Advanced Synthetic Route for 6-Hydroxy-1-indanone: Overcoming Legacy Process Limitations for Commercial Scale-up
The pharmaceutical and fine chemical industries constantly seek robust pathways for constructing complex heterocyclic scaffolds, and the synthesis of 6-hydroxy-1-indanone represents a critical challenge in this domain. Patent CN101597226A introduces a transformative methodology that addresses the longstanding inefficiencies associated with traditional indanone synthesis. By leveraging a direct intramolecular cyclization strategy mediated by trifluoromethanesulfonic acid, this technology bypasses the cumbersome multi-step sequences previously required to install the hydroxyl functionality on the indanone core. This innovation is particularly significant for manufacturers aiming to streamline their supply chains for high-purity pharmaceutical intermediates, as it replaces scarce starting materials with readily available commodity chemicals like phenol.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of 6-hydroxy-1-indanone has been plagued by reliance on convoluted synthetic routes that suffer from poor atom economy and difficult precursor sourcing. One prevalent legacy method involves the use of 6-amino-1-indanone as a starting material, subjecting it to a Sandmeyer reaction to convert the amino group into a hydroxyl group. This approach is inherently flawed because the precursor itself requires a multi-step synthesis, adding significant time and cost to the overall process while generating substantial hazardous waste associated with diazonium chemistry. Another common pathway starts with 6-methoxy-1-indanone, necessitating a harsh demethylation step to reveal the phenol, which often requires aggressive reagents and careful control to prevent side reactions or over-reaction.
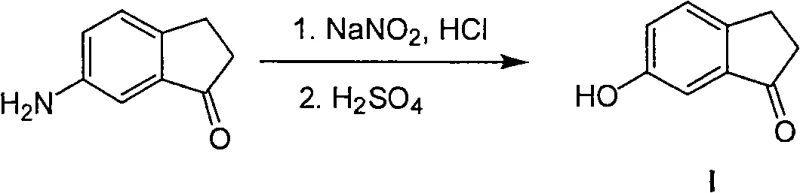
Furthermore, alternative literature methods, such as those described by Corey, often utilize specialized reagents that are not only expensive but also difficult to procure in bulk quantities, creating bottlenecks for commercial scale-up. These conventional pathways collectively result in extended lead times, increased operational expenditures due to multiple isolation steps, and a larger environmental footprint, making them less attractive for modern green chemistry initiatives in API manufacturing.
The Novel Approach
In stark contrast, the novel approach detailed in the patent utilizes a concise two-step sequence that begins with the acylation of phenol, a ubiquitous and low-cost industrial feedstock. The key innovation lies in the subsequent cyclization step, where phenyl acrylate undergoes an intramolecular Friedel-Crafts type acylation promoted by trifluoromethanesulfonic acid. This method effectively constructs the five-membered ring of the indanone system while simultaneously preserving the phenolic hydroxyl group, thereby eliminating the need for protection-deprotection strategies or functional group interconversions found in older methods. The process is characterized by its operational simplicity, requiring only standard heating and extraction workups, which significantly reduces the technical barrier for implementation in diverse manufacturing facilities.
Mechanistic Insights into TfOH-Catalyzed Intramolecular Cyclization
The core of this synthetic breakthrough is the activation of the acrylate double bond by the superacidic nature of trifluoromethanesulfonic acid (TfOH). In this mechanism, the strong proton donor capability of TfOH protonates the beta-carbon of the acrylate moiety or coordinates with the carbonyl oxygen, generating a highly electrophilic species. This activated intermediate is then susceptible to nucleophilic attack by the electron-rich aromatic ring of the phenol group at the ortho-position relative to the ester linkage. This intramolecular electrophilic aromatic substitution closes the five-membered ring, forming the indanone skeleton directly. The use of excess TfOH (5-20 equivalents) ensures that the equilibrium is driven towards the cyclized product and helps to solubilize the organic substrates in the highly polar acidic medium.
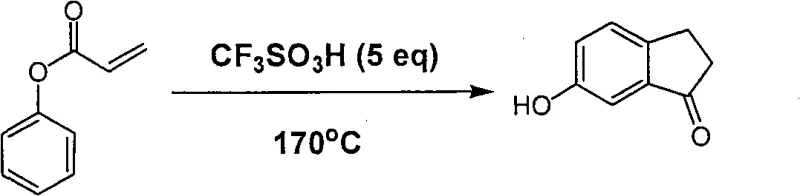
From an impurity control perspective, this mechanism offers distinct advantages by minimizing the formation of regioisomers. Since the tethering of the acrylate chain to the phenol oxygen dictates the proximity of the reaction centers, the cyclization is highly regioselective for the 6-position. Additionally, the high reaction temperatures (120-170°C) facilitate the overcoming of activation energy barriers for the ring closure, ensuring complete conversion of the intermediate ester. The robustness of the reaction conditions allows for a cleaner crude profile, simplifying downstream purification and ensuring that the final API intermediate meets stringent purity specifications required by regulatory bodies.
How to Synthesize 6-Hydroxy-1-indanone Efficiently
The execution of this synthesis is straightforward and amenable to standard laboratory glassware or reactor setups. The process initiates with the formation of phenyl acrylate under mild basic conditions, followed by the critical high-temperature acid-mediated cyclization. Operators must exercise caution during the second step due to the corrosive nature of trifluoromethanesulfonic acid and the elevated temperatures required. Detailed standard operating procedures regarding stoichiometry, addition rates, and safety protocols for handling strong acids are essential for successful replication. For a comprehensive guide on the specific molar ratios and workup procedures validated in the patent examples, please refer to the structured synthesis guide below.
- Prepare phenyl acrylate by reacting phenol with acryloyl chloride in anhydrous methylene chloride using triethylamine as a base at 0°C to room temperature.
- Dissolve the resulting phenyl acrylate in trifluoromethanesulfonic acid (5-20 equivalents) and heat the mixture to 120-170°C under nitrogen protection to induce cyclization.
- Quench the reaction in ice water, extract with dichloromethane, wash with saturated sodium bicarbonate and brine, dry over sodium sulfate, and purify via silica gel chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route presents a compelling value proposition centered on raw material security and process efficiency. By shifting the starting point to phenol and acryloyl chloride, manufacturers can decouple their supply chains from the volatile markets surrounding specialized amino-indanone derivatives. This transition to commodity chemicals not only stabilizes pricing but also ensures a consistent supply of feedstock, mitigating the risk of production stoppages due to precursor shortages. Furthermore, the reduction in the number of synthetic steps directly correlates to lower operational costs, as fewer unit operations mean reduced labor, energy, and solvent consumption per kilogram of finished product.
- Cost Reduction in Manufacturing: The elimination of expensive and multi-step precursor synthesis significantly lowers the overall cost of goods sold. By avoiding the Sandmeyer reaction or demethylation steps, the process removes the need for costly reagents like nitrites or strong demethylating agents, as well as the associated waste disposal costs. The streamlined workflow reduces the total processing time, allowing for higher throughput in existing manufacturing assets without the need for capital-intensive equipment upgrades.
- Enhanced Supply Chain Reliability: Sourcing phenol and acryloyl chloride is far more reliable than procuring niche intermediates like 6-methoxy-1-indanone, which may have limited global suppliers. This diversification of the supply base enhances resilience against geopolitical disruptions or single-source supplier failures. Additionally, the simplified logistics of handling fewer distinct chemical inputs reduce the complexity of inventory management and regulatory compliance documentation.
- Scalability and Environmental Compliance: The process is designed for easy amplification from laboratory to commercial scale, with post-treatment involving standard aqueous workups and extractions that are familiar to plant operators. The reduction in synthetic steps inherently minimizes the generation of hazardous byproducts and solvent waste, aligning with increasingly strict environmental regulations and sustainability goals. This greener profile facilitates smoother regulatory approvals and reduces the long-term liability associated with waste management.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented technology. These insights are derived directly from the experimental data and technical disclosures within the patent documentation to assist decision-makers in evaluating feasibility. Understanding these nuances is crucial for R&D teams planning technology transfer and for procurement teams assessing vendor capabilities.
Q: What are the primary advantages of this synthetic route over traditional Sandmeyer methods?
A: This route eliminates the need for difficult-to-obtain precursors like 6-amino-1-indone or 6-methoxy-1-indone. It utilizes commodity chemicals (phenol and acryloyl chloride) and achieves the core indanone structure in fewer steps with simpler post-treatment.
Q: What are the critical reaction conditions for the cyclization step?
A: The cyclization requires a strong acid promoter, specifically trifluoromethanesulfonic acid (TfOH), used in excess (5-20 equivalents). The reaction temperature is critical, typically ranging between 120°C and 170°C, to ensure efficient ring closure.
Q: Is this process suitable for large-scale manufacturing?
A: Yes, the patent highlights that the method is easy to amplify and suitable for fast laboratory preparation which translates well to pilot scales. The use of standard extraction and chromatography purification techniques supports scalable production workflows.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Hydroxy-1-indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the broader context of drug development and commercial manufacturing. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from bench-scale discovery to full-scale manufacturing is seamless and robust. We are committed to delivering high-purity 6-hydroxy-1-indanone that adheres to stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation to verify identity and assay.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthetic route can be integrated into your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits tailored to your volume needs. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that demonstrate our capability to be your trusted partner in advanced chemical synthesis.
