Scalable Production of High-Purity L-(+)-Selenomethionine via Novel Alkylation Route
Scalable Production of High-Purity L-(+)-Selenomethionine via Novel Alkylation Route
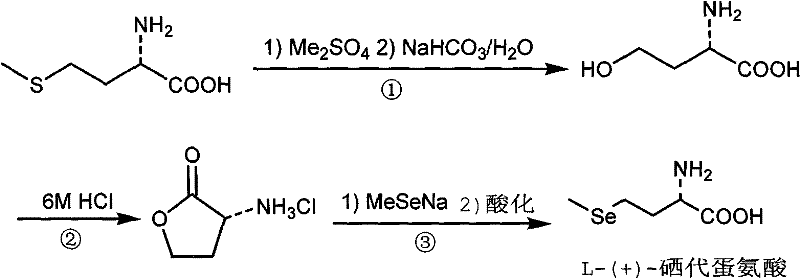
The global demand for high-bioavailability organic selenium compounds has surged, driven by their critical role in antioxidant defense systems and cancer prevention protocols. Patent CN102321004A introduces a transformative synthetic methodology for producing L-(+)-selenomethionine, a premier nutritional and pharmaceutical intermediate. This technology leverages L-(+)-methionine as a chiral pool starting material, utilizing dimethyl sulfate for efficient alkylation followed by a strategic lactonization and selenation sequence. For R&D directors and procurement specialists, this patent represents a pivotal shift away from hazardous cryogenic processes toward a more manageable, ambient-pressure workflow. The methodology not only addresses the chronic instability issues associated with inorganic selenium sources but also provides a robust framework for achieving consistent optical purity, which is paramount for regulatory compliance in the nutraceutical and pharmaceutical sectors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of selenomethionine has been plagued by severe safety hazards and economic inefficiencies that hinder large-scale adoption. Traditional routes, such as the liquid ammonia method, necessitate cryogenic conditions and the handling of pyrophoric metallic sodium, creating unacceptable risks for standard chemical manufacturing facilities. Furthermore, alternative pathways relying on sodium cyanide introduce extreme toxicity concerns, requiring specialized waste treatment infrastructure that drastically inflates operational expenditures. Previous iterations using methyl iodide, while chemically viable, suffer from the high volatility and cost of the alkylating agent, often leading to deep-colored, unstable products that require extensive and yield-eroding purification steps. These legacy methods collectively fail to meet the modern standards of green chemistry, presenting significant bottlenecks for supply chain heads seeking reliable, continuous production capabilities without the burden of complex environmental remediation.
The Novel Approach
The methodology outlined in CN102321004A circumvents these historical impediments by employing dimethyl sulfate as a superior alkylating reagent, which is both economically accessible and operationally safer than its volatile counterparts. This innovative route initiates with the conversion of L-methionine to a 4-hydroxy-alpha-amino acid intermediate, effectively swapping the sulfur moiety for a hydroxyl group under mild aqueous conditions. The subsequent cyclization to alpha-aminobutyrolactone hydrochloride serves as a crucial purification checkpoint, locking the stereochemistry before the final introduction of the selenium atom. By utilizing sodium methylselenide for the ring-opening step, the process achieves high regioselectivity and minimizes side reactions. This streamlined three-step sequence eliminates the need for anhydrous, oxygen-free environments, thereby reducing capital expenditure on specialized reactor hardware and enabling a more straightforward transition from laboratory bench to commercial kilogram scale.
Mechanistic Insights into Dimethyl Sulfate Alkylation and Lactonization
The core chemical innovation lies in the precise control of nucleophilic substitution and intramolecular cyclization kinetics. In the initial step, dimethyl sulfate acts as a potent methylating agent, reacting with the thioether sulfur of L-methionine to form a sulfonium salt intermediate. Subsequent hydrolysis in the presence of sodium bicarbonate facilitates the displacement of the dimethyl sulfide group by a hydroxyl nucleophile, yielding the 4-hydroxy-alpha-amino acid. This transformation is critical because it converts a relatively inert thioether into a reactive alcohol functionality without compromising the chiral center at the alpha-position. The reaction conditions are meticulously balanced to prevent over-alkylation or degradation of the amino acid backbone, ensuring that the intermediate retains the necessary structural integrity for the subsequent cyclization phase.
Following the formation of the hydroxy-acid, the introduction of 6M hydrochloric acid drives an intramolecular esterification, or lactonization, to form the five-membered alpha-aminobutyrolactone ring. This acid-catalyzed cyclization is thermodynamically favorable and serves to protect the amine functionality as a hydrochloride salt, which enhances the stability of the intermediate during isolation. The final step involves the nucleophilic attack of the methylselenide anion (MeSe-) on the gamma-carbon of the lactone ring. This ring-opening reaction is highly specific, regenerating the linear amino acid structure with the selenium atom precisely positioned to mimic the natural methionine architecture. The final acidification to pH 5.5-6.5 is a masterstroke of process chemistry, targeting the isoelectric point of the zwitterionic product to maximize precipitation yield while minimizing the co-crystallization of inorganic salts.
How to Synthesize L-(+)-Selenomethionine Efficiently
Implementing this synthesis requires strict adherence to the stoichiometric ratios and thermal profiles defined in the patent examples to ensure optimal yield and purity. The process begins with the alkylation of L-methionine in a water-methanol solvent system, where temperature control is vital to manage the exothermic nature of the sulfonium salt formation. Following hydrolysis, the crude hydroxy-acid is not isolated but directly subjected to acidic reflux, a telescoping strategy that reduces solvent usage and processing time. The resulting lactone hydrochloride is purified via recrystallization, providing a high-quality substrate for the final selenation. This modular approach allows for quality control checkpoints at the lactone stage, ensuring that any impurities from the initial alkylation do not carry through to the final high-value product.
- React L-(+)-methionine with dimethyl sulfate in water/methanol, followed by hydrolysis with NaHCO3 to form 4-hydroxy-alpha-amino acid.
- Cyclize the 4-hydroxy-alpha-amino acid using 6M hydrochloric acid under reflux to generate alpha-aminobutyrolactone hydrochloride.
- Perform ring-opening addition with sodium methylselenide in DMF/ethanol, followed by acidification to pH 5.5-6.5 to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the economic implications of this patent are profound, primarily driven by the substitution of exotic reagents with commodity chemicals. The reliance on L-methionine, a fermentation-derived amino acid produced in massive global volumes, guarantees a stable and price-resilient supply of the starting material. Unlike methods requiring custom-synthesized bromo-esters or cryogenic liquid ammonia, this route utilizes dimethyl sulfate and hydrochloric acid, which are standard bulk chemicals available from multiple tier-one suppliers worldwide. This diversification of the supply base significantly mitigates the risk of raw material shortages and allows for aggressive cost negotiation, directly impacting the bottom line of the final API or supplement production.
- Cost Reduction in Manufacturing: The elimination of expensive and hazardous reagents such as methyl lithium, tributoxyboron, or excess methyl iodide results in substantial raw material cost savings. Furthermore, the avoidance of specialized equipment for handling pyrophoric metals or maintaining anhydrous, oxygen-free atmospheres drastically reduces capital depreciation and maintenance costs. The simplified workup procedures, which rely on standard distillation and crystallization rather than complex chromatographic separations, lower the consumption of solvents and energy per kilogram of product. These cumulative efficiencies translate into a significantly lower cost of goods sold (COGS), providing a competitive pricing advantage in the global marketplace for selenium supplements.
- Enhanced Supply Chain Reliability: By removing dependencies on reagents with long lead times or restrictive shipping regulations, such as liquid bromine or sodium cyanide, the manufacturing timeline becomes far more predictable. The use of aqueous and alcoholic solvent systems simplifies logistics, as these materials do not require the same level of hazardous material handling infrastructure as ether or liquid ammonia. This operational flexibility allows manufacturers to respond more rapidly to fluctuations in market demand, ensuring consistent delivery schedules for downstream pharmaceutical clients. The robustness of the chemical route means that production campaigns can be extended without the frequent downtime associated with cleaning specialized reactors or treating highly toxic waste streams.
- Scalability and Environmental Compliance: The process generates waste streams that are significantly easier to treat compared to those containing heavy metals or cyanide residues. The primary byproducts involve sulfate salts and dimethyl sulfide, which can be managed through standard industrial wastewater treatment protocols. This alignment with environmental, health, and safety (EHS) standards facilitates smoother regulatory approvals and reduces the liability associated with hazardous waste disposal. The inherent scalability of the batch reactions, demonstrated by the successful transition from gram to multi-gram scales in the patent examples, indicates a low technical risk for scaling up to multi-ton annual production capacities, securing long-term supply continuity.
Frequently Asked Questions (FAQ)
The following technical inquiries address common concerns regarding the implementation and optimization of this synthesis route. These answers are derived directly from the experimental data and comparative analysis provided in the patent documentation, offering clarity on yield expectations, safety protocols, and purity profiles. Understanding these nuances is essential for process engineers evaluating the feasibility of technology transfer and for quality assurance teams establishing specification limits for the final active ingredient.
Q: Why is dimethyl sulfate preferred over methyl iodide in this synthesis?
A: Dimethyl sulfate is significantly less volatile and more cost-effective than methyl iodide. It eliminates the need for hazardous oxidation steps to remove excess alkylating agent, resulting in a cleaner product profile and reduced operational risk.
Q: How does this method ensure the retention of optical purity?
A: The synthesis starts from chiral L-(+)-methionine. The reaction conditions, particularly the mild hydrolysis and controlled acidification steps, are designed to prevent racemization at the alpha-carbon, ensuring the final product remains in the biologically active L-form.
Q: What are the scalability advantages of this route compared to fermentation?
A: Unlike fermentation which suffers from low conversion rates and complex downstream purification of inorganic selenium waste, this chemical route offers defined stoichiometry, faster reaction times, and simpler crystallization-based purification, making it highly suitable for multi-ton commercial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-(+)-Selenomethionine Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic value of adopting advanced synthetic routes like the one described in CN102321004A to secure a competitive edge in the fine chemical market. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from pilot plant to full-scale manufacturing is seamless and efficient. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of L-(+)-selenomethionine meets the highest international pharmacopoeial standards. Our commitment to process excellence means we can deliver high-purity intermediates with consistent optical rotation and minimal impurity profiles, supporting your regulatory filings and product launches.
We invite you to collaborate with us to leverage this cost-effective and safe synthesis technology for your supply chain needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our manufacturing capabilities can enhance your product quality while optimizing your overall procurement budget. Let us be your partner in delivering high-performance selenium solutions to the global market.
