Advanced Synthesis of Ezetimibe Intermediates: A Titanium-Catalyzed Route for Industrial Scale-Up
Introduction to Novel Ezetimibe Synthesis Technologies
The pharmaceutical landscape for cholesterol management has been significantly shaped by the development of Ezetimibe, a potent selective cholesterol absorption inhibitor. As demand for this active pharmaceutical ingredient (API) continues to grow globally, the efficiency and sustainability of its synthetic routes have become paramount concerns for manufacturers. Patent CN102234246A discloses a groundbreaking method for synthesizing Ezetimibe and its key intermediates, addressing critical bottlenecks found in earlier generations of synthesis. This novel approach leverages a combination of trisubstituted chlorosilane and titanium Lewis acid catalysis to construct the crucial azetidinone ring system with high stereoselectivity. For R&D directors and supply chain leaders, understanding this technology is essential, as it represents a shift towards more robust, cost-effective, and environmentally friendly manufacturing processes that ensure long-term supply continuity for this vital cardiovascular medication.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Ezetimibe has relied on routes that, while chemically valid, present severe challenges for industrial scalability and cost efficiency. Early methods, such as those described in US Patent 5767115, utilized a linear sequence involving Grignard reactions catalyzed by palladium complexes like Pd(PPh3)4. As illustrated in the reaction scheme below, these pathways often required multiple protection and deprotection steps, leading to extended production timelines.
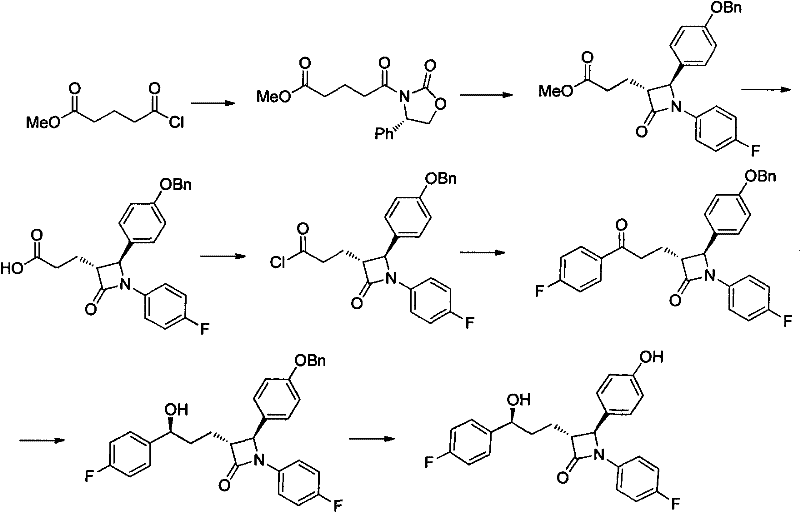
Furthermore, the reliance on precious metal catalysts introduces significant cost volatility and supply chain risks, as palladium prices can fluctuate dramatically. A major technical drawback of these conventional methods is the necessity for column chromatography to purify key intermediates. In a commercial setting, column chromatography is notoriously difficult to scale, often becoming the rate-limiting step that prevents tonnage-level production. Additionally, some routes employed hazardous reagents such as LDA (lithium diisopropylamide) at cryogenic temperatures below -50°C, posing safety risks and requiring specialized, energy-intensive cooling infrastructure that drives up operational expenditures.
The Novel Approach
In stark contrast, the methodology outlined in CN102234246A offers a streamlined, convergent strategy that bypasses these historical inefficiencies. The core innovation lies in the direct coupling of a chiral oxazolidinone derivative with a substituted imine using a titanium Lewis acid system. This reaction proceeds under milder conditions and utilizes readily available reagents, effectively replacing the expensive palladium catalysts with more economical titanium species. The overall transformation is depicted in the following schematic, which highlights the direct formation of the protected intermediate ready for cyclization.
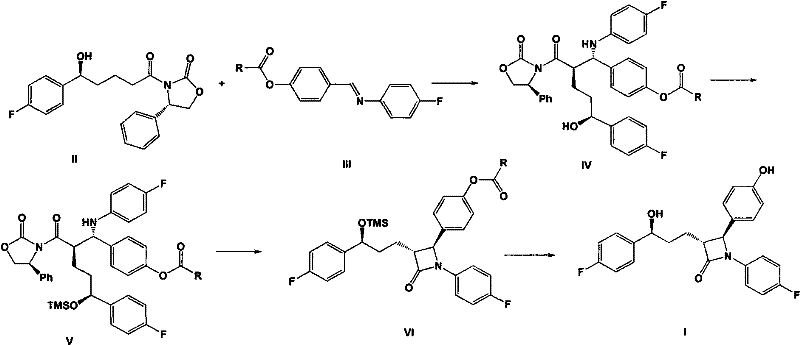
This novel route drastically simplifies the purification workflow. Instead of relying on chromatographic separation, the process allows for the isolation of intermediates and the final product through simple filtration and recrystallization techniques. For instance, the final target product can be obtained with a purity of 99.6% merely by recrystallization from toluene. This shift from chromatography to crystallization is a game-changer for procurement managers, as it implies a significant reduction in solvent consumption, waste generation, and processing time, directly translating to lower manufacturing costs and a smaller environmental footprint.
Mechanistic Insights into Titanium Lewis Acid Catalyzed Cyclization
The success of this synthetic route hinges on the precise orchestration of stereochemistry during the Mannich-type reaction and the subsequent cyclization. The reaction begins with the activation of the imine electrophile by the titanium Lewis acid, likely titanium tetrachloride (TiCl4), which enhances its reactivity towards the nucleophilic enolate derived from the oxazolidinone. The presence of a trisubstituted chlorosilane, such as trimethylchlorosilane (TMSCl), plays a dual role: it acts as a scavenger for water to maintain anhydrous conditions critical for Lewis acid activity, and it temporarily protects the hydroxyl group of the starting material, preventing unwanted side reactions. This careful control of the reaction environment ensures that the newly formed carbon-carbon bond possesses the correct (2R, 1S) stereochemistry required for the biological activity of the final drug.
Following the initial coupling, the intermediate undergoes a fluoride-ion promoted cyclization. The addition of a fluoride source, such as tetrabutylammonium fluoride (TBAF), triggers the removal of the silyl protecting groups and facilitates the intramolecular nucleophilic attack of the nitrogen atom onto the carbonyl carbon. This step closes the four-membered azetidinone ring, which is the structural core of Ezetimibe. The mechanism is highly efficient because the fluoride ion acts as a specific trigger that unmasks the reactive centers only when needed, minimizing the formation of oligomeric byproducts. Finally, a mild acidic workup removes the remaining acyl protecting groups to reveal the free phenolic hydroxyl, yielding the final API intermediate with high optical purity and minimal impurities.
How to Synthesize Ezetimibe Intermediates Efficiently
Implementing this synthesis requires strict adherence to the reaction parameters defined in the patent to ensure optimal yield and stereocontrol. The process involves reacting the chiral oxazolidinone precursor with the specific imine derivative in a solvent like methylene dichloride or toluene. The temperature must be carefully controlled, typically ranging from -78°C to 50°C depending on the specific step, to balance reaction rate and selectivity. The sequential addition of the chlorosilane and the titanium catalyst is critical; adding the Lewis acid too early or too late can lead to incomplete conversion or racemization. Once the coupling is complete, the mixture is treated with BSA (bis(trimethylsilyl)acetamide) to silylate the hydroxyl groups, followed by the fluoride cyclization catalyst. The detailed standardized synthesis steps for replicating this high-efficiency route are provided in the guide below.
- React 3-[(5-(4-fluorophenyl)-(5S)-hydroxyvaleryl]-(4S)-phenyl-oxazolidinone with N-4'-substituted acyloxy benzylidene-4-fluoroaniline using trisubstituted chlorosilane and titanium Lewis acid to form the protected intermediate.
- Treat the resulting intermediate with BSA (bis(trimethylsilyl)acetamide) to generate the silylated species.
- Perform fluoride ion cyclization followed by protonic acid hydrolysis to obtain the final high-purity Ezetimibe target product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this titanium-catalyzed route offers tangible strategic benefits that extend beyond simple chemistry. The primary advantage is the drastic simplification of the supply chain for raw materials. By eliminating the dependency on palladium catalysts, manufacturers insulate themselves from the volatility of precious metal markets and the geopolitical risks associated with their sourcing. Furthermore, the replacement of column chromatography with crystallization reduces the demand for high-purity silica gel and vast quantities of elution solvents, which are significant cost drivers in fine chemical manufacturing. This process intensification allows for higher throughput in existing reactor vessels, effectively increasing capacity without capital expenditure on new equipment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substitution of high-cost reagents with commodity chemicals. Titanium tetrachloride and chlorosilanes are produced on a massive industrial scale and are significantly cheaper than palladium complexes. Additionally, the ability to recycle solvents like toluene and dichloromethane, combined with the reduced need for extensive purification media, lowers the variable cost per kilogram of the intermediate. The high yield reported in the patent examples, reaching over 80% for the final step, further amplifies these savings by maximizing the output from every batch of starting material.
- Enhanced Supply Chain Reliability: Supply continuity is bolstered by the robustness of the reaction conditions. Unlike methods requiring cryogenic temperatures below -50°C or pyrophoric reagents like LDA, this route operates at temperatures that are easily achievable with standard industrial chillers. This reduces the risk of batch failures due to equipment malfunction or temperature excursions. Moreover, the intermediates generated are stable solids that can be isolated and stored, providing flexibility in production scheduling and allowing manufacturers to build inventory buffers against market demand fluctuations without degradation concerns.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is superior. The avoidance of heavy metal waste streams simplifies wastewater treatment and reduces the regulatory burden associated with disposing of palladium-contaminated residues. The use of standard organic solvents allows for established recovery and distillation protocols, minimizing volatile organic compound (VOC) emissions. The process is inherently safer, removing the need for handling highly reactive organometallic reagents under extreme conditions, thereby lowering insurance premiums and operational risks associated with plant safety.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within the patent documentation, providing a reliable basis for feasibility assessments. Understanding these details is crucial for technical teams evaluating the transfer of this technology to pilot or commercial plants.
Q: What are the key advantages of the titanium Lewis acid method over traditional palladium-catalyzed routes?
A: The titanium Lewis acid method eliminates the need for expensive palladium catalysts like Pd(PPh3)4, significantly reducing raw material costs. Furthermore, it avoids complex column chromatography purification steps, relying instead on recrystallization, which is far more suitable for large-scale industrial production.
Q: What purity levels can be achieved with this novel synthesis route?
A: According to the patent data, the final target product can achieve a purity of up to 99.6% after recrystallization. This high purity is critical for pharmaceutical applications where impurity profiles must be strictly controlled.
Q: Is this process suitable for commercial scale-up?
A: Yes, the process is designed for industrial suitability. It utilizes common solvents like methylene dichloride and toluene, operates at manageable temperatures (-78°C to 50°C), and avoids hazardous reagents like LDA, making it safer and easier to scale compared to previous methods.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Intermediate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient synthesis routes in the competitive pharmaceutical market. Our team of expert chemists has extensively analyzed the titanium-catalyzed pathway described in CN102234246A and possesses the technical capability to optimize it for your specific production needs. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from lab-scale success to industrial reality is seamless. Our facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Ezetimibe intermediate meets the highest global regulatory standards.
We invite you to collaborate with us to leverage this advanced technology for your supply chain. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your current manufacturing setup. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our expertise can drive down your costs and secure your supply of this essential cholesterol-lowering agent.
