Advanced Synthesis of Pentafluoroethoxy Cyclotriphosphazene for Next-Gen Lithium Battery Safety
The escalating demand for high-energy-density lithium-ion batteries in electric vehicles and consumer electronics has intensified the focus on thermal safety and fire suppression technologies. Patent CN110066295B introduces a transformative preparation method for pentafluoroethoxy cyclotriphosphazene, a critical flame retardant additive designed to mitigate thermal runaway risks. Unlike conventional synthetic routes that struggle with waste management and selectivity, this innovation leverages a streamlined two-step sequence: a Lewis acid-catalyzed fluorination using hydrogen fluoride gas, followed by a precise etherification reaction. This technical breakthrough addresses the fundamental bottlenecks in producing phosphazene-based electrolyte additives, offering a pathway to higher purity intermediates essential for next-generation battery safety standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of functionalized cyclotriphosphazenes has been plagued by significant process inefficiencies and environmental burdens. Traditional approaches often attempted to substitute ethoxy groups onto hexachlorocyclotriphosphazene prior to fluorination, a strategy that suffered from poor control over mono-substitution states, leading to complex mixtures and low yields. Alternatively, methods utilizing solid fluoride salts like potassium fluoride or sodium fluoride for the fluorination step generated substantial quantities of inorganic solid waste. These solid-state reactions required vigorous stirring to overcome mass transfer limitations caused by chloride byproduct occlusion, necessitating a large excess of fluorinating agents. Furthermore, the high temperatures and extended reaction times required for solid salt fluorination increased energy consumption and risked the loss of volatile hexafluorocyclotriphosphazene intermediates, severely impacting overall process economics and environmental compliance.
The Novel Approach
The methodology disclosed in patent CN110066295B fundamentally reengineers the synthetic logic by prioritizing complete fluorination before etherification. By employing hydrogen fluoride gas in the presence of soluble Lewis acid catalysts such as antimony pentachloride or ferric chloride, the reaction proceeds in a homogeneous liquid phase. This shift eliminates the solid waste issues inherent to salt-based fluorination and ensures thorough mixing without mechanical constraints. The subsequent etherification step utilizes sodium alkoxide under mild conditions to selectively replace a single fluorine atom, capitalizing on the stability of the fully fluorinated intermediate. This reverse-order strategy not only simplifies purification through standard distillation but also drastically reduces the formation of hazardous byproducts, aligning perfectly with modern green chemistry principles and industrial scalability requirements.
Mechanistic Insights into Lewis Acid-Catalyzed Fluorination and Etherification
The core of this innovative process lies in the efficient nucleophilic substitution facilitated by Lewis acid catalysis. In the initial fluorination stage, the electron-deficient phosphorus centers in hexachlorocyclotriphosphazene are activated by catalysts like SbCl5 or TiCl4, enhancing their susceptibility to attack by fluoride ions derived from hydrogen fluoride. This catalytic cycle allows the replacement of all six chlorine atoms with fluorine at relatively low temperatures ranging from 0°C to 60°C, a significant improvement over the harsh conditions needed for non-catalyzed solid-state reactions. The use of aromatic hydrocarbon solvents, such as benzotrifluoride, ensures optimal solubility for both the reactants and the catalyst, maintaining a homogeneous reaction environment that maximizes collision frequency and conversion rates while minimizing side reactions.
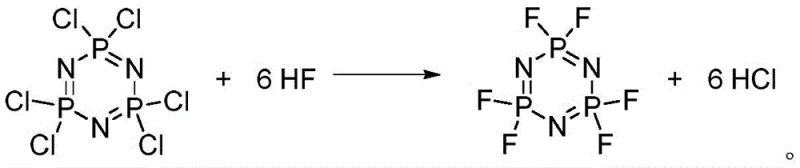
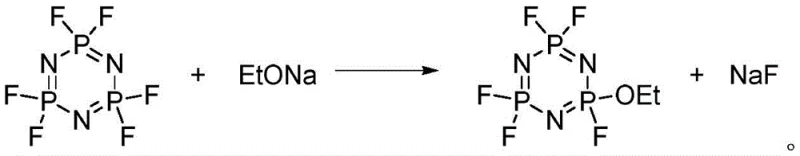
Following the isolation of the hexafluorocyclotriphosphazene intermediate, the mechanism shifts to a classic Williamson ether synthesis pathway. In this second stage, the strong nucleophile sodium ethoxide attacks the electrophilic phosphorus atom of the fluorinated ring. The reaction is conducted at low temperatures, typically between -30°C and 30°C, to kinetically control the substitution and prevent poly-etherification, ensuring the formation of the desired mono-ethoxy product. The leaving group, fluoride ion, precipitates as sodium fluoride, which is easily removed by filtration. This mechanistic precision allows for the production of high-purity pentafluoroethoxy cyclotriphosphazene, as evidenced by the clean spectral data and high HPLC purity reported in the patent examples, confirming the robustness of the chemical transformation.
How to Synthesize Pentafluoroethoxy Cyclotriphosphazene Efficiently
Implementing this synthesis route requires careful attention to reactor material selection and temperature control protocols to handle corrosive reagents safely. The process begins with the charging of hexachlorocyclotriphosphazene and a Lewis acid catalyst into a Hastelloy reactor, followed by the controlled introduction of anhydrous hydrogen fluoride gas. Once the fluorination is complete and the intermediate is distilled, the residue is redissolved in an aprotic polar solvent for the etherification step. Detailed standardized operating procedures regarding molar ratios, specific distillation cuts, and safety interlocks are critical for reproducibility. For the complete step-by-step technical protocol including exact reagent grades and workup procedures, please refer to the standardized guide below.
- Perform fluorination of hexachlorocyclotriphosphazene using hydrogen fluoride gas and a Lewis acid catalyst (e.g., SbCl5) in an aromatic solvent at 0-60°C.
- Isolate the intermediate hexafluorocyclotriphosphazene via distillation under reduced pressure to remove excess HF and solvent.
- Conduct etherification by reacting the intermediate with sodium ethoxide in an aprotic polar solvent at low temperatures (-30 to 30°C) to yield the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the transition to this gas-phase fluorination technology represents a strategic opportunity to optimize cost structures and secure supply continuity. The elimination of solid fluoride salts removes the logistical burden of handling and disposing of tons of inorganic waste, which traditionally incurs significant disposal fees and regulatory overhead. By shifting to a liquid-phase system with gaseous reagents, the process inherently reduces the volume of raw materials required per unit of output, as the stoichiometric efficiency is markedly improved without the need for massive excesses to drive equilibrium. This efficiency translates directly into a leaner bill of materials and reduced operational expenditure on waste treatment infrastructure.
- Cost Reduction in Manufacturing: The adoption of hydrogen fluoride gas coupled with catalytic amounts of Lewis acids significantly lowers the variable costs associated with reagent consumption. Unlike solid salt methods that require large excesses to overcome kinetic barriers and occlusion issues, this method achieves high conversion with near-stoichiometric ratios. Furthermore, the simplified downstream processing, which relies on straightforward distillation rather than complex extraction or washing sequences to remove solids, reduces utility consumption and labor hours. The ability to recycle aromatic solvents like benzotrifluoride further enhances the economic viability, ensuring that the cost reduction in battery additive manufacturing is sustainable and scalable.
- Enhanced Supply Chain Reliability: Sourcing high-purity flame retardant intermediates is often constrained by the limited number of suppliers capable of managing hazardous fluorination chemistries. This patented route utilizes commodity chemicals such as hydrogen fluoride and sodium ethoxide, which are widely available in the global chemical market, reducing dependency on specialized fluorinating agents. The robustness of the reaction conditions, which tolerate standard industrial equipment materials like Hastelloy, means that more contract manufacturing organizations can potentially adopt this technology. This diversification of potential production sites mitigates supply risk and ensures a steady flow of critical materials for lithium battery production lines.
- Scalability and Environmental Compliance: As regulatory pressures on chemical manufacturing intensify, processes that minimize solid waste generation offer a distinct competitive advantage. This technology generates primarily gaseous byproducts like HCl, which can be scrubbed and recovered, and minimal solid waste in the form of NaF. The absence of heavy metal catalysts or persistent organic pollutants simplifies environmental permitting and reduces the long-term liability associated with site remediation. The process is inherently designed for scale-up, with reaction parameters that are easily controlled in large-volume reactors, facilitating the commercial scale-up of complex electronic chemicals without compromising safety or environmental standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation, aiming to clarify the operational benefits for potential partners and licensees. Understanding these nuances is essential for evaluating the feasibility of integrating this route into existing production portfolios.
Q: Why is the HF gas fluorination method superior to solid fluoride salt methods?
A: The HF gas method eliminates the generation of massive inorganic solid wastes associated with potassium or sodium fluoride salts. It also avoids the occlusion of reactants by chloride byproducts, ensuring higher conversion rates and easier purification without excessive reagent usage.
Q: How does this process control the selectivity of mono-substitution?
A: By reversing the traditional order—performing full fluorination first to create a stable hexafluoro-intermediate, followed by controlled low-temperature etherification—the process avoids the difficult selectivity issues of partial substitution on the chloro-precursor.
Q: What are the critical safety parameters for the fluorination step?
A: The reaction is exothermic and requires strict temperature control between 0°C and 60°C, typically using a water bath during HF gas introduction. Hastelloy reactors are recommended due to the corrosive nature of hydrogen fluoride and Lewis acid catalysts.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Pentafluoroethoxy Cyclotriphosphazene Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical role that advanced flame retardants play in the safety and performance of modern energy storage systems. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that laboratory innovations are successfully translated into reliable industrial supply. We maintain stringent purity specifications and operate rigorous QC labs equipped to analyze complex fluorine-containing compounds, guaranteeing that every batch meets the exacting standards required by the battery industry. Our commitment to technical excellence allows us to navigate the complexities of hazardous chemistry with unmatched safety and efficiency.
We invite you to collaborate with us to leverage this patented technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can support your long-term strategic goals in the electronic materials sector.
