Advanced Palladium-Catalyzed Synthesis of Alpha-Substituted Alpha-Amino Acid Esters for Commercial Scale-up
Introduction to Next-Generation Amino Acid Ester Synthesis
The landscape of pharmaceutical intermediate manufacturing is constantly evolving, driven by the need for more efficient and sustainable synthetic routes. Patent CN112920066A introduces a groundbreaking methodology for the preparation of alpha-substituted-alpha-amino acid ester compounds, a structural motif ubiquitous in bioactive molecules and drug candidates. This technology leverages a palladium-catalyzed tandem amination/oxidation process that directly couples readily available aromatic amines with alkenyl ethers. Unlike traditional multi-step sequences, this approach operates under mild conditions, often utilizing air as the oxidant source or benign peroxides, significantly reducing the environmental footprint. The versatility of this method allows for the construction of diverse molecular architectures, addressing the long-standing challenge of synthesizing multi-substituted and multi-functionalized complex amine compounds with high precision.
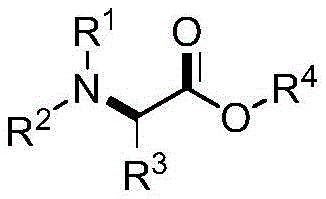
For R&D directors and process chemists, the ability to access these scaffolds without extensive pre-functionalization represents a significant strategic advantage. The core innovation lies in the direct activation of the olefinic bond in the presence of an amine, facilitated by a specialized catalytic system. This not only streamlines the supply chain by reducing the number of discrete unit operations but also enhances the overall atom economy of the process. As the demand for high-purity pharmaceutical intermediates grows, adopting such robust and flexible synthetic platforms becomes critical for maintaining competitiveness in the global market. The following analysis details the mechanistic underpinnings and commercial implications of this novel technology.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of alpha-substituted-alpha-amino acid esters has relied on three primary strategies, each fraught with significant operational and economic drawbacks. The first common strategy involves the esterification of pre-synthesized alpha-substituted alpha-amino acids, which often requires harsh acidic or basic conditions that can compromise sensitive functional groups. The second strategy employs nucleophilic substitution at the alpha-position of an ester, typically necessitating the introduction of leaving groups like halogens, which adds extra synthetic steps and generates stoichiometric salt waste. The third approach utilizes electrophilic substitution on the alpha-carbon of existing amino acid esters, a method that frequently demands rigorous protection and deprotection sequences for the amine group to prevent side reactions. These conventional pathways are not only labor-intensive but also suffer from poor atom economy and limited substrate scope, particularly when dealing with electron-rich or sterically hindered substrates.
The Novel Approach
In stark contrast, the methodology disclosed in CN112920066A circumvents these bottlenecks by employing a direct oxidative coupling strategy. By utilizing simple alkenyl ethers and aromatic amines as starting materials, the process eliminates the need for pre-functionalized precursors or protecting groups. The reaction proceeds via a palladium-catalyzed mechanism that simultaneously forms the carbon-nitrogen and carbon-oxygen bonds required for the ester functionality in a single pot. This tandem transformation is conducted under remarkably mild conditions, typically at temperatures ranging from 20°C to 35°C, and can even proceed in an air atmosphere, removing the need for expensive inert gas setups. The use of ionic liquids as solvents further distinguishes this approach, offering enhanced stability for the catalytic species and facilitating easier product isolation, thereby presenting a superior alternative for cost reduction in pharmaceutical intermediate manufacturing.
Mechanistic Insights into Palladium-Catalyzed Tandem Amination/Oxidation
The success of this synthetic route hinges on a sophisticated catalytic cycle involving palladium species, specialized ligands, and oxidants. The reaction initiates with the coordination of the palladium catalyst, such as palladium acetate or palladium chloride, to the electron-rich double bond of the alkenyl ether. In the presence of nitrogen-based ligands like 5,5-dimethyl-2,2-bipyridine or 4,5-diazafluoren-9-one, the metal center is stabilized, preventing premature precipitation and maintaining high catalytic turnover. The aromatic amine then attacks the activated olefin, forming a key organopalladium intermediate. Subsequent oxidation, mediated by agents like benzoquinone, hydrogen peroxide, or iodobenzene diacetate, regenerates the active palladium species and facilitates the formation of the ester linkage. This intricate dance of electron transfer and bond formation allows for the construction of the complex alpha-amino acid ester skeleton with high regioselectivity.

Furthermore, the choice of solvent plays a pivotal role in the reaction's efficiency and impurity profile. The patent highlights the use of imidazole-type ionic liquids, such as 1-butyl-3-methylimidazolium tetrafluoroborate ([Bmim]BF4). These ionic environments are believed to stabilize charged transition states and improve the solubility of polar intermediates, leading to smoother reaction kinetics. From an impurity control perspective, the mild nature of the reaction minimizes the formation of degradation products often seen in high-temperature processes. The high functional group tolerance means that sensitive moieties like halogens (chlorine, bromine) and alkenes remain intact, reducing the complexity of the crude mixture and simplifying downstream purification. This mechanistic robustness ensures that the process is not just a laboratory curiosity but a viable candidate for reliable pharmaceutical intermediate supplier operations.
How to Synthesize Alpha-Substituted-Alpha-Amino Acid Esters Efficiently
Implementing this synthesis requires careful attention to reagent ratios and reaction parameters to maximize yield and purity. The general protocol involves dissolving the aromatic amine and alkenyl ether in the chosen solvent, followed by the addition of the catalyst, ligand, and oxidant. The reaction is typically stirred at room temperature for 8 to 24 hours, allowing sufficient time for the tandem transformation to reach completion. Detailed standard operating procedures, including specific molar ratios and workup techniques, are essential for reproducibility and scale-up success.
- Dissolve aromatic amine and alkenyl ether in an ionic liquid solvent such as 1-butyl-3-methylimidazolium tetrafluoroborate under an air atmosphere.
- Add palladium catalyst (e.g., palladium acetate), a bipyridine ligand, and an oxidant like hydrogen peroxide or benzoquinone to the reaction mixture.
- Stir the reaction at room temperature (20-35°C) for 8-24 hours, then extract with ethyl acetate and purify via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this technology offers tangible benefits in terms of cost structure and operational reliability. The primary driver for cost reduction is the utilization of commodity chemicals as starting materials. Aromatic amines and alkenyl ethers are widely available in the bulk chemical market, avoiding the premium pricing associated with specialized, pre-functionalized building blocks. Additionally, the elimination of protection/deprotection steps significantly reduces the consumption of auxiliary reagents and solvents, directly lowering the variable cost per kilogram of the final product. The ability to run the reaction under air atmosphere further reduces utility costs by removing the dependency on nitrogen or argon blanketing systems, which is a significant factor in large-scale manufacturing.
- Cost Reduction in Manufacturing: The streamlined nature of this one-pot synthesis drastically simplifies the production workflow. By combining amination and oxidation into a single step, the process reduces the number of reactors required and shortens the overall cycle time. This consolidation of unit operations leads to substantial savings in labor, energy, and equipment depreciation. Furthermore, the high atom economy ensures that a greater proportion of the raw material mass is incorporated into the final product, minimizing waste disposal costs. The use of recyclable ionic liquids, where applicable, adds another layer of economic efficiency by reducing solvent purchase and treatment expenses over the long term.
- Enhanced Supply Chain Reliability: Sourcing risk is mitigated by the reliance on generic, high-volume feedstocks rather than niche intermediates with limited supplier bases. The robustness of the reaction conditions, specifically the tolerance for ambient temperature and air, means that the process is less susceptible to disruptions caused by utility failures or strict environmental controls. This resilience ensures a more consistent supply of high-purity pharmaceutical intermediates, crucial for maintaining uninterrupted production schedules for downstream API manufacturers. The broad substrate scope also allows for flexibility in sourcing; if one specific aniline derivative is unavailable, structurally similar alternatives can often be substituted with minimal process re-optimization.
- Scalability and Environmental Compliance: Scaling this process from gram to ton quantities is facilitated by the absence of hazardous reagents and extreme conditions. The mild temperatures (20-35°C) reduce the thermal load on cooling systems, making it easier to manage exotherms in large reactors. From an environmental standpoint, the reduced generation of salt waste and the potential for solvent recycling align with increasingly stringent global regulations on chemical manufacturing emissions. This compliance readiness future-proofs the supply chain against regulatory tightening, ensuring long-term viability and reducing the risk of production shutdowns due to environmental non-compliance.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and scope of this palladium-catalyzed synthesis. Understanding these nuances is vital for process engineers evaluating the technology for integration into existing manufacturing lines. The answers are derived directly from the experimental data and scope defined in the patent literature, providing a realistic view of the method's capabilities and limitations in a commercial context.
Q: What are the advantages of using ionic liquids in this palladium-catalyzed reaction?
A: Ionic liquids, such as imidazole-type salts, enhance homogeneous catalytic efficiency by stabilizing charged intermediates and improving the solubility of polar reactants, leading to higher yields and easier product separation compared to traditional organic solvents.
Q: Does this method tolerate sensitive functional groups like halogens?
A: Yes, the method exhibits excellent functional group tolerance, successfully preserving halogen substituents (chlorine, bromine) and alkene groups on the substrate without requiring protective group strategies, which simplifies the synthetic route.
Q: What types of oxidants are compatible with this synthesis?
A: The process is versatile regarding oxidants, accommodating benign options like hydrogen peroxide as well as organic oxidants such as benzoquinone and iodobenzene diacetate, allowing for optimization based on cost and safety requirements.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Substituted-Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic methodologies like the one described in CN112920066A. As a leading CDMO partner, we possess the technical expertise to translate such innovative laboratory protocols into robust, commercial-scale processes. Our facilities are equipped to handle complex synthetic pathways, with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We understand that consistency is key in the pharmaceutical industry, which is why our rigorous QC labs and stringent purity specifications ensure that every batch of alpha-substituted-alpha-amino acid esters meets the highest quality standards required for drug substance manufacturing.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our team is ready to provide a Customized Cost-Saving Analysis tailored to your specific molecule, demonstrating how this route can optimize your bill of materials. Please contact our technical procurement team today to request specific COA data and route feasibility assessments. Let us help you secure a competitive advantage through superior chemistry and reliable supply chain execution.
