Advanced Palladium-Catalyzed Synthesis of Alpha-Substituted Alpha-Amino Acid Esters for Commercial Scale-up
Introduction to Next-Generation Amino Acid Ester Synthesis
The pharmaceutical and fine chemical industries are constantly seeking more efficient pathways to construct complex molecular architectures, particularly those serving as critical building blocks for active pharmaceutical ingredients (APIs). Patent CN112920066A introduces a groundbreaking methodology for the preparation of alpha-substituted-alpha-amino acid ester compounds, a class of molecules pivotal in medicinal chemistry due to their versatile reactivity and biological potential. This innovation addresses the long-standing challenges associated with the precise synthesis of multi-substituted and multi-functionalized amine compounds, which have historically required cumbersome multi-step sequences. By leveraging a palladium-catalyzed electron-rich olefin tandem amination/oxidation process, this technology enables the direct transformation of simple aromatic amines and alkenyl ethers into valuable ester derivatives. 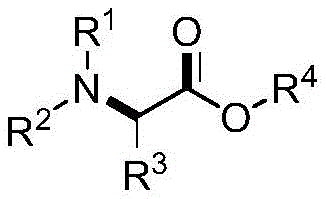 The significance of this development lies in its ability to bypass traditional limitations such as the need for pre-functionalized starting materials or harsh reaction environments. For R&D directors and process chemists, this represents a paradigm shift towards more convergent and atom-economical synthetic strategies. The structural diversity achievable through this method, as defined by the variable R groups in the general formula, allows for the rapid generation of libraries for drug discovery while maintaining a pathway that is inherently scalable for commercial manufacturing. This report analyzes the technical merits and commercial implications of adopting this novel synthesis route for high-purity pharmaceutical intermediates.
The significance of this development lies in its ability to bypass traditional limitations such as the need for pre-functionalized starting materials or harsh reaction environments. For R&D directors and process chemists, this represents a paradigm shift towards more convergent and atom-economical synthetic strategies. The structural diversity achievable through this method, as defined by the variable R groups in the general formula, allows for the rapid generation of libraries for drug discovery while maintaining a pathway that is inherently scalable for commercial manufacturing. This report analyzes the technical merits and commercial implications of adopting this novel synthesis route for high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the construction of alpha-substituted-alpha-amino acid esters has relied on three primary strategies, each fraught with significant operational and economic drawbacks that hinder large-scale production. The first common strategy involves the direct esterification of alpha-substituted-alpha-amino acids, which often necessitates the prior synthesis of the amino acid itself through resolution or asymmetric synthesis, adding substantial cost and time to the supply chain. The second strategy employs nucleophilic substitution with amines at the alpha-position of an ester group; however, this frequently requires the introduction of leaving groups like halogens, generating stoichiometric salt waste and requiring rigorous purification to remove metal residues. The third approach utilizes electrophilic substitution on the alpha-carbon of existing amino acid esters, a method that typically demands harsh reaction conditions and strong bases, leading to poor functional group tolerance and potential racemization of chiral centers. Furthermore, many of these conventional routes require the protection and subsequent deprotection of amine groups, adding at least two additional synthetic steps that drastically reduce overall yield and increase the environmental footprint of the manufacturing process.
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN112920066A offers a streamlined, one-pot solution that fundamentally simplifies the synthetic landscape. By utilizing readily available aromatic amines and alkenyl ethers as direct feedstocks, this palladium-catalyzed protocol eliminates the need for pre-functionalization of the amine substrate. The reaction proceeds under remarkably mild conditions, often at room temperature (20-35°C) and under a simple air atmosphere, removing the capital expenditure associated with inert gas manifolds and cryogenic cooling systems. This novel approach not only enhances step atom economy by combining amination and oxidation into a single transformative event but also demonstrates exceptional substrate universality. The ability to tolerate diverse functional groups—including halogens, ethers, and even terminal alkenes—without the need for protecting groups means that complex, high-value intermediates can be accessed directly from commodity chemicals. This reduction in synthetic complexity translates directly into a more robust and cost-effective supply chain for downstream API manufacturing.
Mechanistic Insights into Palladium-Catalyzed Tandem Amination/Oxidation
The core of this technological breakthrough lies in the sophisticated interplay between the palladium catalyst, the ligand system, and the oxidant within a specialized solvent environment. The reaction mechanism initiates with the coordination of the palladium species to the electron-rich double bond of the alkenyl ether, activating it towards nucleophilic attack by the aromatic amine. Unlike traditional cross-couplings that might require strict anaerobic conditions, this system uniquely leverages molecular oxygen from the air, facilitated by the oxidant (such as benzoquinone or hydrogen peroxide), to regenerate the active palladium species and drive the oxidative functionalization forward. The use of ionic liquids, specifically imidazole-based salts like 1-butyl-3-methylimidazolium tetrafluoroborate ([Bmim]BF4), plays a critical role in stabilizing the charged intermediates and enhancing the homogeneous catalytic efficiency.  This ionic environment creates a unique solvation shell that likely lowers the activation energy for the C-H functionalization step, allowing the reaction to proceed smoothly at ambient temperatures. For the technical team, understanding this mechanism is vital for troubleshooting and optimization; the choice of ligand, such as 5,5-dimethyl-2,2-bipyridine, is crucial for modulating the electronic properties of the palladium center to prevent catalyst deactivation. The result is a highly selective transformation that installs the ester functionality and the amine moiety simultaneously with high regiocontrol, avoiding the formation of unwanted regioisomers that often plague radical-based approaches.
This ionic environment creates a unique solvation shell that likely lowers the activation energy for the C-H functionalization step, allowing the reaction to proceed smoothly at ambient temperatures. For the technical team, understanding this mechanism is vital for troubleshooting and optimization; the choice of ligand, such as 5,5-dimethyl-2,2-bipyridine, is crucial for modulating the electronic properties of the palladium center to prevent catalyst deactivation. The result is a highly selective transformation that installs the ester functionality and the amine moiety simultaneously with high regiocontrol, avoiding the formation of unwanted regioisomers that often plague radical-based approaches.
Furthermore, the impurity profile generated by this method is significantly cleaner compared to nucleophilic substitution routes. Because the reaction does not rely on alkyl halides or strong bases, there is a marked reduction in the formation of elimination byproducts or quaternary ammonium salts. The high functional group tolerance implies that sensitive moieties present on the aromatic ring, such as bromine or trifluoromethyl groups, remain untouched, preserving the chemical integrity required for subsequent cross-coupling reactions in API synthesis. This selectivity is paramount for maintaining high purity specifications without the need for extensive recrystallization or preparative HPLC, which are costly bottlenecks in commercial production. The mechanistic robustness ensures that the process is reproducible across different batches, a key requirement for regulatory compliance in the pharmaceutical sector.
How to Synthesize Alpha-Substituted-Alpha-Amino Acid Esters Efficiently
Implementing this synthesis route in a laboratory or pilot plant setting requires careful attention to the specific ratios of catalyst, ligand, and oxidant as detailed in the patent examples. The process is designed to be operationally simple, involving the mixing of reagents in an ionic liquid or organic solvent followed by stirring under air. However, to achieve the optimal yields reported (up to 91%), precise control over the molar equivalents of the alkenyl ether and the oxidant is necessary. The following guide outlines the standardized procedure derived from the patent data to ensure successful replication of this high-efficiency transformation.
- Charge a reactor with aromatic amine, alkenyl ether, palladium catalyst (e.g., Pd(OAc)2), ligand (e.g., bipyridine derivative), oxidant, and ionic liquid solvent under air atmosphere.
- Stir the reaction mixture at mild temperatures (preferably 20-35°C) for 8 to 24 hours to allow the tandem amination and oxidation to proceed.
- Quench the reaction with water, extract the organic phase with ethyl acetate, dry over anhydrous sodium sulfate, and purify the crude product via column chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this palladium-catalyzed methodology offers tangible strategic advantages that extend beyond mere technical novelty. The primary driver for cost reduction lies in the sourcing of raw materials; aromatic amines and alkenyl ethers are commodity chemicals available in bulk quantities from multiple global suppliers, mitigating the risk of single-source dependency. By eliminating the need for protected amino acid starting materials, which are often expensive and subject to volatile pricing, manufacturers can significantly lower the Bill of Materials (BOM) cost. Additionally, the simplification of the workflow from a multi-step sequence to a one-pot reaction drastically reduces labor costs, solvent consumption, and waste disposal fees. The ability to run the reaction at room temperature further contributes to energy savings, as there is no requirement for heating or cooling infrastructure, making the process inherently greener and more sustainable.
- Cost Reduction in Manufacturing: The elimination of protection and deprotection steps is a major factor in driving down production costs. In traditional synthesis, these steps can account for a significant portion of the total processing time and material usage. By bypassing them, the overall yield is improved, and the consumption of auxiliary reagents is minimized. Furthermore, the use of ionic liquids, while initially more expensive than standard solvents, offers the potential for recycling and reuse, which can amortize the cost over multiple batches. The high atom economy of the tandem reaction ensures that a larger proportion of the starting mass ends up in the final product, reducing the cost per kilogram of the active intermediate.
- Enhanced Supply Chain Reliability: The robustness of this reaction conditions translates directly into supply chain resilience. Since the reaction tolerates air and moisture to a certain extent and uses stable, shelf-stable reagents, the risk of batch failure due to environmental factors is minimized. The broad substrate scope means that if a specific aromatic amine becomes unavailable, structurally similar analogs can often be substituted without re-optimizing the entire process, providing flexibility in raw material sourcing. This adaptability is crucial for maintaining continuous production schedules and meeting tight delivery deadlines for downstream customers.
- Scalability and Environmental Compliance: Scaling this process from gram to ton scale is facilitated by the mild reaction conditions and the absence of hazardous reagents like strong acids or pyrophoric bases. The use of air as the terminal oxidant is particularly advantageous from a safety and environmental perspective, as it avoids the storage and handling of dangerous oxidizing agents. The reduced generation of salt waste and organic byproducts simplifies wastewater treatment and aligns with increasingly stringent environmental regulations. This ease of scale-up ensures that the transition from R&D to commercial production is smooth, reducing the time-to-market for new pharmaceutical products.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These answers are derived directly from the experimental data and claims within patent CN112920066A, providing a reliable basis for decision-making. Understanding these nuances helps stakeholders evaluate the feasibility of integrating this method into their existing manufacturing portfolios.
Q: What are the primary advantages of this palladium-catalyzed method over traditional esterification?
A: Unlike traditional methods requiring pre-functionalized amino acids or harsh nucleophilic substitutions, this novel approach utilizes readily available aromatic amines and alkenyl ethers in a one-pot process. It eliminates the need for amine deprotection steps and operates under mild conditions with high atom economy.
Q: Does this synthesis method tolerate sensitive functional groups like halogens?
A: Yes, the method exhibits excellent functional group tolerance. As demonstrated in the patent data, substrates containing bromine, chlorine, and even unprotected alkene groups (such as allyl ethers) remain intact during the reaction, making it ideal for synthesizing complex pharmaceutical intermediates.
Q: What represent the optimal reaction conditions for maximum yield?
A: The patent indicates that using ionic liquids (such as [Bmim]BF4) as solvents, combined with palladium acetate and specific bipyridine ligands at room temperature (20-35°C) under an air atmosphere, provides the highest yields, reaching up to 91% in optimized examples.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Alpha-Substituted-Alpha-Amino Acid Ester Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of the palladium-catalyzed synthesis route described in CN112920066A for the production of high-value pharmaceutical intermediates. As a leading CDMO partner, we possess the technical expertise and infrastructure to translate this innovative laboratory methodology into a robust commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the benefits of this mild, one-pot process are fully realized at an industrial level. We are committed to delivering products with stringent purity specifications, supported by our rigorous QC labs that utilize advanced analytical techniques to verify the absence of residual palladium and other impurities.
We invite pharmaceutical companies and chemical manufacturers to collaborate with us to leverage this cost-effective synthesis strategy. By partnering with our technical procurement team, you can obtain a Customized Cost-Saving Analysis tailored to your specific target molecules. We encourage you to contact us to request specific COA data and route feasibility assessments, allowing you to validate the commercial viability of this advanced technology for your supply chain needs. Let us help you secure a reliable source of high-purity alpha-amino acid esters while optimizing your manufacturing costs.
