Advanced Manufacturing of High-Purity 1,4-Sorbitan for Pharmaceutical Intermediates
Advanced Manufacturing of High-Purity 1,4-Sorbitan for Pharmaceutical Intermediates
The global demand for high-performance surfactants in the pharmaceutical sector has necessitated a reevaluation of precursor manufacturing standards, particularly for materials intended for parenteral applications. Patent CN101948451A introduces a groundbreaking preparation method for high-purity 1,4-sorbitan, addressing the critical lack of industrial-scale techniques capable of producing injection-grade intermediates. This technology leverages a sophisticated two-stage dehydration protocol followed by a rigorous triple-crystallization refinement, effectively bridging the gap between bulk chemical production and stringent pharmaceutical compliance. By utilizing D-sorbitol as the primary feedstock, the process ensures a reliable supply chain foundation while implementing precise thermal and vacuum controls to dictate reaction selectivity. For R&D directors and procurement specialists, this patent represents a pivotal shift towards obtaining surfactant precursors that meet the exacting purity profiles required for modern drug delivery systems.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial dehydration of sorbitol has been plagued by significant selectivity issues, resulting in a heterogeneous mixture of anhydrous derivatives rather than a single, pure isomer. Traditional acid-catalyzed processes often yield a chaotic blend containing 1,4-anhydrosorbitol alongside unwanted isomers such as 2,5-anhydrosorbitol, 1,6-anhydrosorbitol, and even further dehydrated products like isosorbide. Furthermore, intermolecular dehydration frequently leads to the formation of sorbitan glycosides, complicating the downstream purification landscape immensely. This lack of specificity means that conventional outputs are generally unsuitable for synthesizing polysorbates intended for injection, as the impurity profile poses unacceptable risks for patient safety. Consequently, manufacturers relying on these legacy methods face substantial challenges in quality control, often resulting in batch-to-batch inconsistency that jeopardizes regulatory approval for high-value pharmaceutical formulations.
The Novel Approach
In stark contrast, the methodology outlined in CN101948451A employs a controlled, multi-step strategy designed to maximize the yield of the specific 1,4-isomer while systematically eliminating contaminants. The process initiates with the removal of free water from industrial sorbitol under vacuum before the introduction of the catalyst, preventing premature hydrolysis or uncontrolled side reactions. By carefully modulating the temperature between 90°C and 150°C under reduced pressure during the second dehydration stage, the reaction kinetics are tuned to favor intramolecular cyclization over intermolecular polymerization. Crucially, the innovation extends beyond synthesis into purification, utilizing a three-stage crystallization sequence in lower alcohols that acts as a powerful filter against structural analogs. This approach not only fills the technological void in domestic and international markets but also establishes a new benchmark for producing surfactant intermediates capable of meeting injection-level pharmacopoeia standards.
Mechanistic Insights into Acid-Catalyzed Intramolecular Dehydration
The core chemical transformation involves the acid-catalyzed intramolecular nucleophilic substitution where a hydroxyl group attacks a protonated adjacent carbon, leading to the expulsion of a water molecule and the formation of the cyclic ether ring. In the context of D-sorbitol, which possesses six hydroxyl groups, the challenge lies in directing this reaction specifically between the C1 and C4 positions to form 1,4-sorbitan rather than the thermodynamically competitive 1,6-isomer or other variants. The use of phosphorous acid or sulfuric acid facilitates the protonation of the hydroxyl leaving group, lowering the activation energy for water elimination. However, the selectivity is heavily dependent on the thermal profile; maintaining the reaction within the specified 90-150°C window under vacuum ensures that the kinetic product (1,4-sorbitan) is favored while minimizing the thermodynamic drive towards isosorbide formation. This precise control over reaction conditions is essential for suppressing the formation of glycosidic bonds, which would otherwise irreversibly consume the starting material and degrade the quality of the final intermediate.
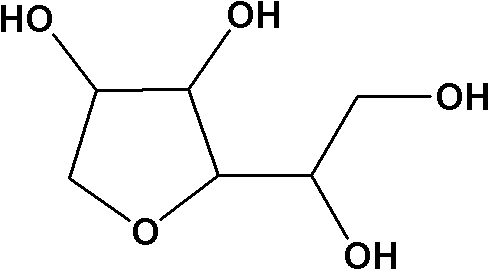
Beyond the primary cyclization, the mechanism of impurity rejection is equally critical to the overall success of this synthetic route. The patent highlights that even with optimized dehydration, the crude product contains a mixture of isomers that cannot be separated by simple distillation due to similar boiling points. Therefore, the purification mechanism relies on the differential solubility of 1,4-sorbitan versus its isomers in lower alcohols like methanol or ethanol at varying temperatures. By executing three sequential crystallizations, the process exploits the subtle differences in lattice energy and solvation shells between the target molecule and impurities such as 2,5-anhydrosorbitol. Each crystallization step incrementally enriches the solid phase with the desired 1,4-configuration, effectively scrubbing the product of trace contaminants that could otherwise catalyze degradation in the final surfactant application. This rigorous purification logic ensures that the final content exceeds 99%, a threshold necessary for the subsequent synthesis of polysorbates used in sensitive biological environments.
How to Synthesize 1,4-Sorbitan Efficiently
The synthesis of high-purity 1,4-sorbitan requires strict adherence to a protocol that balances thermal energy input with vacuum efficiency to drive the equilibrium towards water removal. Operators must first ensure the complete elimination of free water from the industrial sorbitol feedstock to prevent dilution of the acid catalyst and potential hydrolysis of the forming ether bond. Following the dehydration phase, the neutralization step must be performed with precision to remove residual acid without introducing excessive salt impurities that could co-crystallize. The detailed standardized synthesis steps, including specific stoichiometric ratios and cooling rates for the crystallization phases, are provided in the guide below to ensure reproducible results across different manufacturing scales.
- Remove free water from industrial D-sorbitol under vacuum at 120°C, then add an acidic catalyst like phosphorous acid.
- Perform intramolecular dehydration under reduced pressure at 90-150°C to form the cyclic ether structure.
- Neutralize the catalyst, decolorize with activated carbon, and perform three sequential crystallizations in lower alcohol to achieve >99% purity.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this patented process offers distinct strategic advantages centered around product consistency and downstream processing efficiency. By securing a source of 1,4-sorbitan that inherently meets high-purity specifications, manufacturers can significantly reduce the burden on their own quality assurance laboratories and minimize the risk of batch rejection during the final surfactant synthesis. The ability to produce an intermediate that is certified for injection-level applications opens up access to higher-margin pharmaceutical markets that were previously inaccessible due to raw material limitations. Furthermore, the reliance on common industrial feedstocks like D-sorbitol and standard mineral acids ensures that the supply chain remains robust and resistant to the volatility often associated with exotic or specialized reagents. This stability translates directly into more predictable lead times and a reduced need for safety stock, allowing for leaner inventory management strategies.
- Cost Reduction in Manufacturing: The elimination of complex chromatographic separation steps in favor of crystallization significantly lowers the operational expenditure associated with purification. Traditional methods often require expensive resin columns or solvent-intensive extraction processes to achieve similar purity levels, whereas this method utilizes cost-effective lower alcohols that can be recovered and recycled. Additionally, the high selectivity of the reaction reduces the amount of starting material wasted on byproduct formation, thereby improving the overall atom economy of the process. By avoiding the need for extensive post-synthesis remediation to remove toxic catalysts or heavy metal residues, manufacturers can also realize savings in waste disposal and environmental compliance costs.
- Enhanced Supply Chain Reliability: The use of widely available industrial sorbitol as the primary raw material mitigates the risk of supply disruptions that often plague niche chemical sectors. Since the process does not depend on proprietary or single-source catalysts, procurement teams have the flexibility to source reagents from multiple vendors, fostering a competitive pricing environment. The robustness of the dehydration and crystallization steps means that the manufacturing process is less susceptible to minor fluctuations in utility conditions, ensuring a steady output of qualified material. This reliability is crucial for maintaining continuous production schedules for downstream surfactant manufacturing, preventing costly downtime caused by raw material shortages or quality failures.
- Scalability and Environmental Compliance: The process design is inherently scalable, utilizing standard unit operations such as vacuum distillation and crystallization tanks that are common in fine chemical facilities. This compatibility with existing infrastructure allows for rapid capacity expansion without the need for significant capital investment in specialized equipment. From an environmental perspective, the closed-loop nature of the vacuum dehydration system minimizes volatile organic compound emissions, while the use of recyclable alcohols aligns with green chemistry principles. The absence of heavy metal catalysts further simplifies the treatment of process effluents, reducing the environmental footprint and ensuring compliance with increasingly stringent global regulations regarding industrial discharge.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production and application of high-purity 1,4-sorbitan based on the disclosed patent technology. These insights are derived directly from the experimental data and process descriptions to provide clarity on feasibility and performance. Understanding these details is essential for stakeholders evaluating the integration of this intermediate into their existing supply chains or product development pipelines.
Q: Why is traditional sorbitol dehydration insufficient for injection-grade surfactants?
A: Conventional dehydration produces a complex mixture of isomers (1,4; 2,5; 1,6-anhydrosorbitol) and glycosides, lacking the purity required for parenteral pharmaceutical excipients like Polysorbate 80.
Q: What catalysts are suitable for this 1,4-sorbitan synthesis?
A: The patent specifies the use of inorganic acids such as phosphorous acid or sulfuric acid, typically used in amounts ranging from 1% to 3% of the sorbitol weight.
Q: How is the final purity of >99% achieved?
A: High purity is attained through a rigorous triple-crystallization process using lower alcohols like methanol or ethanol, which effectively separates the target 1,4-isomer from side products.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable 1,4-Sorbitan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-purity intermediates in the development of safe and effective pharmaceutical formulations. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and efficiency. Our state-of-the-art facilities are equipped with rigorous QC labs capable of verifying stringent purity specifications, guaranteeing that every batch of 1,4-sorbitan meets the highest industry standards. We are committed to supporting your R&D efforts by providing materials that facilitate the synthesis of injection-grade surfactants, thereby accelerating your time to market.
We invite you to contact our technical procurement team to discuss how our advanced manufacturing capabilities can support your specific requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into how our optimized processes can enhance your bottom line. We encourage potential partners to reach out for specific COA data and route feasibility assessments, allowing us to demonstrate our commitment to quality and transparency in every interaction.
