Revolutionizing Cyclopropane Synthesis: Continuous Copper Catalysis for Commercial Scale-Up
Revolutionizing Cyclopropane Synthesis: Continuous Copper Catalysis for Commercial Scale-Up
The pharmaceutical and agrochemical industries are constantly seeking robust, scalable, and cost-effective methods for constructing strained ring systems, particularly cyclopropanes, which are ubiquitous motifs in bioactive molecules. Patent CN102216243A introduces a transformative approach to the preparation of cyclopropane derivatives by leveraging copper metal or copper oxide catalysts within a continuous flow regime. This technology represents a paradigm shift from traditional batch processes that rely on expensive homogeneous noble metal catalysts. By integrating the catalyst directly into the reactor infrastructure or utilizing packed beds, this method not only simplifies the purification workflow but also significantly enhances process safety when handling energetic diazo precursors. For R&D directors and process chemists, this patent offers a compelling route to high-purity intermediates with a markedly reduced environmental footprint.
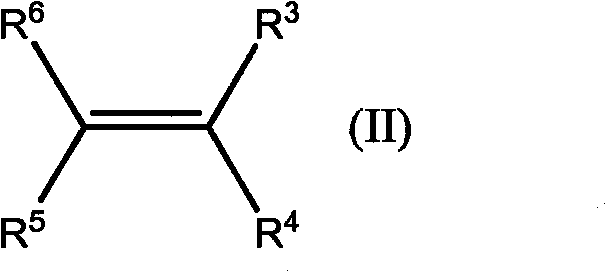
The core innovation lies in the substitution of costly rhodium or palladium complexes with elemental copper or copper oxides. In conventional synthesis, the removal of residual noble metals from the final API intermediate is a critical and often expensive bottleneck, requiring specialized scavengers or chromatography. The heterogeneous nature of the copper catalyst described in this patent allows the reaction to proceed without contaminating the product stream with soluble metal species, thereby streamlining the downstream processing. This is particularly advantageous for the commercial scale-up of complex pharmaceutical intermediates where metal limits are strictly regulated. The ability to run this transformation continuously further amplifies these benefits by ensuring consistent product quality and reducing the variability inherent in batch-to-batch operations.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional cyclopropanation methodologies predominantly utilize homogeneous catalysts based on rhodium, such as Rh2(OAc)4, or palladium complexes. While effective on a small laboratory scale, these systems present substantial challenges when translated to industrial manufacturing. The primary drawback is the high cost of the noble metals themselves, which directly impacts the cost of goods sold (COGS) for the final active ingredient. Moreover, because these catalysts are dissolved in the reaction medium, they necessitate rigorous purification steps to meet stringent regulatory limits on heavy metal residues. These separation processes often involve multiple extraction cycles, filtration through silica or specialized resins, and extensive solvent usage, all of which contribute to higher operational costs and increased waste generation. Additionally, the batch-wise addition of diazo compounds, which are thermally unstable and potentially explosive, poses significant safety risks that require elaborate engineering controls and limit the feasible reaction concentrations.
The Novel Approach
The novel approach detailed in the patent circumvents these issues by employing a continuous flow process catalyzed by copper metal. This method allows for the precise control of reaction parameters such as temperature, pressure, and residence time, which is critical for managing the exothermic decomposition of diazo compounds into carbenes. By utilizing a microreactor or a tubular reactor lined with or packed with copper, the system achieves high surface-area-to-volume ratios that facilitate rapid heat dissipation. This thermal management capability enables the safe use of higher concentrations of reactants, thereby improving space-time yields. Furthermore, the heterogeneous catalyst remains fixed within the reactor, meaning the effluent contains only the product, unreacted starting materials, and solvent. This simplifies the workup to essentially a distillation or crystallization step, eliminating the need for metal scavenging and drastically reducing the complexity of the manufacturing process.
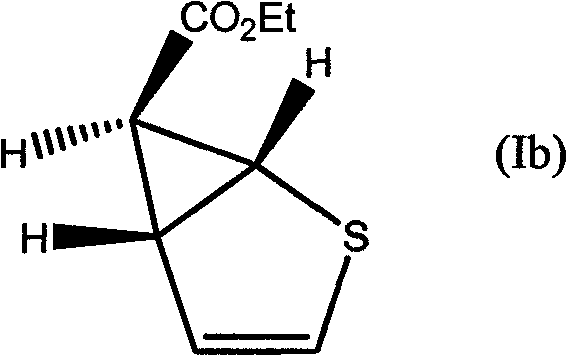
Mechanistic Insights into Copper-Catalyzed Cyclopropanation
The mechanistic pathway involves the in situ generation of a reactive carbene species from a diazo precursor, such as ethyl diazoacetate, upon contact with the copper surface. The copper acts as a Lewis acid, facilitating the extrusion of nitrogen gas to form a copper-carbenoid intermediate. This electrophilic species then undergoes a concerted [2+1] cycloaddition with the electron-rich double bond of the olefin substrate. The patent highlights that the stereochemical outcome of this reaction can be influenced by the specific reaction conditions and the nature of the olefin. For instance, when reacting ethyl diazoacetate with thiophene, the process yields specific stereoisomers, with the trans-isomer often being predominant depending on the thermal profile and residence time within the reactor. Understanding these stereoelectronic interactions is crucial for R&D teams aiming to optimize the diastereomeric ratio for specific biological targets without resorting to difficult chiral separations later in the synthesis.
Impurity control is another critical aspect addressed by the continuous flow architecture. In batch reactors, local hot spots can lead to the dimerization of the carbene species, forming unwanted fumarate or maleate byproducts. The efficient mixing and heat transfer characteristics of the microreactor system minimize these thermal gradients, thereby suppressing dimerization pathways. The patent data indicates that by fine-tuning the residence time and temperature, the formation of dimers can be kept to a minimum while maximizing the conversion of the diazo compound. This level of control ensures a cleaner crude product profile, which reduces the burden on purification units and improves the overall yield of the desired cyclopropane derivative. The ability to recycle unreacted olefins and solvents further enhances the atom economy of the process, aligning with green chemistry principles.
How to Synthesize Cyclopropane Derivatives Efficiently
The synthesis of these valuable intermediates is achieved through a streamlined continuous operation that integrates reagent mixing, reaction, and thermal control into a single unified system. The process begins with the preparation of the diazo compound, which can also be generated continuously to avoid the storage of hazardous materials. This stream is then merged with the olefin feed and pumped through the copper-catalyzed reactor zone. The detailed standardized synthesis steps for implementing this technology are provided in the guide below.
- Prepare the diazo compound precursor, such as ethyl diazoacetate, potentially using a continuous diazo transfer process in a microreactor to ensure safety.
- Mix the olefin substrate (e.g., thiophene or styrene) with the diazo compound solution, optionally using the olefin itself as the solvent to minimize waste.
- Pass the reaction mixture through a copper metal microreactor or a reactor packed with copper particles at elevated temperatures (100-200°C) to facilitate cyclopropanation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this copper-catalyzed continuous process offers tangible strategic advantages beyond mere technical feasibility. The transition from noble metal catalysts to base metal copper represents a fundamental shift in cost structure, removing the volatility associated with rhodium and palladium pricing. This stability allows for more accurate long-term budgeting and reduces the risk of supply chain disruptions caused by geopolitical factors affecting precious metal availability. Additionally, the continuous nature of the process facilitates a smaller physical footprint for manufacturing equipment compared to large batch vessels, enabling production capacity to be scaled through numbering-up rather than building larger facilities. This modularity enhances supply chain resilience and allows for faster response times to market demand fluctuations.
- Cost Reduction in Manufacturing: The elimination of expensive rhodium catalysts results in substantial raw material savings. Since the copper catalyst is heterogeneous and integrated into the reactor, there is no loss of catalyst in the product stream, removing the need for costly recovery or disposal procedures. The process also operates efficiently at higher concentrations, reducing the volume of solvents required per kilogram of product. This reduction in solvent usage lowers both procurement costs for chemicals and the expenses associated with solvent recovery and waste treatment. Consequently, the overall cost of production for these cyclopropane intermediates is significantly optimized, providing a competitive edge in pricing for the final pharmaceutical products.
- Enhanced Supply Chain Reliability: Continuous manufacturing inherently provides a more consistent output rate compared to batch processing, which is subject to turnaround times between batches. The robustness of the copper catalyst, which can be used for extended periods without significant deactivation, ensures uninterrupted production runs. This reliability is critical for maintaining inventory levels of key intermediates and preventing stockouts that could halt downstream API synthesis. Furthermore, the simplified purification train reduces the number of unit operations, decreasing the probability of mechanical failures or bottlenecks. This streamlined workflow translates to shorter lead times and a more dependable supply of high-purity cyclopropane derivatives for global partners.
- Scalability and Environmental Compliance: The microreactor technology described allows for linear scalability; increasing production capacity simply involves running the system for longer durations or paralleling multiple reactor units. This avoids the non-linear engineering challenges often encountered when scaling up batch exothermic reactions. From an environmental perspective, the process generates less waste due to higher selectivity and solvent recycling capabilities. The reduced energy consumption for heating and cooling, owing to the high efficiency of heat exchange in microchannels, contributes to a lower carbon footprint. These factors collectively support compliance with increasingly strict environmental regulations and corporate sustainability goals, making the supply chain more future-proof.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this continuous copper-catalyzed technology. These insights are derived directly from the experimental data and embodiments disclosed in the patent documentation, providing a clear understanding of the process capabilities and limitations for potential adopters.
Q: Why is copper preferred over rhodium for industrial cyclopropanation?
A: Copper metal or copper oxide catalysts are significantly less expensive than rhodium or palladium complexes. Furthermore, heterogeneous copper catalysts can be integrated into the reactor wall or packing, eliminating the need for complex downstream separation steps required for homogeneous noble metal catalysts.
Q: How does this process handle the safety risks of diazo compounds?
A: The process utilizes continuous flow microreactors which offer superior heat transfer and mass transfer capabilities. This minimizes the hold-up volume of hazardous diazo intermediates and allows for precise temperature control, drastically reducing the risk of thermal runaway or explosion associated with batch processing of energetic materials.
Q: Can the solvent be recycled in this continuous method?
A: Yes, the patent describes methods where the olefin acts as the solvent or where solvents like dichloroethane are separated from the product stream via distillation and recycled back into the feed, enhancing the overall atom economy and reducing environmental impact.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Cyclopropane Derivatives Supplier
The technological advancements outlined in patent CN102216243A underscore the immense potential of continuous flow chemistry in modernizing the production of complex organic intermediates. NINGBO INNO PHARMCHEM stands at the forefront of this innovation, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is backed by stringent purity specifications and rigorous QC labs that ensure every batch meets the highest international standards. We understand that transitioning to new manufacturing technologies requires a partner who can navigate both the chemical complexities and the regulatory landscape with equal proficiency.
We invite you to collaborate with us to leverage these cost-effective and safe synthetic routes for your specific project needs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your volume requirements. Please contact us to request specific COA data and route feasibility assessments for your target cyclopropane intermediates. Together, we can optimize your supply chain and accelerate the delivery of life-saving medicines to the market.
