Advanced Manufacturing of Voranolan Fumarate: A High-Yield Technical Breakthrough
Advanced Manufacturing of Voranolan Fumarate: A High-Yield Technical Breakthrough
The pharmaceutical industry continuously seeks robust synthetic pathways for gastric acid secretion inhibitors, specifically targeting the novel compound Voranolan fumarate (Vonoprazan fumarate). Patent CN113861167A discloses a revolutionary preparation method that addresses the longstanding inefficiencies of prior art. This technical insight report analyzes the novel route, which leverages a strategic combination of copper-catalyzed click chemistry and rhodium-catalyzed transannulation to achieve a total yield exceeding 60 percent. For R&D directors and procurement specialists, this represents a pivotal shift from the conventional 39 percent yield baseline, offering a tangible pathway to cost reduction and supply chain stability in the manufacturing of high-purity API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Voranolan fumarate has relied on routes initiating from 5-(2-fluorophenyl)-2-chloro-1H-pyrrole-3-nitrile. While chemically feasible, this conventional approach suffers from inherent structural vulnerabilities. The chloro-pyrrole intermediate is highly susceptible to unwanted side reactions during subsequent functionalization steps, leading to complex impurity profiles that are difficult to separate. Furthermore, the harsh conditions often required to manipulate the nitrile group result in significant material loss, capping the total process yield at approximately 39 percent. For a commercial supply chain, this low efficiency translates to excessive raw material consumption, higher waste disposal costs, and unpredictable batch-to-batch consistency, posing a severe risk to long-term procurement strategies.
The Novel Approach
The methodology outlined in CN113861167A circumvents these pitfalls by constructing the core pyrrole ring late in the synthesis via a denitrogenative transannulation strategy. Instead of struggling with unstable chloro-pyrroles, the process builds a stable triazole intermediate through a highly efficient copper-catalyzed azide-alkyne cycloaddition. This triazole is then seamlessly converted into the target pyrrole scaffold using organorhodium catalysis. This divergence from traditional logic not only simplifies the purification workflow but also drastically improves atom economy. The result is a streamlined process where the total yield is boosted to over 60 percent, providing a reliable foundation for commercial scale-up of complex pharmaceutical intermediates.
Mechanistic Insights into Rhodium-Catalyzed Transannulation
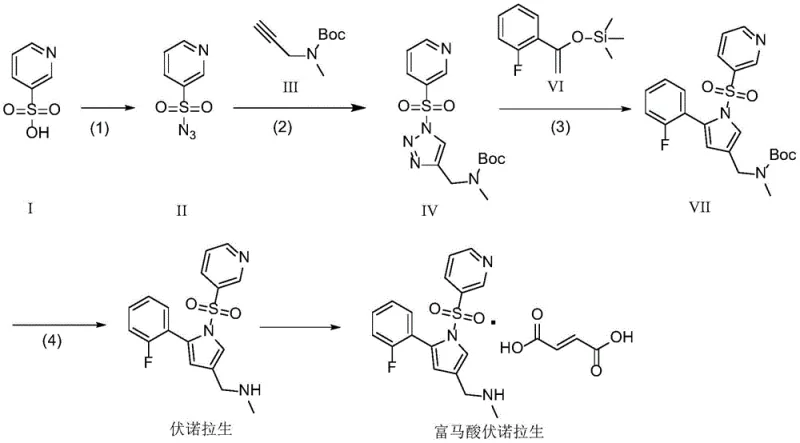
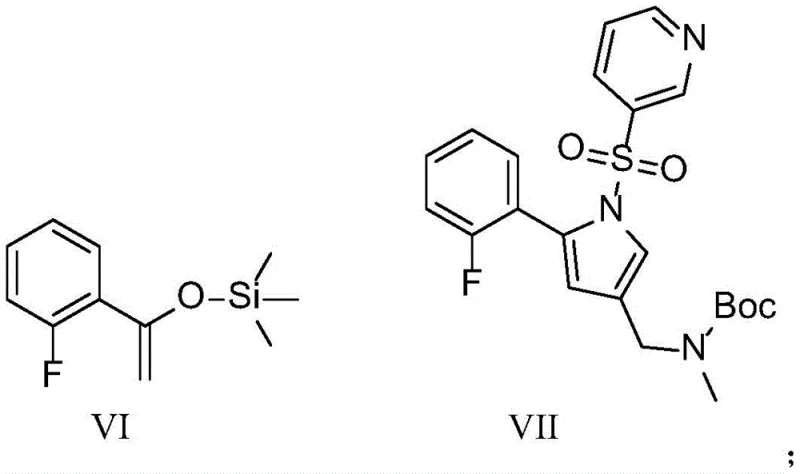
The cornerstone of this novel synthesis is the transformation of the triazole intermediate (Formula IV) into the pyrrole derivative (Formula VII). This step employs a dimeric rhodium acetate catalyst in conjunction with a Lewis acid promoter such as trimethylsilyl trifluoromethanesulfonate (TMSOTf). Mechanistically, the rhodium carbene species generated from the triazole undergoes a [3+2] cycloaddition with the silyl enol ether (Formula VI). This reaction is exceptionally selective, favoring the formation of the desired 1,2,3,5-tetrasubstituted pyrrole ring while minimizing the formation of regioisomers. The mild reaction temperature of 80°C in 1,2-dichloroethane ensures that sensitive functional groups, such as the pyridyl sulfonyl moiety, remain intact throughout the transformation.
Preceding this key step is the formation of the triazole ring via a copper-catalyzed cycloaddition between the sulfonyl azide (Formula II) and the protected propargyl amine (Formula III). Utilizing copper acetate and ligands like 2-aminophenol, this click chemistry reaction proceeds rapidly at room temperature with yields approaching 95 percent. The high fidelity of this step ensures that the substrate fed into the rhodium-catalyzed stage is of exceptional purity, thereby reducing the burden on downstream chromatography. This dual-catalyst strategy effectively decouples the construction of the nitrogen-rich heterocycle from the introduction of the fluorophenyl group, allowing for modular optimization of each stage.
How to Synthesize Voranolan Fumarate Efficiently
The synthesis protocol described in the patent offers a clear, step-by-step guide for producing Voranolan fumarate with industrial viability. The process begins with the activation of pyridine-3-sulfonic acid followed by sequential ring-building reactions that maximize yield at every juncture. By adhering to the specific molar ratios and catalyst loadings detailed in the examples, manufacturers can replicate the high-efficiency profile observed in the patent data. The detailed standardized synthesis steps are provided in the guide below to assist technical teams in process validation.
- Perform azidation on pyridine-3-sulfonic acid to generate the sulfonyl azide intermediate.
- Execute copper-catalyzed cyclization with a propargyl amine derivative to form the triazole precursor.
- Conduct rhodium-catalyzed condensation with a silyl enol ether to construct the pyrrole core, followed by deprotection and salt formation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers compelling economic and operational benefits. The primary driver of value is the substantial increase in overall process yield, which directly correlates to a reduction in the cost of goods sold (COGS). By eliminating the low-yielding steps associated with chloro-pyrrole intermediates, the new method significantly reduces the quantity of starting materials required per kilogram of final API. This efficiency gain is compounded by the use of robust intermediates that are less prone to degradation, thereby minimizing batch failures and ensuring a more consistent supply of high-purity Voranolan fumarate for downstream formulation.
- Cost Reduction in Manufacturing: The elimination of transition metal removal steps associated with less selective catalysts, combined with the high yield of the rhodium-catalyzed step, leads to significant cost optimization. The process avoids the expensive and time-consuming purification protocols necessitated by the side reactions of conventional routes. Furthermore, the ability to use crude intermediates in subsequent steps without extensive isolation reduces solvent consumption and processing time, driving down operational expenditures significantly.
- Enhanced Supply Chain Reliability: The reliance on commercially available and stable starting materials, such as pyridine-3-sulfonic acid and simple silyl enol ethers, mitigates the risk of raw material shortages. The mild reaction conditions, particularly the room temperature cycloaddition, reduce energy consumption and equipment stress, facilitating continuous operation. This robustness ensures that production schedules can be maintained without the delays often caused by the sensitivity of traditional pyrrole precursors to moisture or heat.
- Scalability and Environmental Compliance: The synthetic route is designed with scalability in mind, utilizing solvents like acetonitrile and dichloroethane which are well-understood in large-scale reactor systems. The high atom economy of the click chemistry and transannulation steps results in less chemical waste generation, simplifying effluent treatment and supporting environmental compliance goals. This green chemistry profile aligns with modern regulatory standards, making the facility more sustainable and reducing the long-term liability associated with hazardous waste disposal.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Voranolan fumarate using this patented methodology. These insights are derived directly from the experimental data and process descriptions found in CN113861167A, providing a factual basis for decision-making.
Q: What is the primary advantage of this new synthesis route?
A: The primary advantage is a significant increase in total yield, exceeding 60%, compared to the conventional route which typically yields around 39%. This is achieved through robust intermediate stability and efficient catalytic steps.
Q: What catalysts are critical for the pyrrole ring formation?
A: The process utilizes a dimeric rhodium acetate catalyst combined with an acid catalyst such as trimethylsilyl trifluoromethanesulfonate (TMSOTf) to facilitate the transannulation of the triazole intermediate.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the reaction conditions are relatively mild, with key steps like the cycloaddition occurring at room temperature, and the use of standard solvents facilitates scalable manufacturing and purification.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Voranolan Fumarate Supplier
At NINGBO INNO PHARMCHEM, we understand the critical importance of adopting advanced synthetic routes to maintain competitiveness in the global pharmaceutical market. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the high yields demonstrated in the lab are faithfully reproduced at an industrial scale. We operate stringent purity specifications and utilize rigorous QC labs to guarantee that every batch of Voranolan fumarate meets the highest international standards for API intermediates.
We invite you to collaborate with us to leverage this cutting-edge technology for your supply chain. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and route feasibility assessments to demonstrate how our optimized manufacturing process can enhance your product's profitability and market readiness.
