Scalable Synthesis of Pentafluoroethoxy Cyclotriphosphazene for High-Performance Battery Electrolytes
Scalable Synthesis of Pentafluoroethoxy Cyclotriphosphazene for High-Performance Battery Electrolytes
The rapid expansion of the lithium-ion battery market has necessitated the development of safer, more efficient electrolyte additives, with pentafluoroethoxy cyclotriphosphazene emerging as a critical flame retardant component. Patent CN109422774B introduces a groundbreaking preparation method that addresses the longstanding challenges of yield and purity associated with organophosphorus flame retardants. This technology leverages a sophisticated dual-catalyst system involving alkali metal iodides and phase transfer catalysts to facilitate the nucleophilic substitution of hexafluorocyclotriphosphazene with ethanol. Unlike conventional high-energy pathways, this approach operates under remarkably mild thermal conditions, typically ranging from 20°C to 50°C, which drastically reduces energy consumption and thermal stress on the sensitive phosphazene ring structure. For R&D directors and process engineers, this represents a significant leap forward in controlling the impurity profile of battery-grade chemicals, ensuring that the final electrolyte formulation meets the rigorous safety standards required for next-generation energy storage systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of pentafluoroethoxy cyclotriphosphazene has been plagued by inefficient reaction pathways that compromise both yield and product integrity. Traditional methods often rely on the reaction of hexachlorocyclotriphosphazene with ethoxide, a highly reactive nucleophile that frequently leads to over-substitution, resulting in significant quantities of diethoxy substituted by-products that are difficult to separate. Alternatively, methods utilizing ammonia as an acid-binding agent have been known to introduce amino substituents into the phosphazene ring, creating complex impurity spectra that degrade the electrochemical performance of the final battery electrolyte. These side reactions not only lower the overall conversion rate but also necessitate extensive and costly purification steps, such as multiple recrystallizations or complex chromatographic separations, which are impractical for large-scale manufacturing. Furthermore, the harsh conditions often required for these legacy processes can lead to the degradation of the phosphazene backbone, further diminishing the quality of the flame retardant additive.
The Novel Approach
The methodology disclosed in CN109422774B fundamentally reengineers the synthetic route by employing hexafluorocyclotriphosphazene as the starting material rather than the chlorinated precursor, thereby eliminating the need for aggressive fluorination steps. By utilizing ethanol directly in the presence of a specialized catalyst system, the reaction achieves high selectivity for the mono-ethoxy substitution pattern required for the target molecule. The integration of alkali metal iodides, such as potassium iodide or sodium iodide, acts as a potent nucleophilic promoter, while the phase transfer catalyst ensures efficient interaction between the reactants in the organic phase. This synergistic catalytic effect allows the reaction to proceed with exceptional conversion rates, often exceeding 95%, while maintaining a clean reaction profile with minimal by-product formation. The result is a streamlined process that yields pentafluoroethoxy cyclotriphosphazene with a purity of up to 99.9% following simple filtration and rectification, offering a robust solution for cost reduction in electronic chemical manufacturing.
Mechanistic Insights into Alkali Metal Iodide-Catalyzed Nucleophilic Substitution
The core innovation of this patent lies in the precise modulation of nucleophilicity through the use of alkali metal iodides. In the reaction mechanism, the iodide ion serves as a superior leaving group and nucleophilic activator compared to chloride or fluoride, facilitating the displacement of fluorine atoms on the phosphazene ring by the ethoxy group derived from ethanol. This process is kinetically controlled to prevent poly-substitution, ensuring that the reaction stops predominantly at the desired pentafluoroethoxy stage. The phase transfer catalyst, such as tetrabutylammonium bromide or polyethylene glycol 400, plays a critical role in solubilizing the inorganic base and catalyst species within the organic solvent matrix, creating a homogeneous reaction environment that maximizes collision frequency between reactants. This homogeneity is crucial for maintaining consistent reaction rates throughout the bulk solution, preventing localized hot spots that could trigger decomposition.
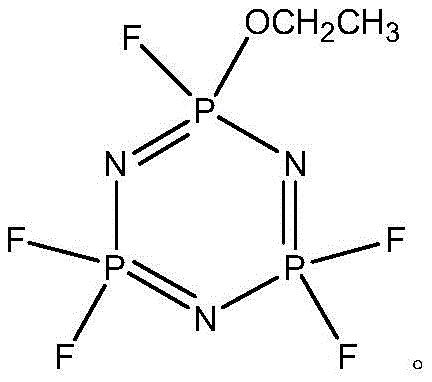
Impurity control is achieved through the careful selection of the acid-binding agent, which neutralizes the hydrogen fluoride generated during the substitution without introducing nucleophilic contaminants like amines. When agents such as sodium carbonate or triethylamine are used, the reaction pH is buffered effectively, preventing the acid-catalyzed degradation of the phosphazene ring. The mild temperature window of 20°C to 50°C further suppresses thermal side reactions, ensuring that the impurity spectrum remains narrow and manageable. For high-purity OLED material and battery electrolyte manufacturers, this level of control over the molecular architecture is essential, as even trace impurities can act as redox shuttles or decomposition initiators within the electrochemical cell. The ability to achieve such high purity through a single rectification step underscores the efficiency of this catalytic system in producing commercial scale-up of complex organophosphorus compounds.
How to Synthesize Pentafluoroethoxy Cyclotriphosphazene Efficiently
The operational protocol for this synthesis is designed for ease of implementation in standard chemical reactors, requiring no specialized high-pressure equipment. The process begins with the dissolution of hexafluorocyclotriphosphazene and ethanol in a selected organic solvent, followed by the sequential addition of the catalyst system and acid binder. The reaction is then allowed to stir under controlled thermal conditions until conversion is complete, monitored via standard analytical techniques. Detailed standardized synthesis steps see the guide below.
- Prepare the reaction mixture by dissolving hexafluorocyclotriphosphazene and ethanol in an organic solvent such as n-hexane or acetonitrile.
- Introduce the dual catalyst system comprising an alkali metal iodide main catalyst and a phase transfer co-catalyst, along with an acid binding agent.
- Maintain the reaction at mild temperatures between 20-50°C, followed by filtration and rectification to isolate the 99.9% pure product.
Commercial Advantages for Procurement and Supply Chain Teams
From a procurement and supply chain perspective, this patented method offers substantial strategic advantages by simplifying the raw material portfolio and reducing processing complexity. The shift away from hazardous fluorinating agents and highly reactive ethoxides towards stable ethanol and common alkali metal salts significantly lowers the barrier to entry for production and reduces the regulatory burden associated with handling dangerous reagents. This simplification translates directly into improved supply chain resilience, as the key inputs are commodity chemicals with robust global availability, mitigating the risk of shortages that often plague specialty fluorine chemistry supply chains. Furthermore, the mild reaction conditions reduce the energy intensity of the manufacturing process, aligning with sustainability goals and lowering utility costs associated with heating and cooling large-scale reactors.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous fluorination steps, combined with the use of low-cost catalysts like potassium iodide, drastically reduces the raw material cost per kilogram of the final product. Additionally, the high conversion rate minimizes the loss of valuable hexafluorocyclotriphosphazene starting material, ensuring that the atom economy of the process is optimized for maximum financial efficiency. The simplified downstream processing, requiring only filtration and rectification rather than complex chromatographic purification, further reduces operational expenditures related to solvent usage and waste disposal.
- Enhanced Supply Chain Reliability: By relying on widely available solvents such as n-hexane, acetonitrile, and dichloroethane, the process ensures that production is not bottlenecked by the availability of niche reagents. The stability of the reaction conditions also allows for flexible scheduling and batch sizing, enabling manufacturers to respond rapidly to fluctuations in demand from the battery sector without compromising product quality. This flexibility is critical for maintaining reducing lead time for high-purity battery materials in a fast-moving market.
- Scalability and Environmental Compliance: The process generates minimal hazardous waste, primarily consisting of inorganic salts that can be easily separated and disposed of according to standard environmental protocols. The absence of heavy metal catalysts or toxic by-products simplifies the environmental impact assessment and permits easier compliance with increasingly stringent global regulations on chemical manufacturing. This environmental compatibility facilitates smoother scaling from pilot plant to full commercial production, ensuring a continuous and reliable supply of this critical battery additive.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims presented in the patent documentation, providing a factual basis for evaluating the process viability. Understanding these details is crucial for technical teams assessing the integration of this method into existing production lines.
Q: What are the primary advantages of the alkali metal iodide catalytic system?
A: The use of alkali metal iodides combined with phase transfer catalysts allows for nucleophilic substitution to proceed efficiently at significantly lower temperatures (20-50°C) compared to traditional methods, minimizing thermal degradation and by-product formation.
Q: How does this method improve purity compared to ethoxide-based routes?
A: Traditional ethoxide routes often generate diethoxy substituted by-products due to excessive nucleophilicity. This patented method controls the reaction kinetics precisely, yielding a target product purity of up to 99.9% after rectification with minimal side reactions.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the process utilizes common organic solvents and avoids hazardous high-pressure fluorination steps, making it highly adaptable for commercial scale-up with simplified downstream processing via filtration and distillation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentafluoroethoxy Cyclotriphosphazene Supplier
As the demand for safer and more efficient lithium battery electrolytes continues to surge, the ability to produce high-quality flame retardant additives like pentafluoroethoxy cyclotriphosphazene at scale becomes a critical competitive advantage. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to deliver consistent, high-volume supply. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, ensuring that every batch of organophosphorus intermediate meets the exacting standards required by top-tier battery manufacturers globally.
We invite you to collaborate with us to optimize your supply chain for battery electrolyte components. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how our advanced catalytic processes can enhance your bottom line. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us partner with you to power the future of energy storage.
