Advanced Synthetic Route for Sitagliptin Intermediates: Cost-Effective Industrial Scale-Up
Advanced Synthetic Route for Sitagliptin Intermediates: Cost-Effective Industrial Scale-Up
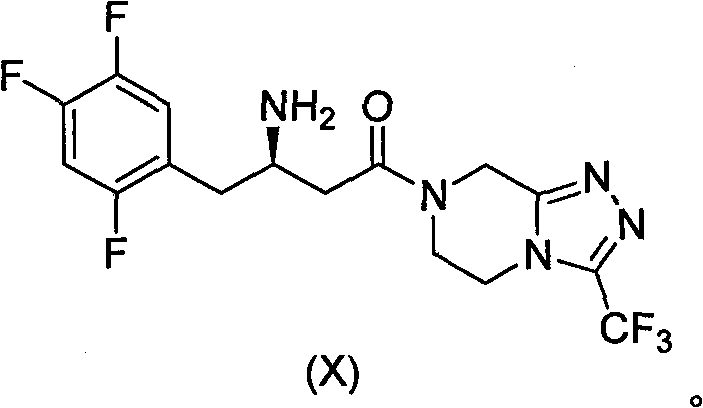
The global demand for antidiabetic medications continues to surge, placing immense pressure on the supply chains of key Active Pharmaceutical Ingredients (APIs) like Sitagliptin. Patent CN102757431A discloses a groundbreaking methodology for synthesizing Sitagliptin intermediates that fundamentally alters the economic landscape of production. Unlike traditional pathways that rely on scarce chiral catalysts, this novel approach leverages classical chiral resolution techniques combined with robust ester protection strategies. By shifting away from precious metal catalysis, this technology offers a viable pathway for cost reduction in API manufacturing while maintaining the rigorous purity standards required by regulatory bodies. The following analysis details how this innovation addresses critical bottlenecks in the current production of DPP-4 inhibitors.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Sitagliptin has been dominated by routes established by the originator, which often involve complex asymmetric catalytic hydrogenation or chiral pool synthesis. As illustrated in prior art such as WO2003004498, early methods utilized chiral induction sources that were difficult to acquire and expensive to implement on a metric ton scale. Furthermore, subsequent improvements described in patents like WO2009064476 relied heavily on sophisticated transition metal catalysts, specifically Rhodium or Ruthenium complexes paired with chiral ligands like BINAP. These catalytic systems not only introduce significant raw material costs but also necessitate stringent removal processes to meet residual metal limits in the final drug substance. Additionally, some legacy routes require hazardous reagents such as n-Butyl Lithium and cryogenic temperatures as low as -78°C, creating substantial safety risks and energy burdens that hinder commercial scale-up of complex pharmaceutical intermediates.
The Novel Approach
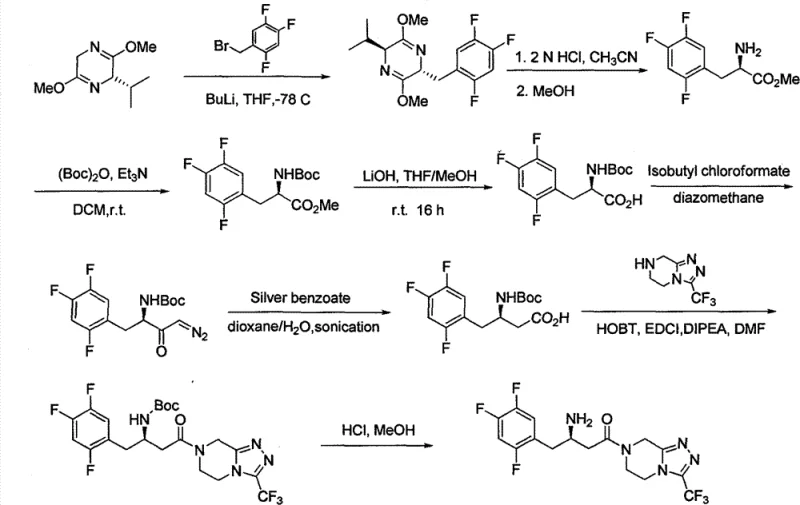
In stark contrast, the methodology presented in CN102757431A introduces a streamlined synthetic sequence that bypasses the need for asymmetric hydrogenation entirely. The core innovation lies in the construction of a racemic intermediate followed by a highly efficient chiral resolution step. The process begins with the condensation of a trifluorophenyl ketone derivative with benzyl alcohol to form a stable beta-keto ester, followed by enamine formation and reduction. This route operates under mild thermal conditions ranging from 0°C to 110°C, utilizing common organic solvents like toluene and acetonitrile. By decoupling the stereochemistry establishment from the carbon-carbon bond formation, the process allows for the use of inexpensive, achiral starting materials. This strategic pivot transforms the manufacturing profile, offering a reliable sitagliptin intermediate supplier alternative that is both economically superior and operationally simpler than catalytic asymmetric routes.
Mechanistic Insights into Chiral Resolution and Ester Protection
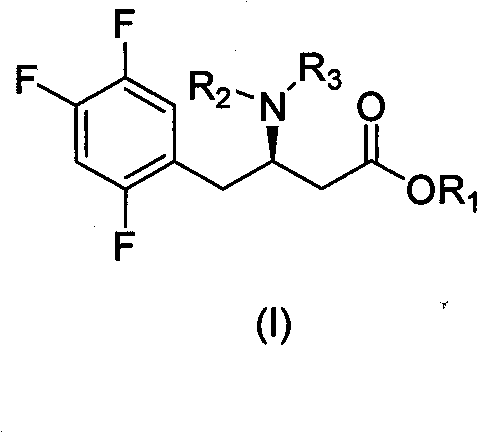
The heart of this synthetic strategy is the meticulous control of stereochemistry through diastereomeric salt formation rather than kinetic resolution via catalysis. In the critical resolution step, the racemic amine intermediate is treated with D-dibenzoyl tartaric acid in a mixed solvent system of acetonitrile and water. The mechanism relies on the differential solubility of the resulting diastereomeric salts; the desired (R)-enantiomer salt precipitates selectively upon cooling, while the unwanted (S)-enantiomer remains in the mother liquor. This physical separation is driven by thermodynamic stability and crystal lattice energy differences, allowing for the isolation of the chiral amine with an enantiomeric excess (ee) value reaching 99.5% after recrystallization. This approach effectively circumvents the sensitivity of transition metal catalysts to poisons and the variability often seen in asymmetric hydrogenation batches.
Furthermore, the use of the benzyl ester protecting group plays a pivotal role in impurity control and process robustness. The benzyl group provides sufficient stability during the enamine formation and reduction steps, preventing side reactions such as self-condensation or hydrolysis that could plague free acid intermediates. Crucially, the benzyl group can be cleanly removed via catalytic hydrogenolysis in a later stage using standard Pd-C catalysts under mild pressure, regenerating the free acid without affecting the sensitive trifluorophenyl moiety or the newly established chiral center. This orthogonal protection strategy ensures that the impurity profile remains clean throughout the synthesis, facilitating easier downstream purification and ensuring the final API meets the strict specifications for high-purity pharmaceutical intermediates.
How to Synthesize Sitagliptin Intermediate Efficiently
The synthesis of the key chiral intermediate involves a logical sequence of protection, functionalization, and resolution. The process initiates with the esterification of the keto-acid precursor, followed by conversion to an enamine using ammonium sources. Subsequent reduction yields the racemic amine, which is then subjected to chiral resolution. The detailed operational parameters, including specific molar ratios, solvent choices, and temperature profiles for each transformation, are critical for maximizing yield and optical purity. For process chemists looking to implement this technology, understanding the nuances of the crystallization step is paramount to success. The standardized synthesis steps for this pathway are outlined below.
- Condensation of the trifluorophenyl ketone precursor with benzyl alcohol to form the protected beta-keto ester intermediate.
- Formation of the enamine derivative using ammonium acetate or amine sources, followed by reduction to the racemic amine.
- Chiral resolution of the racemic amine using D-dibenzoyl tartaric acid to isolate the desired (R)-enantiomer with high optical purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route translates directly into enhanced margin protection and supply security. The elimination of proprietary chiral catalysts removes a significant single-point-of-failure risk often associated with specialized reagent sourcing. Moreover, the reliance on commodity chemicals such as benzyl alcohol, ammonium acetate, and tartaric acid derivatives ensures that raw material availability is not constrained by niche supplier capacity. This shift towards bulk chemical inputs drastically simplifies the procurement landscape and mitigates the volatility associated with precious metal markets.
- Cost Reduction in Manufacturing: The most immediate financial impact stems from the complete removal of expensive Rhodium or Ruthenium catalysts and chiral ligands like BINAP from the process. These transition metal complexes represent a substantial portion of the variable cost in traditional routes. By replacing them with stoichiometric amounts of inexpensive resolving agents like D-dibenzoyl tartaric acid, the direct material cost is significantly reduced. Additionally, the avoidance of cryogenic operations eliminates the need for specialized low-temperature reactors and the high energy consumption associated with maintaining minus seventy-eight degrees Celsius, leading to substantial operational expenditure savings.
- Enhanced Supply Chain Reliability: The robustness of this chemistry enhances supply continuity by reducing batch failure rates. Traditional asymmetric hydrogenation can be sensitive to trace impurities in solvents or substrates, leading to inconsistent conversion and extended cycle times. The resolution-based approach described here is far more forgiving and reproducible, ensuring consistent lead times for reducing lead time for high-purity pharmaceutical intermediates. The use of stable intermediates also allows for potential campaign manufacturing, where key precursors can be stockpiled without degradation, providing a buffer against demand fluctuations.
- Scalability and Environmental Compliance: From an EHS (Environment, Health, and Safety) perspective, this route offers distinct advantages for large-scale production. The absence of pyrophoric reagents like n-Butyl Lithium removes a major safety hazard, simplifying facility requirements and insurance costs. Furthermore, the solvent systems employed (toluene, acetonitrile, ethyl acetate) are well-understood and easily recoverable through standard distillation protocols, minimizing waste generation. This aligns with modern green chemistry principles, facilitating smoother regulatory approvals and reducing the environmental footprint of the manufacturing process.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this novel synthetic pathway. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on the feasibility and advantages of the technology for potential partners and licensees.
Q: How does this new method reduce production costs compared to traditional routes?
A: This method eliminates the need for expensive transition metal catalysts like Rhodium or Ruthenium complexes and avoids cryogenic conditions (-78°C), significantly lowering raw material and energy costs.
Q: What is the optical purity achievable with this chiral resolution process?
A: Through recrystallization of the D-dibenzoyl tartaric acid salt, the process achieves an ee value of up to 99.5%, meeting stringent pharmaceutical specifications.
Q: Is this synthetic route suitable for large-scale commercial manufacturing?
A: Yes, the route utilizes mild reaction conditions (0-110°C), common solvents like toluene and acetonitrile, and avoids hazardous reagents like n-Butyl Lithium, making it highly scalable and safe for industrial production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Sitagliptin Intermediate Supplier
The technological advancements detailed in patent CN102757431A represent a significant opportunity for optimizing the production of Sitagliptin intermediates. At NINGBO INNO PHARMCHEM, we possess the technical expertise to translate these laboratory-scale innovations into robust, commercial-grade manufacturing processes. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this route are fully realized in practice. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of intermediate meets the exacting standards required for global pharmaceutical registration.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage this cost-effective synthesis. By partnering with our technical procurement team, you can access a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our optimized manufacturing capabilities can strengthen your supply chain for this critical antidiabetic medication.
