists, understanding the nuances of this pathway is essential for securing a reliable rosuvastatin calcium intermediate supplier capable of meeting stringent purity specifications while optimizing manufacturing expenditures.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Prior to this innovation, the synthesis of key intermediates like (3R)-tert-butyldimethylsilyloxy-5-oxo-6-triphenylphosphine alkene capronate faced substantial economic and technical hurdles. Existing literature, such as PCT application WO/2006/091771, describes routes necessitating bulky carboxy protecting groups to ensure ester stability, which inadvertently complicates the selective mono-methyl hydrolysis in early stages. When smaller protecting groups like methyl are employed to simplify the structure, the selectivity of hydrolysis drops precipitously, leading to poor yields and difficult purification challenges that render the process economically unviable for large-scale operations. Furthermore, alternative approaches documented in journals like the Journal of Organic Chemistry rely on expensive anhydride compounds as starting materials. These anhydrides not only drive up the raw material costs significantly but also suffer from low reaction yields in the initial steps, creating a compounding negative effect on the overall process mass intensity. Such inefficiencies result in higher waste generation and increased environmental burden, making these conventional pathways unsuitable for modern, green chemistry-compliant manufacturing facilities seeking cost reduction in pharmaceutical intermediates manufacturing.
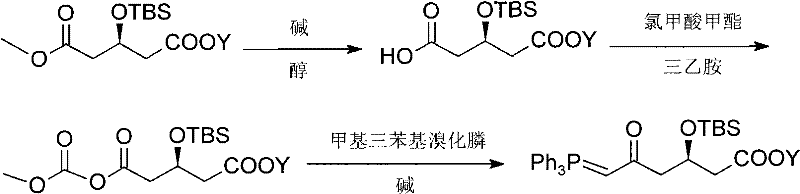
The Novel Approach
The methodology outlined in CN101735272A fundamentally reengineers the synthetic logic by starting from commodity chemicals, thereby bypassing the cost and complexity associated with specialized anhydrides or cumbersome protecting group manipulations. The novel route initiates with the formation of a vinyl Grignard reagent from vinyl chloride and magnesium metal, which is then coupled with (R)-epichlorohydrin under cuprous chloride catalysis. This strategic choice allows for the rapid construction of the carbon backbone with the correct chirality established early in the sequence. Subsequent steps involve a straightforward nucleophilic substitution with sodium cyanide, followed by alcoholysis to install the ester functionality directly. The process avoids the pitfalls of selective hydrolysis seen in prior art by designing the molecule such that the ester group is introduced in a stable form that does not require later removal or modification. The final stages employ a robust oxidation using potassium permanganate and a classic Wittig reaction to install the terminal alkene-phosphonium moiety. This streamlined approach ensures mild reaction conditions, typically ranging from cryogenic temperatures for sensitive steps to moderate heating for others, facilitating easier thermal management and safer operation in a commercial plant setting.
Mechanistic Insights into Cu-Catalyzed Grignard Coupling and Wittig Olefination
The cornerstone of this synthesis lies in the precise control of stereochemistry and functional group tolerance during the carbon-carbon bond-forming events. The initial Grignard reaction involves the generation of vinyl magnesium chloride, which acts as a potent nucleophile. However, direct addition to epoxides can often lead to regio-irregularities. The inclusion of cuprous chloride (CuCl) as a catalyst is critical; it likely forms a higher-order cuprate species in situ that directs the nucleophilic attack specifically to the less hindered carbon of the (R)-epichlorohydrin ring, ensuring the formation of (2R)-1-chloro-2-hydroxyl-4-pentene with high regioselectivity. The reaction is conducted in tetrahydrofuran (THF), a solvent that stabilizes the Grignard reagent and facilitates the coordination of the copper catalyst. Temperature control is paramount here, with the reaction initiated at low temperatures (-35°C to -25°C) to suppress side reactions such as homocoupling of the Grignard reagent or polymerization of the vinyl chloride, before being allowed to warm slightly to drive completion. This careful modulation of reaction parameters ensures that the chiral center derived from the epoxide is preserved without racemization, a critical quality attribute for the final API.
In the final transformation, the conversion of the keto-ester intermediate to the target phosphonium salt via the Wittig reaction demonstrates excellent chemoselectivity. The process involves the generation of a phosphorus ylide from methyltriphenylphosphonium bromide using n-butyl lithium as a strong base. This ylide then attacks the ketone carbonyl of the intermediate (Formula V), formed via the activation of the carboxylic acid with methyl chloroformate. The use of N-methylmorpholine as a base in the preceding acylation step is particularly noteworthy; it acts as a non-nucleophilic base that scavenges the HCl generated during mixed anhydride formation without attacking the sensitive ester or silyl ether functionalities. The subsequent Wittig olefination proceeds under strictly anhydrous conditions at low temperatures (-60°C to -55°C) to favor the formation of the desired alkene geometry and prevent ylide decomposition. The final product is isolated as a crystalline solid after rigorous washing and recrystallization, achieving an enantiomeric excess (ee) of greater than 99%, which underscores the robustness of the stereochemical control throughout the seven-step sequence.
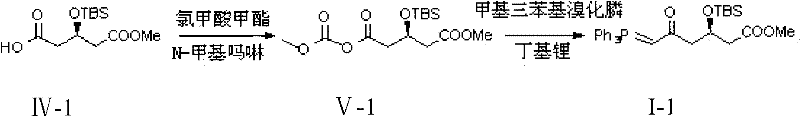
How to Synthesize Rosuvastatin Calcium Intermediate Efficiently
The synthesis of this high-value intermediate requires strict adherence to the optimized protocol detailed in the patent to ensure maximum yield and purity. The process is divided into distinct operational units: Grignard formation, chain extension via cyanide, esterification, protection, oxidation, activation, and olefination. Each step has been fine-tuned regarding stoichiometry, solvent choice, and temperature profiles to minimize byproduct formation. For instance, the oxidation step utilizes potassium permanganate in an acetone-water mixture, a cost-effective oxidant system that avoids the use of heavy metal catalysts like chromium or ruthenium, simplifying downstream waste treatment. The detailed standardized synthesis steps, including specific molar ratios such as the 0.10:1 ratio of cuprous chloride to epichlorohydrin and the 3.0:1 ratio of permanganate to substrate, are critical for reproducibility. Operators must ensure rigorous exclusion of moisture during the Grignard and Wittig steps, while the aqueous workups in the intermediate stages are designed to efficiently remove inorganic salts and polar impurities.
- Prepare vinyl magnesium chloride via Grignard reaction and couple with (R)-epichlorohydrin using cuprous chloride catalyst to form (2R)-1-chloro-2-hydroxyl-4-pentene.
- Perform nucleophilic substitution with sodium cyanide followed by alcoholysis to generate the protected ester intermediate.
- Execute hydroxyl protection, potassium permanganate oxidation, mixed anhydride formation, and final Wittig olefination to yield the target phosphonium salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this synthetic route offers transformative benefits for supply chain stability and cost management. By shifting the starting material base from specialized, high-cost anhydrides to bulk commodities like vinyl chloride and magnesium, the raw material cost profile is drastically improved. Vinyl chloride is a petrochemical derivative produced on a massive global scale, ensuring consistent availability and shielding the manufacturing process from the supply volatility often associated with fine chemical building blocks. Furthermore, the elimination of complex protecting group strategies reduces the total number of unit operations and the consumption of auxiliary reagents, directly translating to lower operational expenditures. The process is designed with scalability in mind; the use of common solvents such as dichloromethane, THF, and ethyl acetate simplifies solvent recovery and recycling systems, further enhancing the economic efficiency of the production line. This alignment with green chemistry principles also mitigates regulatory risks associated with hazardous waste disposal.
- Cost Reduction in Manufacturing: The replacement of expensive anhydride starting materials with low-cost vinyl chloride and (R)-epichlorohydrin results in substantial raw material savings. Additionally, the avoidance of bulky protecting groups eliminates the need for specific hydrolysis steps that typically suffer from low yields, thereby improving the overall throughput of the plant. The use of potassium permanganate as an oxidant instead of precious metal catalysts removes the cost burden of metal recovery and validation, while the high yields reported in the embodiments (often exceeding 80% for key steps) maximize the utilization of input materials. These factors combine to create a significantly leaner cost structure compared to legacy processes.
- Enhanced Supply Chain Reliability: Sourcing reliability is bolstered by the use of universally available reagents. Unlike specialized chiral building blocks that may have limited suppliers, the primary inputs for this route are produced by multiple major chemical manufacturers globally. This diversification of the supply base reduces the risk of single-source dependency and potential disruptions. Moreover, the process stability described in the patent, characterized by mild conditions and robust intermediates, ensures consistent batch-to-batch quality. This predictability allows for more accurate production planning and inventory management, reducing the need for excessive safety stock and enabling a more responsive supply chain capable of meeting fluctuating market demands for rosuvastatin calcium.
- Scalability and Environmental Compliance: The process is inherently scalable due to its reliance on standard unit operations such as extraction, distillation, and crystallization, which are well-understood in industrial engineering. The waste profile is favorable; the primary inorganic byproducts are magnesium and copper salts, which are easier to treat and dispose of compared to heavy metal waste from other oxidation methods. The high atom economy of the Grignard and Wittig steps, combined with efficient solvent recovery protocols, minimizes the environmental footprint. This compliance with environmental standards facilitates smoother regulatory approvals and reduces the long-term liability associated with waste management, making the facility more sustainable and socially responsible.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific experimental data and comparative analysis provided in the patent documentation. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer or scale-up. The answers reflect the empirical results obtained during the optimization of the seven-step sequence, highlighting the reproducibility and robustness of the method under varying conditions.
Q: What are the key advantages of this synthesis route over prior art methods?
A: Unlike prior art requiring expensive anhydride starting materials or bulky protecting groups that complicate hydrolysis, this method utilizes low-cost commodity chemicals like vinyl chloride and achieves high yields through mild, stable reaction conditions suitable for large-scale production.
Q: How is stereochemical integrity maintained during the synthesis?
A: The process relies on (R)-epichlorohydrin as the chiral source. The subsequent nucleophilic substitution and protection steps are designed to proceed with inversion or retention as required, ensuring the final product maintains high enantiomeric excess (ee > 99%).
Q: Is this process suitable for industrial scale-up?
A: Yes, the patent explicitly highlights the use of common solvents (THF, DCM), manageable temperature ranges (-60°C to 60°C), and straightforward workup procedures (extractions, crystallization), making it highly adaptable for commercial manufacturing environments.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Rosuvastatin Calcium Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the production of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and efficient. We are committed to delivering products that meet stringent purity specifications, utilizing our rigorous QC labs to verify every batch against the highest industry standards. Our facility is equipped to handle the specific requirements of this synthesis, including the safe handling of Grignard reagents and the precise temperature control needed for the Wittig olefination, guaranteeing a consistent supply of high-purity rosuvastatin calcium intermediates for your global operations.
We invite you to collaborate with us to optimize your supply chain and reduce your overall manufacturing costs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals in the pharmaceutical sector.
