Advanced Synthesis of Cefotiam Dihydrochloride: A High-Yield Route for Commercial API Manufacturing
Advanced Synthesis of Cefotiam Dihydrochloride: A High-Yield Route for Commercial API Manufacturing
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for the production of critical antibiotic intermediates. Patent CN101045733B, published in late 2011, introduces a transformative preparation method for Cefotiam Dihydrochloride, a second-generation cephalosporin antibiotic widely used for its potent activity against Gram-positive and Gram-negative bacteria. This technical disclosure addresses long-standing inefficiencies in the acylation of the cephalosporin nucleus, specifically targeting the 7-amino position of 7-ACMT (7-amino-3-[(1-(2-dimethylaminoethyl)-1H-tetrazol-5-yl)thiomethyl]-3-cephem-4-carboxylic acid). By shifting away from unstable acylating agents and complex protecting group manipulations, this innovation offers a direct, high-yield route that is exceptionally well-suited for large-scale commercial manufacturing. For R&D directors and procurement strategists, understanding this shift is crucial for optimizing supply chains and reducing the cost of goods sold (COGS) for downstream API production.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the introduction of the 2-amino-4-thiazole ethanoyl side chain at the 7-position of the cephalosporin nucleus has been fraught with chemical challenges. One prevalent conventional method, often referred to as Method One in technical literature, relies on the use of 4-chloro-3-oxobutyryl chloride (COBC) as the initial acylating agent. This approach necessitates a subsequent cyclization reaction with thiourea to form the thiazole ring. However, COBC is notoriously unstable, presenting significant hazards and logistical difficulties regarding transportation and storage, which severely hampers its suitability for continuous industrial production. Furthermore, the two-step reaction sequence involving COBC typically results in a cumulative total recovery rate of merely 50% to 60%. This low efficiency not only inflates raw material costs but also generates substantial waste, creating a burden on environmental compliance and downstream purification processes. The instability of the reagents combined with the mediocre yield creates a bottleneck that limits the scalability of this traditional route.
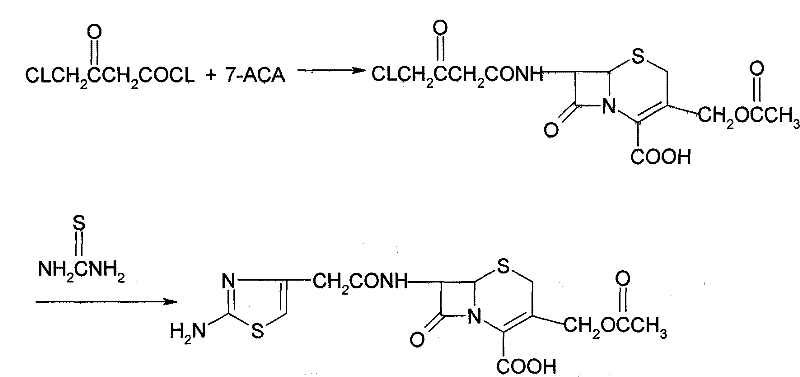
The Novel Approach
In stark contrast to the legacy methods, the novel approach detailed in the patent utilizes 2-(2-amino-4-thiazolyl)acetic acid (ATA) as the primary feedstock. This method bypasses the instability issues of COBC entirely by converting ATA directly into its hydrochloride salt form, ATC.HCl, through a controlled chlorination process. This activated species then serves as the acylating agent for 7-ACMT. The elimination of protecting group strategies, which were previously required to manage the reactivity of the amino group on the thiazole ring, represents a major process intensification. By removing the need for subsequent deprotection steps—whether enzymatic or acidic—the overall synthetic sequence is drastically shortened. This streamlining not only simplifies the operational workflow but also inherently boosts the overall yield, with the patent reporting yields reaching approximately 84%. This leap in efficiency transforms the economic landscape of producing this critical antibiotic intermediate.
Mechanistic Insights into ATC.HCl Mediated Acylation
The core of this technological advancement lies in the precise generation and utilization of the ATC.HCl intermediate. The mechanism begins with the suspension of ATA in a suitable solvent system, such as dichloromethane or acetonitrile, followed by the introduction of dry hydrogen chloride gas. This step protonates the amino group and activates the carboxylic acid functionality. Subsequent addition of a chlorinating agent, such as phosphorus pentachloride or phosphorus oxychloride, at controlled low temperatures (0 to 30°C) converts the acid into the corresponding acid chloride hydrochloride salt. This in-situ generation ensures high reactivity while minimizing decomposition. The resulting ATC.HCl is then reacted with 7-ACMT, which serves as the beta-lactam nucleus. The 7-ACMT itself is typically derived from 7-ACA through a substitution reaction with DMMT, as illustrated in the preparatory chemistry below.
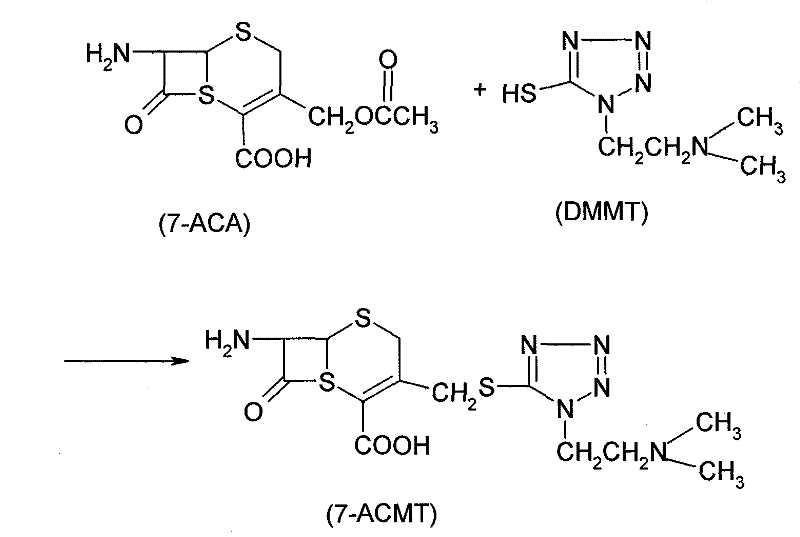
During the acylation phase, the 7-ACMT is dissolved in an aqueous alkaline solution to ensure the amino group at the 7-position is nucleophilic enough to attack the carbonyl carbon of the ATC.HCl. The reaction is maintained at temperatures between -10°C and 35°C to balance reaction kinetics with the stability of the beta-lactam ring. Following the acylation, the workup procedure is elegantly simple: the organic solvent is separated, and the aqueous phase containing the product is acidified with hydrochloric acid. The addition of a hydrophilic solvent, such as acetone or ethanol, in a specific ratio induces the crystallization of the final Cefotiam Dihydrochloride. This direct crystallization from the reaction mixture minimizes product loss and ensures high purity without the need for extensive chromatographic purification, a critical factor for GMP compliance.
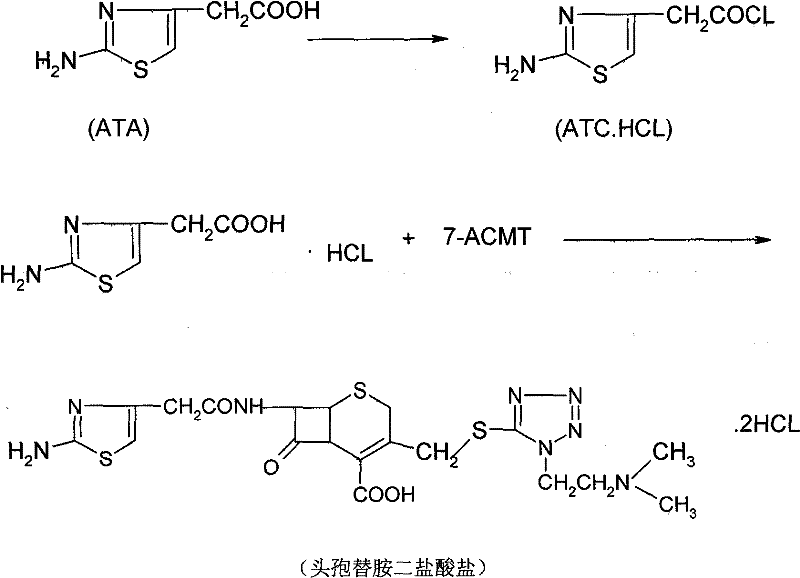
How to Synthesize Cefotiam Dihydrochloride Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and temperature control to maximize the formation of the ATC.HCl intermediate and its subsequent coupling efficiency. The process is designed to be robust, tolerating slight variations in conditions while maintaining high purity profiles essential for pharmaceutical applications. Operators must ensure the rigorous exclusion of moisture during the chlorination step to prevent hydrolysis of the activated acid chloride. The following guide outlines the standardized operational parameters derived from the patent examples, providing a clear roadmap for process engineers to replicate this high-yield methodology in a pilot or commercial plant setting.
- Prepare ATC.HCl by reacting ATA with dry hydrogen chloride gas and a chlorinating agent (e.g., phosphorus pentachloride) in a nitrile or chlorinated solvent at 0-30°C.
- Dissolve 7-ACMT in an aqueous alkaline solution and react with the prepared ATC.HCl at temperatures between -10°C and 35°C to perform the acylation.
- Separate the organic solvent, acidify the aqueous phase with hydrochloric acid, add a hydrophilic solvent like acetone, and crystallize the final Cefotiam Dihydrochloride product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this ATC-based synthesis route offers compelling strategic advantages that extend beyond simple yield metrics. The primary value driver is the stabilization of the supply chain through the use of robust, commercially available raw materials. Unlike the volatile COBC used in older methods, ATA is a stable solid that can be sourced reliably and stored without specialized containment, significantly reducing inventory risks and logistics costs. This stability translates directly into enhanced supply continuity, ensuring that production schedules for the downstream API are not disrupted by reagent degradation or supply shortages. Furthermore, the simplification of the process flow—specifically the removal of protecting group installation and removal steps—reduces the total number of unit operations, thereby lowering labor costs and equipment occupancy time.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the substantial increase in overall yield and the reduction in material consumption. By achieving yields in the mid-80% range compared to the 50-60% of conventional methods, the effective cost per kilogram of the active intermediate is drastically lowered. Additionally, the elimination of expensive protecting group reagents and the enzymes or acids required for their removal further strips away unnecessary cost layers. The process also utilizes common, recoverable solvents like dichloromethane and acetone, which facilitates solvent recycling programs and reduces waste disposal expenses, contributing to a leaner manufacturing cost structure.
- Enhanced Supply Chain Reliability: Reliability in the pharmaceutical supply chain is paramount, and this method enhances it by removing single points of failure associated with unstable reagents. The ability to generate the acylating agent in situ from stable precursors means that manufacturers are less dependent on Just-In-Time delivery of sensitive chemicals. This flexibility allows for larger batch sizes and longer campaign runs, optimizing plant utilization rates. Moreover, the robustness of the crystallization step ensures consistent product quality and particle size distribution, which is critical for downstream formulation processes, reducing the risk of batch rejection and ensuring a steady flow of material to the API finishing lines.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is superior due to its atom economy and reduced waste generation. The avoidance of heavy metal catalysts or toxic reagents simplifies the effluent treatment process, making it easier to meet stringent environmental regulations. The straightforward workup procedure, which relies on pH adjustment and solvent precipitation rather than complex extractions or chromatography, is inherently easier to scale from kilogram to multi-ton production. This scalability ensures that the process can grow with market demand without requiring disproportionate increases in capital expenditure for specialized waste treatment or purification infrastructure.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this patented synthesis method. These insights are derived directly from the experimental data and process descriptions provided in the patent documentation, offering clarity on yield expectations, solvent choices, and purity profiles. Understanding these details is essential for technical teams evaluating the feasibility of adopting this route for their specific manufacturing capabilities.
Q: What is the primary advantage of using ATC.HCl over the COBC method for Cefotiam synthesis?
A: The ATC.HCl method eliminates the need for unstable 4-chloro-3-oxobutyryl chloride (COBC) and avoids complex protecting group strategies, resulting in a significantly higher total yield (approx. 84% vs 50-60%) and simpler industrial operation.
Q: What solvents are recommended for the crystallization of Cefotiam Dihydrochloride in this process?
A: The patent specifies using hydrophilic solvents such as lower alcohols (ethanol, isopropanol) or ketones (acetone) added in a volume 3 to 6 times that of the water layer to induce effective crystallization of the dihydrochloride salt.
Q: How does this method improve supply chain stability for antibiotic intermediates?
A: By utilizing stable raw materials like ATA and avoiding the transportation risks associated with unstable acylating agents like COBC, this method ensures consistent production schedules and reduces the risk of batch failures due to reagent degradation.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefotiam Dihydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize that the adoption of advanced synthetic routes like the one described in CN101045733B is key to maintaining competitiveness in the global antibiotic market. As a leading CDMO and supplier, we possess the technical expertise to translate these laboratory-scale innovations into robust, commercial-scale processes. Our facilities are equipped to handle complex acylation reactions and precise crystallization protocols, ensuring that we can deliver high-purity Cefotiam Dihydrochloride that meets the most rigorous pharmacopoeial standards. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, leveraging our rigorous QC labs to guarantee stringent purity specifications and consistent impurity profiles for every batch we produce.
We invite pharmaceutical partners to collaborate with us to leverage this cost-effective and scalable technology for their supply chains. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality needs. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our optimized manufacturing capabilities can drive value and reliability for your antibiotic portfolio.
