Revolutionizing 6-Hydroxy-1-indanone Production: A Direct Cyclization Strategy for Commercial Scale-up
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient, cost-effective, and safer synthetic routes. A pivotal advancement in this domain is detailed in patent CN101597226B, which introduces a novel method for synthesizing 6-hydroxy-1-indanone (also referred to as 6-hydroxyl-1-anisindione). This compound serves as a critical building block in medicinal chemistry and complex organic synthesis, yet its historical production has been plagued by inefficiencies. The patented technology addresses these challenges by replacing convoluted multi-step sequences with a streamlined, direct cyclization strategy. By leveraging the potent catalytic properties of trifluoromethanesulfonic acid (TfOH), this innovation transforms simple, commercially available starting materials—phenol and acryloyl chloride—into the target indanone scaffold with remarkable efficiency. For R&D directors and procurement specialists alike, this represents a significant opportunity to optimize supply chains and reduce the cost of goods sold (COGS) for downstream API manufacturing.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of 6-hydroxy-1-indanone has relied on methodologies that are inherently cumbersome and fraught with operational hazards. One prevalent traditional route, often cited in literature such as Farmaco Ed. Sci. (1983), involves the use of 6-amino-1-indanone as a starting material. This approach necessitates a Sandmeyer reaction to convert the amino group into a hydroxyl group. The Sandmeyer reaction is notorious for its requirement of unstable diazonium salts, which pose significant safety risks on a large scale due to their potential for explosive decomposition. Furthermore, the precursor 6-amino-1-indanone itself is not a commodity chemical; it must be synthesized through multiple steps, compounding the overall cost and time investment. Another common method involves the demethylation of 6-methoxy-1-indanone. While chemically feasible, this route shifts the bottleneck to the availability of the methoxy precursor, which also requires a multi-step synthesis to produce. These legacy methods create a fragile supply chain where delays in early-stage intermediates can halt entire production lines.
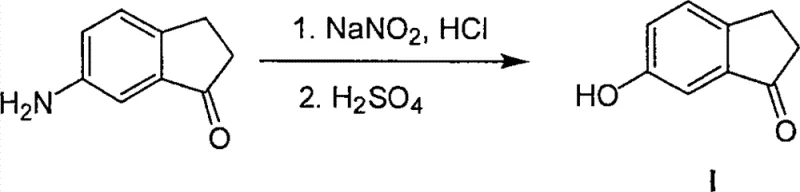
The Novel Approach
In stark contrast to these legacy pathways, the methodology disclosed in CN101597226B offers a paradigm shift by utilizing a direct construction of the indanone core from phenol. This new approach bypasses the need for pre-functionalized indanone precursors entirely. Instead, it employs a two-step sequence: first, the esterification of phenol with acryloyl chloride to form phenyl acrylate, followed by an intramolecular cyclization mediated by trifluoromethanesulfonic acid. This strategy is groundbreaking because it utilizes phenol, one of the most abundant and inexpensive petrochemical feedstocks available globally. By eliminating the dependency on scarce or difficult-to-synthesize starting materials, this method drastically simplifies the procurement landscape. The reaction conditions, while requiring strong acid catalysis, are operationally straightforward, involving standard heating and extraction workups that are easily adaptable to existing reactor infrastructure in fine chemical plants.
Mechanistic Insights into TfOH-Catalyzed Intramolecular Acylation
The core of this technological breakthrough lies in the utilization of trifluoromethanesulfonic acid (TfOH) as both a solvent and a superacid catalyst. TfOH is one of the strongest known Brønsted acids, possessing a Hammett acidity function far superior to sulfuric acid. In the context of this synthesis, TfOH plays a dual role: it activates the carbonyl group of the acrylate ester towards electrophilic attack and facilitates the subsequent aromatization or tautomerization steps required to form the stable ketone. The mechanism likely proceeds via the protonation of the carbonyl oxygen of the phenyl acrylate intermediate, generating a highly reactive acylium ion species. This electrophile then undergoes an intramolecular Friedel-Crafts acylation at the ortho-position of the phenol ring. The high temperature range of 120-170°C specified in the patent is crucial; it provides the necessary activation energy to overcome the kinetic barrier of this cyclization, ensuring that the reaction proceeds to completion rather than stalling at intermediate stages.
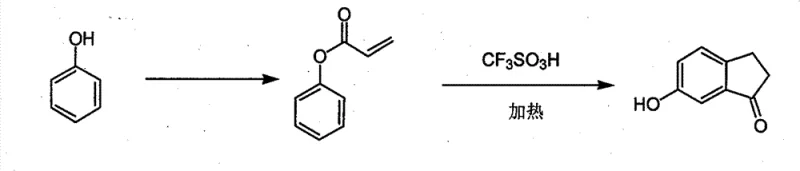
From an impurity control perspective, this mechanism offers distinct advantages over metal-catalyzed alternatives. Traditional Friedel-Crafts reactions often employ Lewis acids like aluminum chloride, which generate stoichiometric amounts of hazardous waste and can lead to complex metal-chelated impurities that are difficult to remove. The use of TfOH, while corrosive, avoids the introduction of transition metals into the product stream. This is particularly vital for pharmaceutical intermediates, where residual metal limits are strictly regulated by ICH guidelines. The patent data indicates that by optimizing the equivalent ratio of TfOH (between 5 to 20 equivalents) and maintaining the temperature at 170°C, yields of approximately 38-40% can be achieved. While the yield might appear moderate compared to some idealized lab reactions, the simplicity of the workup—quenching in ice water and extracting with dichloromethane—allows for high purity recovery without the need for complex distillation or recrystallization protocols that often degrade sensitive molecules.
How to Synthesize 6-Hydroxy-1-indanone Efficiently
Implementing this synthesis in a laboratory or pilot plant setting requires careful attention to the exothermic nature of the initial esterification and the corrosive environment of the cyclization step. The process begins with the formation of phenyl acrylate, where phenol is dissolved in anhydrous methylene chloride and treated with triethylamine as a base scavenger. Acryloyl chloride is added dropwise at 0°C to control the exotherm, followed by stirring at room temperature. Once the ester is isolated, the critical cyclization step commences. The ester is dissolved directly into trifluoromethanesulfonic acid, and the mixture is heated to 170°C under an inert nitrogen atmosphere. Detailed standardized operating procedures regarding safety handling of TfOH and specific quenching protocols are essential for safe execution.
- React phenol with acryloyl chloride in the presence of triethylamine in anhydrous methylene chloride at 0°C to room temperature to form phenyl acrylate.
- Dissolve the resulting phenyl acrylate in trifluoromethanesulfonic acid (5-20 equivalents) and heat the mixture to 120-170°C under nitrogen protection.
- Quench the reaction in ice water, extract with dichloromethane, wash, dry, and purify via silica gel column chromatography to isolate 6-hydroxy-1-indanone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical elegance. The primary advantage lies in the raw material profile. By anchoring the synthesis on phenol and acryloyl chloride, manufacturers can leverage the massive global production capacity of these commodity chemicals. Unlike specialized intermediates that may have single-source suppliers and long lead times, phenol is produced in millions of tons annually for the plastics and resin industries, ensuring a stable and competitive pricing structure. This shift effectively decouples the production of 6-hydroxy-1-indanone from the bottlenecks associated with niche fine chemical suppliers, thereby enhancing supply chain resilience against market volatility.
- Cost Reduction in Manufacturing: The economic implications of this route are profound. By eliminating the multi-step synthesis of precursors like 6-amino-1-indanone or 6-methoxy-1-indanone, the overall processing time and labor costs are significantly reduced. Furthermore, the avoidance of expensive transition metal catalysts removes the need for costly metal scavenging steps and the associated waste disposal fees. Although trifluoromethanesulfonic acid is a premium reagent, its use in a recyclable or recoverable manner, combined with the low cost of the carbon backbone sources, results in a favorable overall cost structure. The simplified purification process further contributes to cost efficiency by reducing solvent consumption and energy usage during isolation.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the complexity of synthetic routes. Each additional step in a traditional synthesis introduces a potential point of failure, whether it be a low-yielding reaction or a difficult purification. This two-step process minimizes these risks. The robustness of the reaction conditions means that batch-to-batch variability is reduced, leading to more predictable production schedules. Additionally, the use of common solvents like methylene chloride and standard extraction techniques ensures that the process can be transferred between different manufacturing sites with minimal requalification effort, providing flexibility in sourcing and production location.
- Scalability and Environmental Compliance: As regulatory pressures regarding environmental impact intensify, the choice of synthetic route becomes a compliance issue as well as an economic one. This method avoids the generation of heavy metal waste streams associated with Lewis acid catalysts and eliminates the hazardous diazonium byproducts of Sandmeyer chemistry. The waste profile is primarily acidic aqueous waste and organic solvents, which are well-understood and manageable within modern wastewater treatment facilities. The scalability of the process is supported by the fact that high-temperature acid-mediated cyclizations are unit operations commonly found in bulk chemical manufacturing, allowing for seamless scale-up from pilot batches to multi-ton commercial production without the need for specialized equipment.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and technical specifications outlined in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: What are the primary advantages of the TfOH cyclization method over traditional Sandmeyer routes?
A: The TfOH cyclization method eliminates the need for hazardous diazonium salts and multi-step precursor synthesis required in Sandmeyer reactions. It utilizes commodity chemicals like phenol and acryloyl chloride, significantly simplifying the supply chain and reducing raw material costs while improving operational safety.
Q: How does the reaction temperature affect the yield of 6-hydroxy-1-indanone?
A: According to patent data, temperature is a critical factor. Reactions conducted at 170°C achieve yields around 38-40%, whereas lowering the temperature to 120°C drastically reduces the yield to approximately 5%. Optimal thermal energy is required to drive the intramolecular acylation effectively.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is highly scalable due to the use of robust reagents and a straightforward two-step sequence. The avoidance of sensitive intermediates and the use of standard extraction and chromatography purification techniques make it amenable to commercial scale-up from kilogram to tonnage levels.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 6-Hydroxy-1-indanone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from a patented laboratory method to a robust commercial process requires deep technical expertise and rigorous quality control. As a leading CDMO and supplier in the fine chemical sector, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our facility is equipped to handle the corrosive nature of superacid chemistry safely and efficiently, ensuring that the benefits of the CN101597226B route are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of 6-hydroxy-1-indanone meets the exacting standards required for pharmaceutical applications.
We invite procurement leaders and R&D teams to collaborate with us to evaluate the feasibility of this cost-effective route for your specific projects. By partnering with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume requirements. We encourage you to reach out today to obtain specific COA data and comprehensive route feasibility assessments, ensuring that your supply chain is optimized for both performance and profitability in the competitive global market.
