Revolutionizing Veterinary Antibiotic Production: The GCLE-Based Synthesis of Cefquinome Sulfate
The pharmaceutical landscape for veterinary antibiotics is constantly evolving, driven by the need for more efficient, cost-effective, and high-purity manufacturing processes. A pivotal advancement in this domain is detailed in Chinese Patent CN102002058B, which discloses a novel synthetic method for Cefquinome sulfate, a fourth-generation cephalosporin exclusively designed for animal health. This patent represents a significant departure from traditional methodologies by utilizing GCLE (7-phenylacetyl amido-3-chloromethyl cephalosporanic acid p-methoxybenzyl ester) as the foundational starting material. Unlike conventional routes that rely heavily on 7-ACA, this innovative approach leverages the inherent reactivity of the C-3 chloromethyl group on the GCLE nucleus. By substituting the chlorine with iodine and subsequently reacting with 5,6,7,8-tetrahydroquinoline, the process achieves a streamlined pathway that effectively circumvents the harsh reaction conditions and complex purification challenges associated with legacy technologies. For R&D directors and procurement managers alike, this development signals a robust opportunity to optimize the supply chain for high-value veterinary intermediates while ensuring stringent quality standards.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Cefquinome has been dominated by routes originating from 7-Aminocephalosporanic Acid (7-ACA), as exemplified by prior art such as U.S. Patent 4845087 and 4667028. These conventional pathways are fraught with significant operational inefficiencies that pose challenges for large-scale manufacturing. A primary drawback is the reliance on expensive and sensitive reagents like iodotrimethylsilane for silylation reactions, which necessitates strictly anhydrous and oxygen-free environments. Such苛刻 requirements not only escalate equipment costs but also introduce substantial risks regarding batch-to-batch consistency and safety. Furthermore, these traditional methods often involve multiple solvent switches and complex protection-deprotection sequences that generate considerable waste streams. The resulting reaction mixtures are notoriously difficult to purify, frequently yielding products with unsatisfactory color and purity profiles that require extensive downstream processing. These factors collectively contribute to a high cost of goods sold (COGS) and extended lead times, creating bottlenecks for reliable agrochemical intermediate supplier networks aiming to meet global demand.
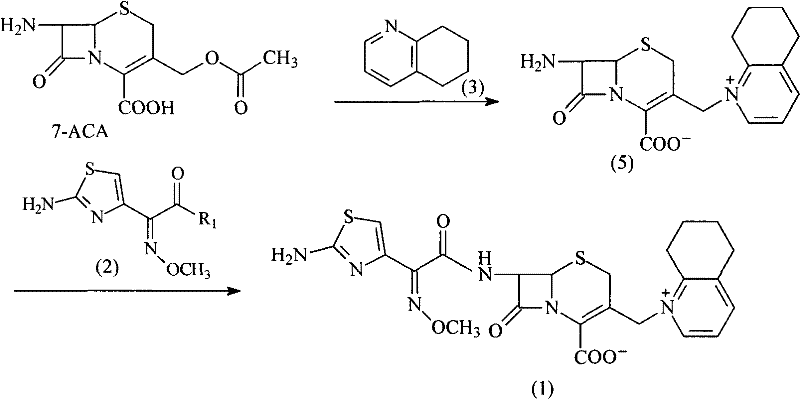
The Novel Approach
In stark contrast, the methodology outlined in CN102002058B introduces a paradigm shift by employing GCLE, a derivative that inherently possesses protected C-4 carboxyl and C-7 amino groups. This structural advantage eliminates the need for the cumbersome protection steps required when starting from 7-ACA. The core innovation lies in the nucleophilic substitution at the C-3 position, where the chloromethyl group is first activated via iodination using potassium iodide (KI) and then displaced by 5,6,7,8-tetrahydroquinoline. This sequence proceeds under remarkably mild conditions, typically ranging from room temperature to -10°C, thereby significantly reducing energy consumption and thermal stress on the beta-lactam ring. The subsequent deprotection using phosphorus pentachloride and phenol, followed by acylation with the AE-active ester, is executed with high selectivity. This streamlined workflow not only simplifies the operational protocol but also enhances the overall yield and product quality, making it an ideal candidate for cost reduction in veterinary drug manufacturing.
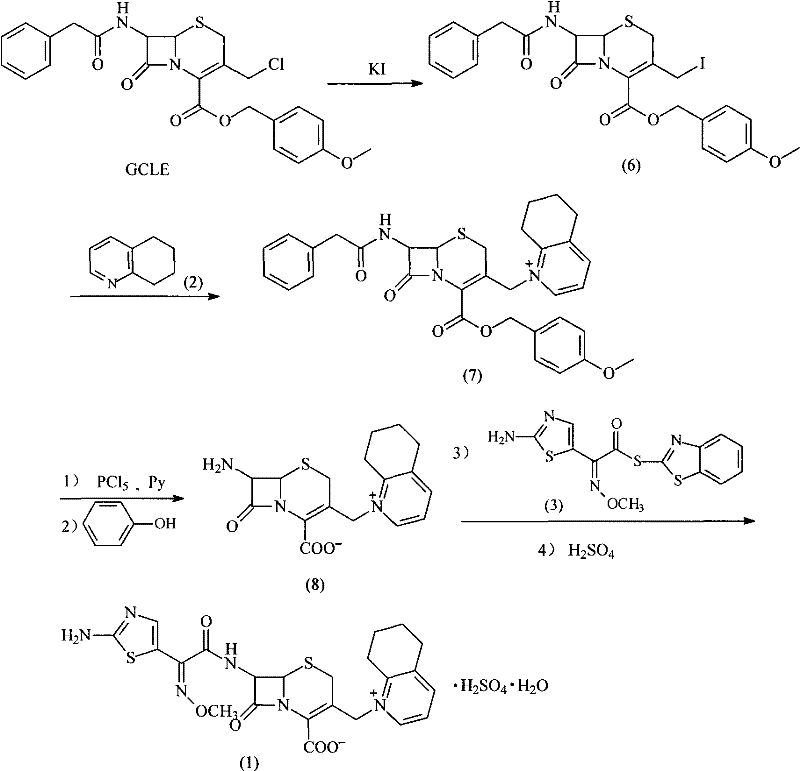
Mechanistic Insights into GCLE-Based Nucleophilic Substitution
The chemical elegance of this process is rooted in the enhanced reactivity of the GCLE nucleus. The presence of the chloromethyl group at the C-3 position serves as a superior leaving group precursor compared to the acetoxymethyl group found in 7-ACA derivatives. In the initial step, the displacement of chlorine by iodine generates an intermediate iodomethyl species in situ, which is significantly more electrophilic. This facilitates a rapid and clean quaternization reaction with the nitrogen atom of 5,6,7,8-tetrahydroquinoline. The mechanism avoids the formation of unstable silyl intermediates, which are prone to hydrolysis and side reactions in conventional routes. By maintaining the integrity of the beta-lactam ring through mild temperature control and avoiding strong Lewis acids until the specific deprotection stage, the process minimizes the formation of ring-opened impurities. This mechanistic precision is crucial for R&D teams focused on impurity profiling, as it ensures a cleaner reaction profile that simplifies the isolation of the target molecule.
Furthermore, the deprotection strategy utilizing phosphorus pentachloride (PCl5) and phenol is meticulously designed to remove the phenylacetyl and p-methoxybenzyl protecting groups without compromising the newly formed quaternary ammonium structure. The reaction likely proceeds through the formation of an acid chloride intermediate at the C-4 position, which is then hydrolyzed or alcoholized to reveal the free carboxylic acid. Simultaneously, the amide bond at the C-7 position is cleaved to liberate the free amino group necessary for the final acylation. The use of phenol acts as a scavenger and stabilizer during this acidic phase, preventing degradation of the sensitive cephalosporin core. This controlled deprotection ensures that the resulting intermediate (8) retains high optical purity and structural fidelity, setting the stage for the final coupling reaction with the AE-active ester to proceed with high efficiency and minimal epimerization.
How to Synthesize Cefquinome Sulfate Efficiently
The practical implementation of this synthesis involves a carefully orchestrated sequence of reactions that balance reactivity with stability. The process begins with the halogen exchange and quaternization in a suitable organic solvent such as dichloromethane or acetone, followed by a precise deprotection sequence at low temperatures. The final step involves coupling with the side chain active ester under basic conditions to form the sulfate salt. Each stage is monitored via TLC or HPLC to ensure complete conversion before proceeding, thereby maximizing yield and minimizing waste. For technical teams looking to implement this route, attention to solvent selection and temperature gradients is paramount to replicate the high purity reported in the patent examples.
- React GCLE with Potassium Iodide (KI) followed by 5,6,7,8-tetrahydroquinoline to form the quaternary ammonium intermediate.
- Perform deprotection of the C-7 amino and C-4 carboxyl groups using Phosphorus Pentachloride (PCl5) and Phenol.
- Couple the resulting intermediate with AE-active ester in the presence of triethylamine and crystallize as the sulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this GCLE-based route offers transformative benefits for procurement managers and supply chain heads tasked with optimizing the sourcing of veterinary pharmaceutical intermediates. The most immediate impact is observed in raw material costs; GCLE is derived from penicillin G through well-established fermentation and enzymatic processes, making it significantly more abundant and cost-effective than 7-ACA. The patent explicitly notes that the price of GCLE is merely a fraction of 7-ACA, which directly translates to substantial cost savings in the bill of materials. Moreover, the elimination of expensive silylating reagents like iodotrimethylsilane further reduces the input costs. This economic efficiency is compounded by the simplified process flow, which requires fewer unit operations and less solvent consumption, thereby lowering utility and waste disposal expenses. For organizations focused on cost reduction in pharmaceutical intermediates manufacturing, this route presents a compelling value proposition.
- Cost Reduction in Manufacturing: The transition to a GCLE-based synthesis fundamentally alters the cost structure of Cefquinome production. By bypassing the need for costly protection groups and expensive silylating agents, the process achieves a leaner manufacturing footprint. The use of common reagents like potassium iodide and phosphorus pentachloride, which are commodity chemicals with stable pricing, insulates the production process from the volatility associated with specialty reagents. Additionally, the high yields reported in the patent examples indicate a more efficient utilization of starting materials, reducing the effective cost per kilogram of the final API. This efficiency allows manufacturers to offer more competitive pricing without sacrificing margins, a critical factor in the highly price-sensitive veterinary market.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the availability of GCLE. As a mature intermediate with a decade-long production history, GCLE benefits from a robust global supply network, unlike some niche reagents required for 7-ACA based routes. The simplicity of the reaction conditions—operating at near-ambient or mildly cooled temperatures—reduces the dependency on specialized cryogenic equipment, making the process easier to transfer between different manufacturing sites. This flexibility ensures continuity of supply even in the face of regional disruptions. Furthermore, the reduced complexity of the purification steps means that production cycles are shorter, enabling faster turnaround times and reducing lead time for high-purity veterinary antibiotics.
- Scalability and Environmental Compliance: The environmental profile of this synthesis is markedly improved, aligning with modern green chemistry principles and regulatory expectations. The avoidance of hazardous silylating agents reduces the generation of silicon-containing waste, which can be difficult to treat. The process relies on standard extraction and crystallization techniques that are easily scalable from pilot plant to commercial tonnage. The use of solvents like dichloromethane and acetone, while requiring proper management, allows for efficient recovery and recycling systems. This scalability ensures that the commercial scale-up of complex veterinary intermediates can be achieved with minimal environmental impact, facilitating smoother regulatory approvals and long-term sustainability of the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis of Cefquinome sulfate using the GCLE route. These insights are derived directly from the experimental data and comparative analysis provided in the patent literature, offering clarity on the feasibility and advantages of this method for industry stakeholders.
Q: Why is GCLE preferred over 7-ACA for Cefquinome synthesis?
A: GCLE offers pre-protected C-4 and C-7 positions, eliminating the need for complex protection/deprotection steps required with 7-ACA. Additionally, the C-3 chloromethyl group is highly reactive, allowing for milder reaction conditions and avoiding expensive silylating agents.
Q: What are the key purity advantages of this new method?
A: The process utilizes specific crystallization steps after the C-3 substitution and final acylation, effectively removing impurities. The avoidance of harsh silylation reagents reduces side reactions, resulting in superior product color and higher HPLC purity.
Q: Is this process suitable for large-scale industrial production?
A: Yes, the method uses inexpensive and readily available raw materials like GCLE and KI. The reaction conditions are mild (ranging from room temperature to -10°C), and the work-up involves standard extraction and crystallization, making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefquinome Sulfate Supplier
The technological advancements detailed in CN102002058B underscore the potential for high-efficiency production of fourth-generation cephalosporins. At NINGBO INNO PHARMCHEM, we leverage our extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production to bring such innovative processes to life. Our state-of-the-art facilities are equipped to handle the specific requirements of beta-lactam chemistry, ensuring that every batch meets stringent purity specifications. With our rigorous QC labs and commitment to process optimization, we are uniquely positioned to deliver high-purity Cefquinome sulfate that adheres to the highest international standards, providing our partners with a secure and quality-assured supply source.
We invite forward-thinking pharmaceutical companies to collaborate with us to unlock the full commercial potential of this synthesis route. Our technical team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating exactly how this GCLE-based method can enhance your bottom line. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and economic efficiency.
