Optimizing Cefquinome Sulfate Production via Streamlined 7-ACA Modification for Veterinary Applications
Optimizing Cefquinome Sulfate Production via Streamlined 7-ACA Modification for Veterinary Applications
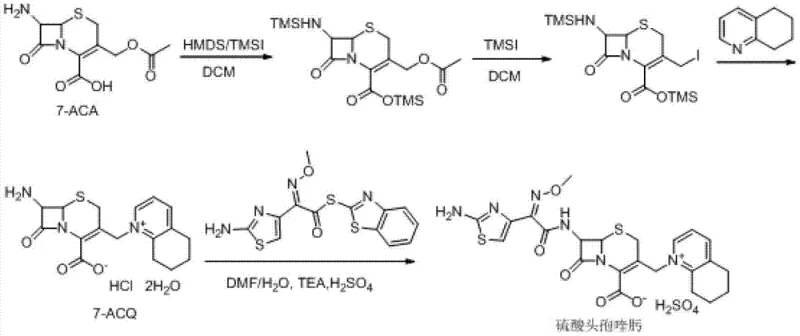
The global demand for fourth-generation cephalosporin antibiotics in the veterinary sector continues to rise, driven by the need for effective treatments against resistant bacterial strains in livestock. Patent CN103193799A introduces a transformative chemical synthesis method for Cefquinome Sulfate, a critical animal-specific antibiotic known for its broad spectrum and low toxicity. This innovation addresses longstanding inefficiencies in manufacturing by utilizing 7-aminocephalosporanic acid (7-ACA) as a starting material and employing a novel one-pot quaternization strategy. The technical breakthrough lies in the strategic use of hexamethyldisilazane (HMDS) and catalytic iodotrimethylsilane (TMSI) to protect functional groups, followed by a streamlined reaction with 5,6,7,8-tetrahydroquinoline. For procurement leaders and R&D directors, this patent represents a significant opportunity to optimize production costs while maintaining stringent quality standards required for veterinary drug registration.
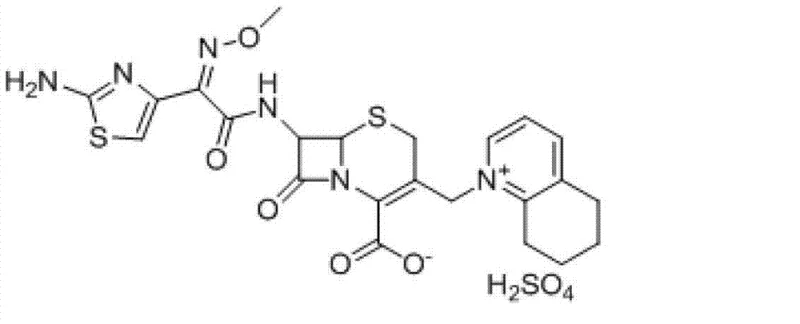
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Cefquinome Sulfate has been plagued by high operational costs and complex purification steps that hinder scalability. As illustrated in the prior art pathways, traditional Route One relies on the initial formation of cefotaxime acid, which is notoriously unstable and prone to degradation, leading to yield losses. Furthermore, this route necessitates the use of excessive amounts of iodotrimethylsilane (up to 7 equivalents) and 5,6,7,8-tetrahydroquinoline (up to 9 equivalents), both of which are high-cost reagents that severely impact the overall manufacturing budget. Additionally, the final salt formation step in Route One requires anion-exchange resins like Amberlite LA-2, which are often imported materials subject to supply chain volatility and price fluctuations. Route Two attempts to mitigate some issues by reacting 7-ACA directly but still suffers from high reagent consumption, requiring 5 equivalents of TMSI and 7 equivalents of the quinoline derivative. Route Three utilizes GCLE as a starting material, which is less readily available and more expensive than 7-ACA, and involves harsh deprotection conditions using phosphorus pentachloride and phenol, posing safety and environmental challenges.
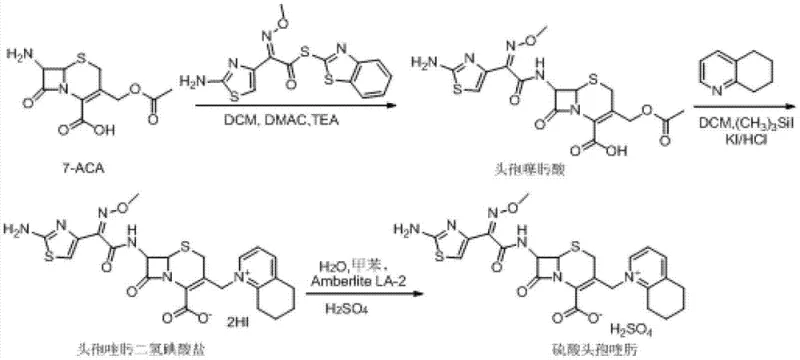
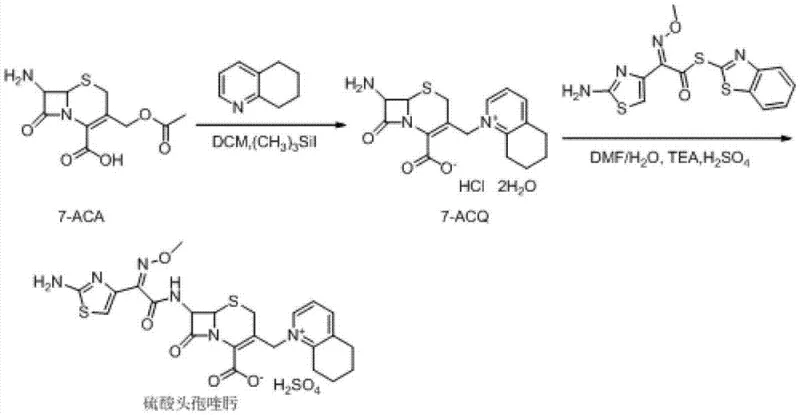
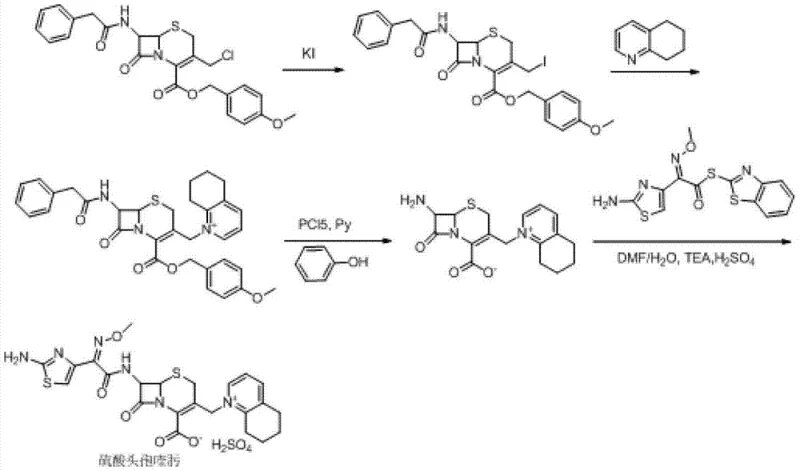
The Novel Approach
The patented methodology offers a robust alternative by fundamentally redesigning the reaction sequence to maximize atom economy and minimize reagent waste. By initiating the process with the silylation of 7-ACA using HMDS and a catalytic amount of TMSI, the amino and carboxyl groups are effectively protected without the need for stoichiometric excesses typically seen in older methods. The subsequent one-pot quaternization at the C-3 position allows for the direct introduction of the 5,6,7,8-tetrahydroquinoline moiety using significantly reduced molar ratios—specifically 1 to 3 equivalents compared to the 7 to 9 equivalents required previously. This approach not only lowers the raw material cost but also simplifies the workup procedure by eliminating the need for ion-exchange resin purification. The final acylation with AE-active ester and subsequent salt formation with sulfuric acid yields a product with high crystallinity and purity, suitable for direct formulation. This streamlined workflow demonstrates a clear path toward cost reduction in veterinary antibiotic manufacturing while ensuring consistent product quality.
Mechanistic Insights into Silylation and Quaternization Chemistry
The core of this synthesis lies in the precise control of silylation and nucleophilic substitution mechanisms within the cephalosporin nucleus. The initial reaction between 7-ACA and hexamethyldisilazane (HMDS), catalyzed by a trace amount of iodotrimethylsilane (TMSI), generates a bis-silylated intermediate where both the C-7 amino group and the C-4 carboxyl group are protected as trimethylsilyl derivatives. This protection is crucial as it enhances the solubility of the beta-lactam intermediate in organic solvents like dichloromethane and prevents unwanted side reactions during the subsequent quaternization step. The presence of the silyl groups also activates the C-3 acetoxymethyl leaving group, making it more susceptible to nucleophilic attack. When N,N-diethylaniline and additional TMSI are introduced, they facilitate the generation of a reactive iodomethyl species in situ, which is then rapidly displaced by the nitrogen lone pair of 5,6,7,8-tetrahydroquinoline. This quaternization forms the stable pyridinium salt structure characteristic of fourth-generation cephalosporins, locking the molecule into its bioactive conformation.
Impurity control is meticulously managed through pH regulation and solvent selection during the acylation and crystallization phases. The reaction of the 7-ACQ intermediate with the AE-active ester is conducted in a mixed solvent system of DMF and deionized water, with triethylamine used to maintain the pH between 8.0 and 9.0. This specific pH range is critical for maximizing the nucleophilicity of the C-7 amino group while minimizing hydrolysis of the beta-lactam ring, a common degradation pathway in cephalosporin synthesis. Following acylation, the product is isolated not through complex chromatography but via a straightforward extraction and acidification protocol. Adjusting the pH to 1.5–2.0 with sulfuric acid ensures the formation of the monosulfate salt, which precipitates efficiently upon the addition of isopropanol. HPLC monitoring throughout the process confirms that residual starting materials are kept below 2.0 mg/mL, ensuring that the final API meets the rigorous purity specifications demanded by regulatory bodies for veterinary medicines.
How to Synthesize Cefquinome Sulfate Efficiently
The implementation of this synthesis route requires careful attention to temperature control and reagent addition rates to ensure optimal yield and safety. The process begins with the reflux of 7-ACA in dichloromethane with HMDS and catalytic TMSI until the solution clarifies, indicating complete silylation. After cooling to 0°C, the quaternization reagents are added sequentially, maintaining low temperatures to prevent thermal degradation of the sensitive beta-lactam ring. The detailed standardized operating procedures, including exact molar ratios, stirring speeds, and crystallization times, are essential for reproducing the high yields reported in the patent embodiments. For manufacturing teams looking to adopt this technology, adhering to these specific parameters is vital for achieving the reported purity of >98% and yield of >85%.
- Protect 7-ACA amino and carboxyl groups using hexamethyldisilazane (HMDS) and catalytic iodotrimethylsilane (TMSI) in dichloromethane under reflux.
- Perform one-pot quaternization at the C-3 position by adding N,N-diethylaniline, additional TMSI, and 5,6,7,8-tetrahydroquinoline to form the 7-ACQ intermediate.
- React the 7-ACQ intermediate with AE-active ester in a DMF/water mixture, followed by acidification with sulfuric acid and crystallization with isopropanol to obtain the final sulfate salt.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this patented synthesis route offers tangible benefits that extend beyond simple chemistry improvements. The primary advantage is the substantial reduction in the consumption of high-value raw materials, specifically iodotrimethylsilane and 5,6,7,8-tetrahydroquinoline. By lowering the stoichiometric requirements from nearly 10 equivalents in traditional methods to just 1-3 equivalents, the direct material cost per kilogram of the active pharmaceutical ingredient is significantly decreased. This efficiency gain allows manufacturers to offer more competitive pricing structures to their downstream veterinary pharmaceutical clients, enhancing market share potential in a price-sensitive industry. Furthermore, the simplified process flow reduces the overall processing time and utility consumption, contributing to a leaner manufacturing operation.
- Cost Reduction in Manufacturing: The elimination of expensive anion-exchange resins such as Amberlite LA-2 removes a significant cost center and logistical hurdle. In previous methods, the reliance on imported resins introduced variability in supply and added substantial expense to the purification stage. By replacing this step with a direct acidification and crystallization protocol using sulfuric acid and isopropanol, the process becomes more self-contained and less dependent on external specialty chemical suppliers. Additionally, the use of 7-ACA as the starting material leverages a widely available and cost-effective bulk commodity, avoiding the premium pricing associated with specialized starting materials like GCLE used in other routes.
- Enhanced Supply Chain Reliability: The robustness of this synthetic route enhances supply chain continuity by reducing the number of critical raw materials that could become bottlenecks. Since the process avoids the use of unstable intermediates like cefotaxime acid, which degrades easily and complicates inventory management, production scheduling becomes more predictable. The ability to perform the quaternization in a one-pot manner reduces the number of isolation and purification steps, thereby minimizing material loss and increasing the overall throughput of the facility. This reliability is crucial for meeting the consistent demand cycles of the animal health market, where seasonal disease outbreaks can create sudden spikes in antibiotic requirements.
- Scalability and Environmental Compliance: From an environmental and safety perspective, this method offers a cleaner profile that facilitates easier regulatory approval and scale-up. The reduction in hazardous reagents like phosphorus pentachloride and phenol, which are required in alternative routes, lowers the burden on waste treatment facilities and reduces the risk of workplace exposure incidents. The solvent system primarily utilizes dichloromethane, DMF, and isopropanol, all of which are standard industrial solvents with well-established recovery and recycling protocols. This alignment with green chemistry principles not only reduces disposal costs but also positions the manufacturer as a responsible partner for global pharmaceutical companies increasingly focused on sustainability metrics in their supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Cefquinome Sulfate using this advanced methodology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing a factual basis for decision-making. Understanding these details helps stakeholders evaluate the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does this new synthesis route reduce raw material costs compared to traditional methods?
A: The patented process significantly reduces the molar equivalents of expensive reagents like iodotrimethylsilane (TMSI) and 5,6,7,8-tetrahydroquinoline. Traditional routes often require 5 to 9 equivalents of these reagents, whereas this method utilizes catalytic amounts initially and reduces the total stoichiometric requirement to 1-3 equivalents, drastically lowering the bill of materials.
Q: What are the purity specifications achievable with this manufacturing process?
A: The process is designed to achieve high purity levels suitable for veterinary pharmaceutical standards. Experimental embodiments demonstrate that the final Cefquinome Sulfate product can reach purity levels greater than 98% as determined by HPLC area normalization, with the intermediate 7-ACQ achieving purity over 97.4%.
Q: Does this method eliminate the need for imported ion-exchange resins?
A: Yes, a key advantage of this synthesis route is the elimination of the anion-exchange resin step (such as Amberlite LA-2) required in previous methods for salt formation. By directly forming the sulfate salt through acidification and crystallization, the process removes dependency on specific imported resin supplies, enhancing supply chain stability.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cefquinome Sulfate Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes for high-value veterinary antibiotics like Cefquinome Sulfate. Our technical team has extensively analyzed the process described in CN103193799A and possesses the expertise to implement this low-cost, high-yield methodology on an industrial scale. We have extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to pilot plant is seamless. Our facility is equipped with rigorous QC labs capable of verifying stringent purity specifications, ensuring that every batch of Cefquinome Sulfate meets the >98% purity benchmark required for regulatory compliance in major global markets.
We invite procurement leaders and R&D directors to collaborate with us to optimize their supply chains for veterinary drug intermediates. By leveraging our technical capabilities, you can achieve significant cost reductions and secure a stable supply of this critical antibiotic. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our manufacturing excellence can support your business growth.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
