Revolutionizing TUDCA Production: A Silane-Catalyzed Route for Commercial Scale-Up
Revolutionizing TUDCA Production: A Silane-Catalyzed Route for Commercial Scale-Up
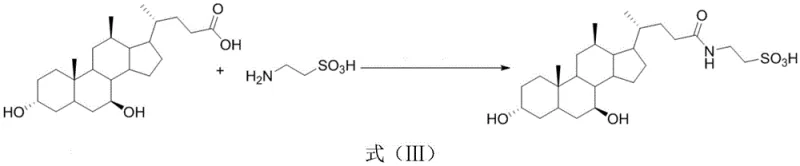
The pharmaceutical industry constantly seeks robust, scalable, and economically viable pathways for synthesizing high-value active pharmaceutical ingredients (APIs) and their precursors. A significant breakthrough in this domain is documented in patent CN112588319A, which discloses a novel application of silane compounds for the synthesis of tauroursodeoxycholic acid (TUDCA). TUDCA, a conjugated bile acid, is increasingly recognized for its potent hepatoprotective, anti-apoptotic, and choleretic properties, making it a critical component in treating cholestatic liver diseases. The disclosed technology leverages a unique dual-catalyst system comprising hydrosilane and aminosilane to facilitate the direct amidation of ursodeoxycholic acid (UDCA) with taurine. This innovation represents a paradigm shift from traditional peptide coupling strategies, offering a streamlined one-pot process that operates under remarkably mild conditions. For global procurement and R&D teams, this patent signals a new standard for efficiency in bile acid derivative manufacturing, promising enhanced purity profiles and reduced operational complexity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the chemical synthesis of tauroursodeoxycholic acid has been plagued by significant technical and economic hurdles that hinder large-scale industrial adoption. Traditional methodologies generally fall into three categories: the mixed acid anhydride-phenol ester method, the condensing agent method, and the activated thioester method. The mixed anhydride approach often necessitates the use of highly toxic solvents such as dioxane, posing severe safety risks and requiring extensive waste management protocols that drive up operational costs. Furthermore, this route typically involves multiple synthetic steps, leading to cumulative yield losses and a complex impurity profile that complicates downstream purification. Alternatively, the condensing agent method, while direct, relies on expensive reagents like 4-(4,6-dimethoxy-1,3,5-triazin-2-yl)-4-methylmorpholinium chloride (DMTMM). The high cost of these coupling agents makes the final API prohibitively expensive for many applications. Finally, the activated thioester method, though chemically elegant, involves difficult-to-handle intermediates and oxidation steps that are notoriously challenging to control on a multi-ton scale, rendering it unsuitable for cost-effective commercial production.
The Novel Approach
In stark contrast to these legacy techniques, the silane-catalyzed synthesis described in the patent offers a transformative solution that addresses the core inefficiencies of prior art. By employing a synergistic combination of hydrosilane and aminosilane, the process enables the direct formation of the amide bond between the carboxyl group of ursodeoxycholic acid and the amino group of taurine in a single reaction vessel. This one-pot strategy eliminates the need for isolating unstable activated intermediates, thereby drastically simplifying the workflow. The reaction proceeds efficiently in tetrahydrofuran (THF), a solvent that has been empirically determined to provide superior yields compared to alternatives like dichloromethane or acetonitrile. Crucially, the process operates at ambient temperatures ranging from 20°C to 30°C, removing the need for energy-intensive heating or cryogenic cooling. This mild thermal profile not only reduces utility costs but also minimizes the risk of thermal degradation of the sensitive bile acid scaffold, ensuring a cleaner reaction output.
Mechanistic Insights into Silane-Catalyzed Amidation
The efficacy of this novel synthetic route lies in the unique mechanistic role played by the silane catalysts, which function through a distinct activation pathway compared to traditional carbodiimides or uronium salts. The hydrosilane component acts as a potent activating agent for the carboxylic acid moiety of ursodeoxycholic acid. Upon interaction, the hydrosilane reacts in situ with the carboxyl group to generate a highly reactive silicon-based carboxylic ester intermediate. This silyl ester is significantly more electrophilic than the parent acid, facilitating nucleophilic attack. Concurrently, the aminosilane additive plays a critical catalytic role by stabilizing the transition state and promoting the conversion of the silicon-based ester. This dual-catalyst system ensures that the activation energy barrier for amide bond formation is sufficiently lowered, allowing the reaction to proceed rapidly even at room temperature. The presence of the aminosilane prevents the hydrolysis of the activated intermediate and directs the reaction trajectory exclusively towards the desired amide product, thereby suppressing side reactions that typically lead to impurities.
From an impurity control perspective, this mechanism offers substantial advantages for meeting stringent regulatory standards required for a reliable pharmaceutical intermediates supplier. Traditional coupling reagents often leave behind urea byproducts or require extensive washing to remove phosphine oxides, which can be difficult to purge completely. In the silane-catalyzed system, the byproducts are primarily siloxanes or silyl ethers, which are generally more volatile or easily separable during the aqueous workup. The patent data highlights that maintaining the molar ratio of hydrosilane to ursodeoxycholic acid at greater than or equal to 1:1 is critical for driving the reaction to completion. Furthermore, the specific selection of tetrahydrofuran as the solvent creates an optimal solvation environment that keeps the reactants in solution while allowing the final product to precipitate upon acidification. This inherent difference in solubility between the reactants and the product facilitates a straightforward isolation process, resulting in a crude product that is already of high purity before the final recrystallization step.
How to Synthesize Tauroursodeoxycholic Acid Efficiently
Implementing this synthesis requires precise control over stoichiometry and reaction parameters to maximize yield and purity. The process begins with the dissolution of ursodeoxycholic acid in tetrahydrofuran, followed by the sequential addition of the silane catalysts and the taurine nucleophile. The reaction mixture is then maintained under controlled agitation to ensure homogeneity and efficient mass transfer. Following the reaction period, a specific workup protocol involving pH adjustment and solvent extraction is employed to isolate the target molecule. The detailed standardized operating procedures for this synthesis, including exact mixing times and filtration parameters, are outlined in the technical guide below.
- Dissolve ursodeoxycholic acid in tetrahydrofuran and add hydrosilane and aminosilane catalysts under stirring.
- Add taurine to the reaction mixture and maintain temperature between 20-30°C for 6-10 hours to form the crude product.
- Purify the crude product through extraction, acidification to pH 1-3, and recrystallization using an acetone-water mixture.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this silane-catalyzed technology translates into tangible strategic benefits that extend beyond simple yield improvements. The elimination of expensive coupling reagents like DMTMM directly impacts the bill of materials, offering a pathway for significant cost reduction in API manufacturing without compromising quality. Furthermore, the simplified one-pot nature of the reaction reduces the number of unit operations required, which in turn lowers labor costs and decreases the overall cycle time for production batches. This efficiency gain allows manufacturers to respond more agilely to market demand fluctuations, ensuring a more stable supply of this critical hepatoprotective agent.
- Cost Reduction in Manufacturing: The primary driver for cost optimization in this process is the substitution of high-value condensing agents with catalytic amounts of silanes. Traditional methods consume stoichiometric quantities of expensive activators, whereas this method utilizes hydrosilane and aminosilane in catalytic or near-stoichiometric ratios that are far more economical. Additionally, the avoidance of toxic solvents like dioxane reduces the costs associated with hazardous waste disposal and environmental compliance. The mild reaction conditions also imply lower energy consumption for heating or cooling, further contributing to a leaner manufacturing cost structure that enhances competitiveness in the global marketplace.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable raw materials. Unlike specialized peptide coupling reagents that may have long lead times or limited suppliers, hydrosilanes and aminosilanes are commodity chemicals with robust global supply networks. This availability reduces the risk of production stoppages due to raw material shortages. Moreover, the robustness of the reaction conditions—specifically the tolerance for a broad temperature range of 20°C to 30°C—means that the process is less susceptible to minor fluctuations in plant utilities, ensuring consistent batch-to-batch quality and reliable delivery schedules for downstream customers.
- Scalability and Environmental Compliance: Scaling chemical processes often introduces new challenges regarding heat dissafety and mixing, but this method is inherently designed for scalability. The exotherm of the reaction is manageable under the described conditions, and the use of THF allows for efficient heat transfer. From an environmental standpoint, the process aligns with green chemistry principles by avoiding heavy metals and toxic reagents. The high atom economy and the ability to recycle solvents contribute to a reduced environmental footprint, helping manufacturers meet increasingly strict regulatory requirements for sustainable pharmaceutical production and facilitating smoother regulatory approvals in key markets.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and optimization of this silane-catalyzed synthesis route. These insights are derived directly from the experimental data and comparative examples provided in the patent literature, offering practical guidance for process engineers and quality assurance teams looking to adopt this technology.
Q: What are the advantages of the silane-catalyzed method over traditional condensing agents?
A: Unlike traditional methods using expensive condensing agents like DMTMM or toxic reagents like dioxane, the silane-catalyzed method utilizes mild conditions (20-30°C) and avoids heavy metal contamination, significantly reducing purification costs and environmental impact.
Q: What is the optimal solvent system for this synthesis?
A: Experimental data indicates that tetrahydrofuran (THF) is the superior solvent, achieving yields up to 85%, whereas solvents like dichloromethane, acetonitrile, or methanol result in significantly lower yields below 55%.
Q: How is high purity achieved in the final TUDCA product?
A: High purity is ensured through a specific workup procedure involving ethyl acetate extraction, acidification of the aqueous phase to pH 1-3 to precipitate the crude solid, followed by recrystallization in an acetone-water mixture (8:3 ratio).
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tauroursodeoxycholic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the silane-catalyzed synthesis route for producing high-purity tauroursodeoxycholic acid. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative patent technology into commercial reality. Our facilities are equipped to handle complex organic syntheses with precision, ensuring that every batch meets the rigorous quality standards demanded by the pharmaceutical industry. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, allowing us to offer flexible manufacturing solutions that grow with your needs. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize advanced analytical techniques to verify the identity and potency of every intermediate and API we produce.
We invite global partners to collaborate with us to leverage this cost-effective and environmentally friendly synthesis method. Whether you require custom synthesis services or bulk supply of TUDCA intermediates, our team is ready to support your project goals. Please contact our technical procurement team to request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to reach out for specific COA data and route feasibility assessments to understand how our optimized silane-catalyzed process can enhance your supply chain efficiency and product profitability.
