Advanced Enzymatic Synthesis of Ezetimibe: A Breakthrough in Commercial Scale-Up and Purity Control
Introduction to Next-Generation Ezetimibe Manufacturing
The global demand for cholesterol-lowering agents continues to drive innovation in the synthesis of Ezetimibe, a critical active pharmaceutical ingredient. Patent CN113621663A introduces a transformative synthesis process that addresses long-standing challenges in yield, selectivity, and environmental safety. Unlike conventional methods that struggle with harsh reaction conditions and complex purification, this novel approach leverages a sophisticated combination of 1,4-addition chemistry and enzymatic chiral reduction. By utilizing specific ketoreductases, the process achieves exceptional stereocontrol, effectively freezing the rotation of chemical bonds at the target site to ensure the formation of the correct chiral center. This technological leap not only enhances product purity but also streamlines the manufacturing workflow, positioning it as a superior choice for modern pharmaceutical production lines seeking efficiency and compliance.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Ezetimibe has been plagued by significant technical hurdles that impact both cost and safety. Legacy routes, such as those disclosed by Schering Corporation, often involve lengthy multi-step sequences, including up to seven distinct reaction stages. These traditional pathways frequently rely on hazardous reagents like pivaloyl chloride and borane, which pose substantial safety risks due to their toxic and explosive nature. Furthermore, earlier methods utilizing Nigishi reactions or chiral lactones often suffer from poor stereoselectivity, resulting in trans/cis ratios that necessitate difficult and yield-reducing separation processes. The reliance on expensive catalysts like palladium on carbon and oxidants like sodium periodate further inflates production costs, rendering these older methodologies economically unviable for competitive large-scale manufacturing in today's regulated environment.
The Novel Approach
In stark contrast, the process outlined in patent CN113621663A offers a streamlined and chemically elegant solution that bypasses these historical bottlenecks. The new methodology initiates with a strategic hydroxyl protection step followed by a highly efficient 1,4-addition reaction, which replaces the cumbersome substitution and elimination steps of the past. This fundamental shift in synthetic strategy drastically reduces reaction time and minimizes the generation of by-products. Crucially, the final step employs a biocatalytic reduction using specific ketoreductases, which operates under mild aqueous conditions. This enzymatic step not only achieves the necessary chiral reduction with high fidelity but also simultaneously removes the protecting group in a one-pot operation. This integration of steps simplifies the post-treatment workflow, eliminates the need for toxic heavy metal catalysts, and ensures a final product with purity levels exceeding industry standards, making it ideally suited for reliable pharmaceutical intermediate supplier operations.
Mechanistic Insights into Enzymatic Chiral Reduction
The core innovation of this synthesis lies in the precise mechanistic action of the ketoreductase enzyme during the final transformation stage. Under the influence of specific chiral enzymes, such as ES-KRED-135 or ES-KRED-136, the carbonyl group of Intermediate II binds tightly to the active center of the enzyme. This binding interaction effectively forces the translation and rotation of the chemical bonds at the reactive site to become frozen in a specific conformation. By restricting the molecular freedom of the substrate, the enzyme promotes the reaction to proceed exclusively in the desired direction, thereby facilitating the formation of the target chiral bond with exceptional specificity. This biological precision eliminates the formation of unwanted enantiomers that typically plague chemical catalysis, ensuring that the resulting Ezetimibe possesses the correct stereochemistry required for therapeutic efficacy without the need for extensive chiral chromatography.
Furthermore, the reaction design ingeniously combines chiral reduction with deprotection, a dual-functionality that is rare in synthetic organic chemistry. As the ketoreductase reduces the ketone to the corresponding alcohol, the acidic conditions maintained during the workup (pH 3-5) facilitate the cleavage of the silyl protecting group introduced in the first step. This tandem process means that the intermediate does not need to be isolated and purified between reduction and deprotection, significantly reducing solvent consumption and processing time. The result is a cleaner reaction profile with a simplified impurity spectrum, which is critical for meeting the stringent regulatory requirements for API intermediates. The ability to control the reaction temperature between 20-35°C further underscores the mildness of the process, preserving the integrity of the sensitive beta-lactam ring structure throughout the synthesis.
How to Synthesize Ezetimibe Efficiently
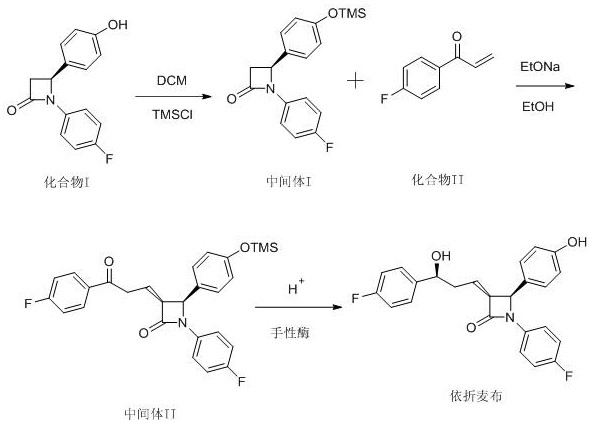
The synthesis of Ezetimibe via this patented route involves a logical sequence of protection, addition, and biocatalytic reduction that can be readily adapted for pilot and commercial scales. The process begins with the protection of the starting lactam, followed by a base-mediated coupling with a fluorophenyl ketone derivative, and concludes with the enzymatic finishing step. Each stage is optimized for high conversion and ease of isolation, utilizing common solvents like dichloromethane and ethanol. The detailed standardized synthesis steps, including specific mass ratios, temperature controls, and stirring speeds required to replicate the high yields reported in the patent, are provided in the technical guide below.
- Protect the hydroxyl group of Compound I using TMSCl in dichloromethane to form Intermediate I.
- Perform a 1,4-addition reaction between Intermediate I and Compound II using an organic base to generate Intermediate II.
- Execute enzymatic chiral reduction and simultaneous deprotection using ketoreductase to yield final Ezetimibe product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this enzymatic synthesis route presents a compelling value proposition centered on risk mitigation and operational efficiency. By transitioning away from processes that require expensive transition metal catalysts and hazardous reagents, manufacturers can significantly reduce the complexity of their supply chains. The elimination of heavy metals removes the need for costly and time-consuming scavenging steps, which are often required to meet residual metal specifications in pharmaceutical products. This simplification directly translates to lower operational expenditures and a reduced environmental footprint, aligning with the growing corporate mandates for green chemistry and sustainable manufacturing practices across the global pharmaceutical industry.
- Cost Reduction in Manufacturing: The replacement of traditional chemical catalysts with biocatalysts offers a profound opportunity for cost optimization in API manufacturing. Traditional routes often incur high costs associated with the purchase of precious metal catalysts and the subsequent disposal of metal-contaminated waste streams. In this novel process, the use of recyclable or biodegradable enzymes, combined with the avoidance of toxic reagents like borane, drastically lowers the raw material costs. Additionally, the one-pot nature of the final reduction and deprotection step reduces solvent usage and energy consumption, leading to substantial cost savings in utilities and waste treatment without compromising on the quality of the final active ingredient.
- Enhanced Supply Chain Reliability: Supply chain resilience is critically dependent on the availability and safety of raw materials. The reagents utilized in this process, such as TMSCl and common organic bases, are commodity chemicals with stable global supply lines, unlike some specialized chiral ligands or unstable organometallic reagents used in older methods. The mild reaction conditions, particularly the ambient temperature range for the enzymatic step, reduce the risk of thermal runaways or safety incidents that could halt production. This operational stability ensures consistent batch-to-batch quality and reliable delivery schedules, allowing partners to maintain lean inventory levels with confidence in the continuity of supply for this high-value pharmaceutical intermediate.
- Scalability and Environmental Compliance: Scaling chemical processes from the laboratory to multi-ton production often reveals hidden bottlenecks related to heat transfer and mixing, especially in exothermic reactions involving hazardous reagents. This enzymatic route is inherently scalable because biocatalytic reactions typically proceed efficiently in aqueous or mixed aqueous-organic systems with manageable heat profiles. The process generates significantly less hazardous waste, simplifying compliance with increasingly strict environmental regulations regarding effluent discharge. The high selectivity of the enzyme minimizes the formation of side products, which reduces the burden on downstream purification units and allows for a more compact and efficient plant footprint, facilitating rapid commercial scale-up of complex pharmaceutical intermediates.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis presented in the patent documentation, providing clarity on how this method outperforms legacy techniques in terms of purity, yield, and operational safety. Understanding these details is essential for technical teams evaluating the feasibility of integrating this route into their existing manufacturing infrastructure.
Q: How does the enzymatic route improve upon traditional chemical synthesis for Ezetimibe?
A: Traditional methods often rely on harsh reagents like borane or expensive palladium catalysts with poor stereoselectivity. This patented enzymatic route utilizes specific ketoreductases to achieve high stereoselectivity under mild conditions, eliminating the need for toxic heavy metals and complex purification steps.
Q: What are the key advantages of the 1,4-addition strategy in this process?
A: The 1,4-addition strategy replaces traditional substitution and elimination reactions, significantly shortening reaction time and reducing the number of synthetic steps. This leads to higher overall yields and a more robust process suitable for industrial mass production.
Q: Can this process be scaled for commercial API manufacturing?
A: Yes, the process is designed for industrial suitability. It operates under mild temperatures (20-35°C for enzymatic step) and avoids explosive or highly toxic reagents, making it safer and more cost-effective for large-scale commercial API manufacturing compared to legacy routes.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Ezetimibe Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to remain competitive in the global pharmaceutical market. Our team of expert chemists has thoroughly analyzed the potential of this enzymatic route and is fully prepared to support its translation from patent to production. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition to this greener, more efficient process is seamless. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of Ezetimibe intermediate meets the highest international standards for safety and efficacy.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this innovative synthesis technology. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific production volumes and regional regulatory requirements. We encourage you to contact us today to request specific COA data and comprehensive route feasibility assessments, allowing you to make informed decisions that will enhance your supply chain resilience and drive down the total cost of ownership for your cholesterol-lowering drug portfolio.
