Advanced Synthetic Route for Dabigatran Etexilate and Analogues for Commercial Scale-up
The pharmaceutical landscape for anticoagulant therapies continues to evolve, driven by the urgent clinical need for effective thromboembolic disease treatments. Patent CN110981858A discloses a groundbreaking preparation method for Dabigatran Etexilate and its structural analogues, addressing critical bottlenecks in current manufacturing paradigms. This novel approach leverages a concise synthetic strategy starting from 3-[(3-amino-4-methylaminobenzoyl)pyridine-2-ylamino]ethyl propionate and 2-(4-cyanophenylamino)isopropyl acetate. By streamlining the condensation and cyclization steps, the technology offers a robust pathway that enhances operability and product yield. For R&D directors and supply chain leaders, this represents a significant opportunity to optimize the production of high-purity pharmaceutical intermediates while mitigating the risks associated with complex, low-yield legacy processes.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthesis routes for Dabigatran Etexilate have historically been plagued by excessive step counts and convoluted purification protocols that hinder efficient commercialization. Existing methods often require harsh reaction conditions that degrade sensitive functional groups, leading to poor impurity profiles and necessitating expensive chromatographic separations. Furthermore, the reliance on scarce or costly reagents in older pathways creates substantial volatility in raw material pricing and supply continuity. These inefficiencies not only inflate the cost of goods sold but also extend lead times, making it difficult for manufacturers to respond agilely to market demand fluctuations. The cumulative effect of these drawbacks is a manufacturing process that is fragile, expensive, and ill-suited for the rigorous quality standards required in modern anticoagulant drug production.
The Novel Approach
In stark contrast, the methodology outlined in the patent introduces a streamlined butt-joint reaction strategy that drastically simplifies the molecular construction of the target compound. By utilizing specific alkali reagents and advanced condensing agents, the new route achieves high conversion rates under mild thermal conditions, preserving the integrity of the molecular scaffold. This approach eliminates several intermediate isolation steps, thereby reducing solvent consumption and waste generation significantly. The operational simplicity allows for easier scale-up, as the reaction parameters are forgiving and the workup procedures involve standard crystallization techniques rather than complex extractions. This shift towards a more direct synthetic logic provides a reliable pharma intermediates supplier with the capability to deliver consistent quality at a reduced operational burden.
Mechanistic Insights into Condensation and Benzimidazole Cyclization
The core of this technological advancement lies in the precise orchestration of amide bond formation followed by an intramolecular cyclization to construct the benzimidazole core. The initial step involves the activation of the carboxylic acid moiety using coupling reagents such as 1-(3-dimethylaminopropyl)-3-ethylcarbodiimide hydrochloride (EDCI) in conjunction with 1-hydroxybenzotriazole (HOBt). This activation generates a highly reactive O-acylisourea intermediate that rapidly reacts with the amine nucleophile to form the peptide-like bond with minimal racemization or side products. Following this condensation, the intermediate undergoes an acid-catalyzed cyclization in an alcohol solvent, where the proximity of the amino and amidine groups facilitates the ring closure to form the stable benzimidazole structure. This mechanistic pathway is crucial for ensuring the correct regiochemistry and high purity of the final active pharmaceutical ingredient precursor.
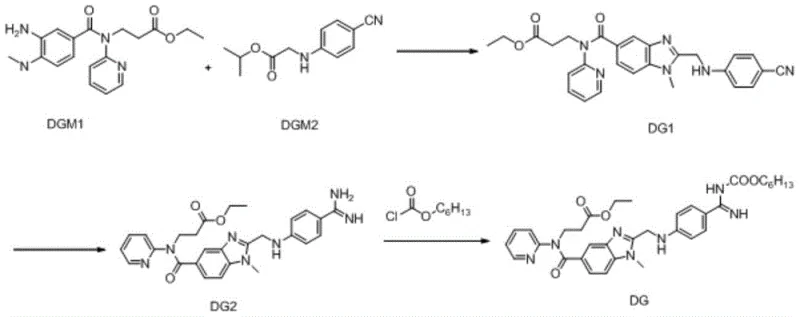
Furthermore, the subsequent functionalization steps demonstrate exceptional control over chemoselectivity, particularly during the formation of the carbamate linkage. The reaction of the benzimidazole intermediate with n-hexyl chloroformate proceeds efficiently under basic conditions, where the base scavenges the generated hydrochloric acid to drive the equilibrium forward. The use of mild bases like potassium carbonate or sodium carbonate prevents the hydrolysis of the ester groups present elsewhere in the molecule, a common pitfall in less optimized protocols. This careful balance of reactivity ensures that the final product retains all necessary functional groups for biological activity while achieving the desired lipophilicity for oral absorption. Such mechanistic precision is vital for producing high-purity OLED material or pharmaceutical grades where trace impurities can compromise safety profiles.
How to Synthesize Dabigatran Etexilate Efficiently
The execution of this synthesis requires strict adherence to stoichiometric ratios and temperature controls to maximize yield and minimize byproduct formation. The process begins with the dissolution of starting materials in dichloromethane at controlled low temperatures to manage the exothermic nature of the coupling reaction. Detailed standardized operating procedures for each stage, including specific molar ratios of condensing agents and precise pH adjustments during workup, are essential for reproducibility. For a comprehensive guide on the exact experimental conditions and purification methods, please refer to the structured protocol below.
- Condense 3-[(3-amino-4-methylaminobenzoyl)pyridine-2-ylamino]ethyl propionate with 2-(4-cyanophenylamino)isopropyl acetate using EDCI/HOBt coupling agents.
- Perform acid-catalyzed cyclization in alcohol solvent followed by ammoniation to form the benzimidazole intermediate DG2.
- React the intermediate DG2 with n-hexyl chloroformate under alkaline conditions to form the final amido bond of Dabigatran Etexilate.
Commercial Advantages for Procurement and Supply Chain Teams
From a strategic procurement perspective, this synthetic route offers transformative benefits that directly impact the bottom line and supply chain resilience. The reduction in synthetic steps translates to a significant decrease in man-hours and equipment occupancy time, allowing for higher throughput within existing facility footprints. Moreover, the substitution of exotic reagents with commodity chemicals like carbonates and common organic solvents reduces exposure to volatile raw material markets. This stability is critical for maintaining consistent pricing structures and ensuring long-term supply security for downstream API manufacturers who rely on just-in-time delivery models.
- Cost Reduction in Manufacturing: The elimination of transition metal catalysts and the reduction of purification steps drastically lower the operational expenditure associated with waste treatment and solvent recovery. By avoiding expensive heavy metal removal processes, the overall cost of production is substantially decreased without compromising product quality. Additionally, the high yields reported in the patent examples mean that less raw material is wasted, further enhancing the economic efficiency of the manufacturing process.
- Enhanced Supply Chain Reliability: The reliance on widely available starting materials such as substituted anilines and simple amino acids ensures that the supply chain is not vulnerable to single-source bottlenecks. This diversification of raw material sources allows for greater flexibility in sourcing strategies, reducing the risk of production stoppages due to supplier shortages. Consequently, lead times for high-purity pharmaceutical intermediates can be shortened, enabling faster response to market demands.
- Scalability and Environmental Compliance: The process utilizes green chemistry principles by minimizing solvent usage and employing aqueous workups where possible, which simplifies compliance with increasingly stringent environmental regulations. The robustness of the reaction conditions allows for seamless translation from laboratory scale to multi-ton commercial production, ensuring that quality remains consistent regardless of batch size. This scalability is essential for meeting the growing global demand for anticoagulant therapies.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the patent specifications and are intended to clarify the feasibility and advantages of adopting this route for industrial applications. Understanding these details is key for stakeholders evaluating the potential for technology transfer or licensing agreements.
Q: What are the primary advantages of this new synthetic route for Dabigatran Etexilate?
A: The disclosed method significantly shortens the synthetic route compared to conventional multi-step processes, utilizing readily available starting materials like DGM1 and DGM2. It operates under mild reaction conditions with high isolated yields, simplifying purification and reducing overall production time.
Q: How does this process address impurity control in pharmaceutical intermediates?
A: By employing specific condensing agents like EDCI and HOBt, the reaction minimizes side reactions typically associated with harsher coupling methods. The subsequent recrystallization steps described in the patent ensure high purity specifications suitable for downstream API synthesis.
Q: Is this synthesis method suitable for large-scale industrial manufacturing?
A: Yes, the process is designed for scalability, utilizing common solvents like dichloromethane, ethanol, and acetone which are easy to recover. The operational simplicity and robust yield profiles make it highly viable for commercial scale-up of complex pharmaceutical intermediates.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dabigatran Etexilate Supplier
At NINGBO INNO PHARMCHEM, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project moves seamlessly from bench to plant. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that validate every batch against international pharmacopeia standards. We understand the critical nature of anticoagulant supply chains and are equipped to handle the complex chemistry required for Dabigatran Etexilate and its analogues with precision and reliability.
We invite you to engage with our technical procurement team to discuss how this innovative synthesis route can be tailored to your specific volume requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper visibility into the potential economic benefits for your organization. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will empower your decision-making process.
