Advanced Synthetic Route for L-(+)-Selenomethionine: Technical Breakthroughs and Commercial Scalability
Advanced Synthetic Route for L-(+)-Selenomethionine: Technical Breakthroughs and Commercial Scalability
The global demand for high-bioavailability organic selenium compounds has surged, driven by stringent regulations in animal nutrition and growing interest in oncology prevention therapies. Patent CN102321004A introduces a transformative synthetic methodology for producing L-(+)-selenomethionine, a critical nutritional ingredient and pharmaceutical intermediate. Unlike legacy processes that rely on hazardous reagents or complex fermentation purification, this invention leverages a concise three-step chemical transformation starting from abundant L-(+)-methionine. The core innovation lies in the strategic use of dimethyl sulfate for selective alkylation and subsequent hydrolysis, bypassing the severe safety and environmental bottlenecks associated with liquid ammonia or cyanide-based chemistries. For R&D directors and procurement strategists, this patent represents a pivotal shift towards safer, more cost-effective manufacturing paradigms. By replacing exotic reagents with commodity chemicals, the process not only enhances operational safety but also significantly stabilizes the supply chain against raw material volatility. This report provides a deep technical dissection of the mechanism, a comparative analysis against conventional methods, and a strategic evaluation of its commercial viability for reliable nutritional ingredients suppliers seeking to optimize their portfolio.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of selenomethionine has been plagued by severe technical and safety hurdles that hinder scalable production. Early methodologies, such as the liquid ammonia method, necessitate cryogenic reaction conditions and the use of metallic sodium, creating substantial explosion risks and requiring expensive, specialized pressure vessels that drive up capital expenditure. Alternative routes involving methylselenopropionaldehyde simulate outdated methionine production but introduce highly toxic sodium cyanide, complicating waste treatment and imposing rigorous environmental compliance costs on manufacturers. Furthermore, aminobutyrolactone methods starting from gamma-butyrolactone often yield racemic mixtures requiring difficult chiral separations, while utilizing liquid bromine near its boiling point poses acute corrosion and operator safety challenges. Other approaches employing methyllithium demand strictly anhydrous and oxygen-free environments, making them prohibitively expensive for bulk chemical manufacturing due to the high cost of reagents and the complexity of maintaining inert atmospheres on a multi-ton scale. These cumulative limitations result in low overall yields, unstable product quality, and significant environmental liabilities, rendering many traditional routes economically unviable for modern high-volume production facilities.
The Novel Approach
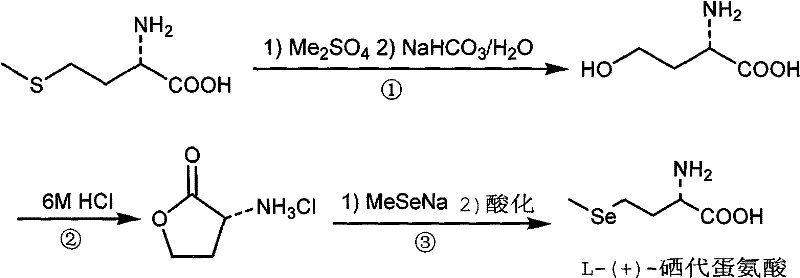
The methodology disclosed in CN102321004A fundamentally reengineers the synthetic pathway by utilizing dimethyl sulfate as a温和 yet effective alkylating agent to convert L-(+)-methionine into a key hydroxy-intermediate. This approach elegantly sidesteps the need for cryogenic conditions or pyrophoric reagents, operating instead in aqueous or alcoholic media that are compatible with standard stainless steel reactor systems. The process flow is streamlined into three distinct stages: initial methylation and hydrolysis to form 4-hydroxy-alpha-amino acid, acid-catalyzed cyclization to generate the lactone intermediate, and finally, nucleophilic ring-opening with sodium methylselenide. This sequence ensures that the chiral integrity of the starting L-methionine is preserved throughout the transformation, eliminating the need for costly resolution steps associated with racemic synthesis. Moreover, the use of dimethyl sulfate, a widely available commodity chemical, drastically reduces raw material costs compared to specialized organolithium or alkyl halide reagents. The reaction conditions are sufficiently mild to allow for straightforward workup procedures, primarily involving distillation and recrystallization, which simplifies downstream processing and enhances the overall throughput of the manufacturing line.
Mechanistic Insights into Dimethyl Sulfate Alkylation and Lactonization
The chemical elegance of this synthesis lies in the precise manipulation of functional groups to install the selenium moiety while maintaining stereochemical fidelity. The first stage involves the reaction of the sulfur atom in L-(+)-methionine with dimethyl sulfate, forming a sulfonium salt intermediate which is subsequently hydrolyzed under basic conditions using sodium bicarbonate. This hydrolysis step is critical as it cleaves the methylthio group to reveal the primary hydroxyl functionality, yielding 4-hydroxy-alpha-amino acid without racemizing the alpha-carbon center. Following isolation, the hydroxy-acid undergoes an intramolecular esterification catalyzed by concentrated hydrochloric acid. This cyclization is driven by the formation of a stable five-membered lactone ring, specifically alpha-aminobutyrolactone hydrochloride, which serves as a highly activated electrophile for the subsequent substitution. The protonation of the hydroxyl group by HCl facilitates water elimination, generating a reactive cyclic oxonium species that is primed for nucleophilic attack. This mechanistic pathway ensures that the amino group remains protected as a hydrochloride salt, preventing unwanted side reactions and maintaining the structural integrity of the backbone throughout the harsh acidic conditions.
The final and most crucial step involves the nucleophilic displacement of the lactone oxygen by the methylselenide anion. Sodium methylselenide, generated in situ or added as a pre-formed solution in ethanol, acts as a potent soft nucleophile that attacks the gamma-carbon of the lactone ring. This ring-opening reaction is thermodynamically favorable due to the relief of ring strain inherent in the four or five-membered cyclic ester structure. The reaction proceeds through a tetrahedral intermediate which collapses to reform the carbonyl group, effectively swapping the oxygen linker for a selenium linker. Careful control of the pH during the final acidification step, typically adjusting to a range of 5.5 to 6.5 using acetic acid, is essential to precipitate the zwitterionic L-(+)-selenomethionine product while keeping inorganic salts in solution. This pH-dependent crystallization mechanism is a key factor in achieving high purity, as it exploits the isoelectric point of the amino acid to separate it from residual selenium species and reaction byproducts, thereby minimizing the need for complex chromatographic purification and ensuring a robust impurity profile suitable for pharmaceutical or nutritional applications.
How to Synthesize L-(+)-Selenomethionine Efficiently
Implementing this synthetic route requires strict adherence to the stoichiometric ratios and thermal profiles outlined in the patent examples to maximize yield and purity. The process begins with the alkylation of L-(+)-methionine using a slight molar excess of dimethyl sulfate, typically in a water-methanol solvent system, followed by a controlled hydrolysis phase. Operators must monitor the distillation of solvents carefully to concentrate the intermediate before introducing the acid for cyclization. The subsequent ring-opening with sodium methylselenide demands anhydrous conditions for the reagent preparation but can be conducted in polar aprotic or protic solvents like DMF or ethanol under reflux. Detailed standard operating procedures regarding temperature ramping, addition rates, and crystallization kinetics are essential for technology transfer.
- React L-(+)-methionine with dimethyl sulfate followed by hydrolysis with sodium bicarbonate to generate 4-hydroxy-alpha-amino acid.
- Perform acid-catalyzed cyclization using 6M hydrochloric acid under reflux to form alpha-aminobutyrolactone hydrochloride.
- Execute nucleophilic ring-opening using sodium methylselenide in DMF/ethanol, followed by acidification to pH 5.5-6.5 to isolate the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this dimethyl sulfate-based methodology offers profound strategic advantages beyond mere technical feasibility. The primary value driver is the substantial reduction in raw material costs achieved by substituting expensive, specialty reagents like methyllithium or methyl iodide with dimethyl sulfate, which is produced on a massive industrial scale for other applications. This shift not only lowers the direct bill of materials but also mitigates supply risk, as dimethyl sulfate is less prone to the market volatility that affects niche organometallic compounds. Furthermore, the elimination of hazardous reagents such as liquid ammonia or cyanide simplifies regulatory compliance and reduces the overhead costs associated with hazardous waste disposal and specialized storage infrastructure. The process inherently supports a leaner manufacturing model by reducing the number of unit operations and avoiding complex purification steps like chiral chromatography, which translates directly into lower conversion costs and higher asset utilization rates for production facilities.
- Cost Reduction in Manufacturing: The economic argument for this process is compelling, primarily driven by the replacement of high-cost reagents with commodity chemicals. By utilizing dimethyl sulfate instead of volatile alkyl halides or air-sensitive organometallics, manufacturers can achieve significant savings in raw material procurement without compromising reaction efficiency. Additionally, the simplified workup procedure, which relies on distillation and recrystallization rather than energy-intensive drying or complex extraction sequences, reduces utility consumption and labor hours per batch. The avoidance of expensive catalysts or ligands further contributes to a lower cost of goods sold, enabling competitive pricing strategies in the global nutritional ingredients market while maintaining healthy margins.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the reliance on widely available starting materials like L-(+)-methionine and dimethyl sulfate, both of which have established, multi-vendor global supply networks. This diversification reduces dependency on single-source suppliers for exotic reagents, thereby minimizing the risk of production stoppages due to raw material shortages. The robustness of the reaction conditions also means that the process is less sensitive to minor variations in reagent quality, allowing for greater flexibility in sourcing. Consequently, manufacturers can maintain consistent production schedules and shorter lead times, ensuring reliable delivery to downstream customers in the pharmaceutical and animal feed sectors even during periods of market disruption.
- Scalability and Environmental Compliance: From an environmental and scaling perspective, this route is exceptionally well-suited for large-volume production. The absence of heavy metal catalysts or persistent organic pollutants simplifies wastewater treatment and aligns with increasingly stringent green chemistry regulations. The process generates fewer hazardous byproducts, reducing the burden on EHS departments and lowering the costs associated with environmental remediation. Moreover, the mild reaction temperatures and pressures allow for the use of standard glass-lined or stainless steel reactors, facilitating seamless scale-up from pilot batches to commercial tonnage without the need for bespoke engineering solutions. This scalability ensures that supply can be rapidly expanded to meet surging market demand for organic selenium supplements.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. They are derived from a critical analysis of the patent's background and experimental data, focusing on the practical implications for industrial adoption. Understanding these nuances is vital for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: Why is the dimethyl sulfate route superior to traditional liquid ammonia methods for selenomethionine?
A: Traditional liquid ammonia methods require cryogenic temperatures and hazardous metallic sodium, posing significant safety risks and equipment costs. The dimethyl sulfate route operates under mild conditions using readily available commodity chemicals, eliminating the need for specialized anhydrous or cryogenic infrastructure while simplifying downstream purification.
Q: How does this synthetic method address impurity control compared to bromine-based routes?
A: Bromine-based routes often introduce halogenated impurities and require handling toxic liquid bromine near its boiling point. This novel method utilizes a clean hydrolysis and lactonization sequence that avoids halogenation entirely, resulting in a cleaner crude profile that facilitates easier recrystallization and higher final purity without extensive chromatographic separation.
Q: Is this process suitable for large-scale industrial production of nutritional ingredients?
A: Yes, the process is designed for scalability. It utilizes standard reactor materials compatible with aqueous and alcoholic solvents, avoids pyrophoric reagents like methyllithium, and employs simple distillation and recrystallization unit operations. This makes it highly adaptable for commercial scale-up from pilot plants to multi-ton annual production capacities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable L-(+)-Selenomethionine Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthetic technologies to meet the evolving demands of the global nutraceutical and pharmaceutical industries. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of patent CN102321004A are fully realized in practical manufacturing settings. We are committed to delivering high-purity L-(+)-selenomethionine that meets stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle complex amino acid derivatives with the utmost care, ensuring batch-to-batch consistency and regulatory compliance for food and pharma applications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this methodology. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your volume needs, ensuring a seamless transition to a more efficient and reliable source of high-quality organic selenium ingredients.
