Revolutionizing Enantioselective Cyclization with Advanced Chiral Bisphosphine Ligands
Revolutionizing Enantioselective Cyclization with Advanced Chiral Bisphosphine Ligands
The landscape of asymmetric catalysis is undergoing a significant transformation driven by the need for more sustainable and cost-effective manufacturing processes for complex pharmaceutical intermediates. Patent CN109293700B introduces a groundbreaking class of chiral bisphosphine ligands, specifically designed to overcome the longstanding limitations of nickel-catalyzed reductive cyclization reactions. This technology addresses the critical challenge of constructing chiral tertiary alcohols, a structural motif prevalent in bioactive natural products and drug candidates, which has historically been difficult to access with high stereocontrol. The disclosed ligands, exemplified by the structure in Formula I, enable reactions to proceed with unprecedented efficiency, utilizing trace amounts of nickel catalyst while delivering superior enantiomeric ratios.
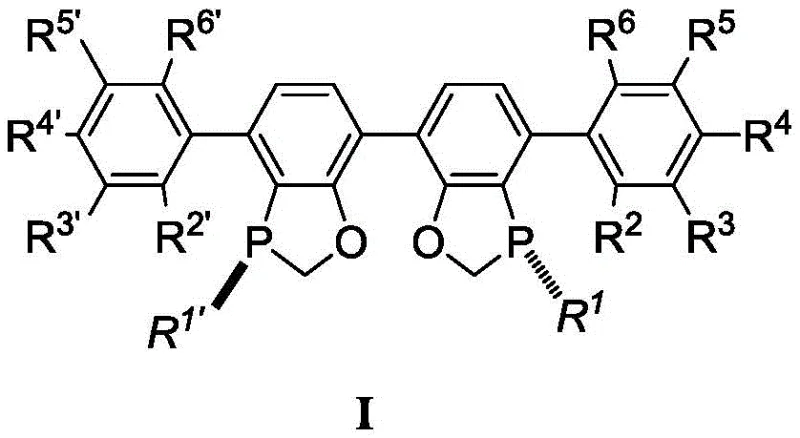
For R&D directors and process chemists, the implications of this innovation are profound. Traditional methods often suffer from low catalytic turnover, requiring high loadings of expensive transition metals that complicate downstream purification and increase the environmental footprint. In contrast, the ligands described in this patent facilitate a streamlined pathway to high-purity chiral pyrrole and piperidine derivatives. By fine-tuning the steric bulk and electronic properties through the diverse R groups on the biaryl backbone, manufacturers can achieve precise control over the reaction outcome. This level of customization ensures that the technology is not merely a laboratory curiosity but a robust platform adaptable to various substrate scopes required in modern drug discovery and development pipelines.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the enantioselective cyclization of N-alkynones has been plagued by inefficiencies that hinder commercial viability. Conventional nickel-catalyzed systems typically demand catalyst loadings ranging from 5 to 30 mol%, which is economically prohibitive for large-scale synthesis. High metal loading not only inflates raw material costs but also introduces significant challenges in removing residual nickel from the final API, a critical quality attribute regulated by strict international guidelines. Furthermore, existing ligand systems often struggle to induce sufficient chirality during the formation of tetra-substituted carbon centers, resulting in mediocre enantiomeric excess values that necessitate costly recrystallization or chromatographic separation steps. These inefficiencies create bottlenecks in the supply chain, extending lead times and reducing the overall throughput of manufacturing facilities.
The Novel Approach
The novel approach presented in this patent fundamentally shifts the paradigm by introducing a specialized bisphosphine ligand architecture that maximizes catalytic efficiency. As demonstrated in the experimental data, the new ligand system allows for nickel loadings as low as 0.05 mol% while achieving yields exceeding 98% and enantiomeric ratios of 99:1. This drastic reduction in catalyst requirement is achieved through the unique spatial arrangement of the phosphine atoms and the rigid biaryl scaffold, which creates a highly defined chiral pocket around the metal center. This design stabilizes the key organometallic intermediates and accelerates the rate-determining steps of the catalytic cycle. Consequently, manufacturers can realize substantial process intensification, reducing reaction times and simplifying workup procedures, thereby enhancing the overall sustainability and economic feasibility of producing complex chiral intermediates.
Mechanistic Insights into Nickel-Catalyzed Reductive Cyclization
The efficacy of this chiral bisphosphine ligand stems from its ability to modulate the electronic density and steric environment of the nickel center during the reductive cyclization process. The reaction mechanism involves the oxidative addition of the nickel(0) species to the substrate, followed by migratory insertion and subsequent reductive elimination to form the cyclic product. The bulky tert-butyl groups on the phosphorus atoms, combined with the substituted aryl rings, prevent the formation of inactive nickel aggregates and suppress competing side reactions such as linear oligomerization. This ensures that the catalytic cycle remains productive over thousands of turnovers. The ligand's rigidity locks the conformation of the transition state, effectively differentiating between the prochiral faces of the ketone substrate to deliver the desired enantiomer with high fidelity.
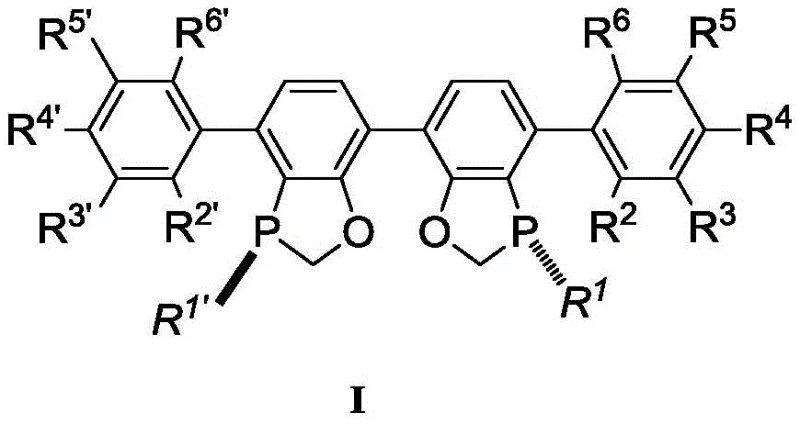
Impurity control is another critical aspect where this technology excels. In traditional high-loading scenarios, the accumulation of metal byproducts and ligand degradation products can contaminate the reaction mixture. However, the ultra-low loading enabled by this ligand minimizes the introduction of extraneous species. Furthermore, the high selectivity reduces the formation of diastereomers and regioisomers, simplifying the impurity profile. This clean reaction output is particularly advantageous for regulatory compliance, as it reduces the burden on analytical teams to identify and quantify trace impurities. The robust nature of the ligand under reaction conditions also means that it maintains its integrity throughout the process, preventing the release of free phosphine or phosphine oxides that could interfere with downstream processing or affect the stability of the final product.
How to Synthesize (S,S)-DI-BI-DIME Efficiently
The synthesis of the flagship ligand (S,S)-DI-BI-DIME (L4) outlined in the patent represents a masterclass in modern organic synthesis, balancing complexity with operational simplicity. The route begins with readily available phosphine oxide precursors and proceeds through a series of high-yielding transformations including silyl protection, bromination, and palladium-catalyzed cross-coupling. Each step is optimized to minimize waste and maximize atom economy, reflecting a design philosophy geared towards industrial scalability. The final reduction step converts the stable phosphine oxide into the active phosphine species using polymethylhydrosiloxane, a safe and effective reducing agent. This comprehensive synthetic strategy ensures that the ligand can be produced reliably in multi-kilogram quantities to support commercial manufacturing demands.
- Protection and Bromination: Start with the phosphine oxide precursor, protect the hydroxyl group with TBDPSCl, and perform regioselective bromination using NBS.
- Biaryl Coupling: Execute a palladium-catalyzed homocoupling or Suzuki coupling to form the biphenyl backbone, followed by deprotection to reveal the phenolic hydroxyls.
- Triflation and Final Reduction: Convert phenols to triflates, couple with boronic acids to install aryl groups, and finally reduce the phosphine oxides to phosphines using silane reagents.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this ligand technology translates into tangible strategic benefits that extend beyond mere technical performance. The most significant advantage lies in the drastic reduction of transition metal consumption. By lowering the nickel requirement from typical percentages to trace parts-per-million levels, companies can significantly lower their raw material costs and reduce dependency on volatile metal markets. This efficiency also alleviates the logistical burden associated with sourcing and handling large quantities of catalysts. Moreover, the simplified purification process resulting from lower metal residues means faster batch release times and reduced solvent consumption for chromatography, directly impacting the cost of goods sold and improving the overall margin profile of the manufactured intermediates.
- Cost Reduction in Manufacturing: The ultra-low catalyst loading eliminates the need for expensive metal scavengers and extensive purification protocols, leading to substantial operational savings. The high yield and selectivity further reduce waste disposal costs and improve material throughput, ensuring that every kilogram of starting material is converted into valuable product with minimal loss.
- Enhanced Supply Chain Reliability: The robust synthesis of the ligand itself relies on common chemical building blocks and standard reaction conditions, mitigating the risk of supply disruptions associated with exotic reagents. This stability ensures a consistent supply of the catalyst, allowing production planners to forecast timelines with greater accuracy and maintain continuous manufacturing operations without unexpected delays caused by reagent shortages.
- Scalability and Environmental Compliance: The process is inherently greener due to the reduced metal waste and solvent usage, aligning with increasingly stringent environmental regulations. The ability to scale this chemistry from gram to ton scale without losing efficiency provides a clear pathway for commercial expansion, allowing companies to meet growing market demand for chiral intermediates while maintaining a sustainable corporate footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this chiral ligand technology. These insights are derived directly from the patent data and are intended to assist decision-makers in evaluating the feasibility of integrating this solution into their existing workflows. Understanding the nuances of catalyst handling, substrate compatibility, and regulatory implications is essential for maximizing the value proposition of this innovation.
Q: What is the primary advantage of this chiral ligand over traditional phosphine ligands?
A: The primary advantage is the ability to operate at ultra-low catalyst loadings (as low as 0.05 mol%) while maintaining exceptional enantioselectivity (up to 99:1 er) and high yields, significantly reducing transition metal contamination and cost.
Q: Can this ligand be used for substrates other than N-alkynones?
A: While optimized for the enantioselective reductive cyclization of N-alkynones to form chiral tertiary alcohols, the steric and electronic properties of the ligand suggest potential applicability in other nickel-catalyzed asymmetric transformations requiring bulky, electron-rich phosphine environments.
Q: Is the synthesis of this ligand scalable for industrial production?
A: Yes, the synthetic route utilizes robust reactions such as Suzuki coupling and standard protection/deprotection strategies with commercially available starting materials, making it highly amenable to kilogram-scale manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Chiral Bisphosphine Ligand Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of advanced catalytic technologies like the chiral bisphosphine ligands described in CN109293700B. As a premier CDMO partner, we possess the technical expertise and infrastructure to translate these innovative laboratory protocols into commercial reality. Our team has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your transition from pilot plant to full-scale manufacturing is seamless. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of ligand or intermediate meets the highest industry standards, providing you with the confidence needed to advance your drug candidates through clinical trials.
We invite you to collaborate with us to leverage this cutting-edge chemistry for your next project. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific production volumes and requirements. Contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our supply chain solutions can drive efficiency and innovation in your pharmaceutical manufacturing operations.
