Revolutionizing Mupirocin Production: A Solvent-Free Purification Strategy for Commercial Scale
The pharmaceutical industry is constantly seeking more sustainable and efficient methods for producing critical antibiotics, and the recent advancements detailed in patent CN110606844B represent a significant leap forward in the purification of Mupirocin. This patent introduces a groundbreaking solvent-free purification strategy that fundamentally alters the downstream processing landscape for this potent broad-spectrum antibiotic. Unlike conventional techniques that rely heavily on hazardous organic solvents, this novel approach leverages the precise interplay of aqueous chemistry, macroporous resin chromatography, and membrane technology to achieve exceptional purity levels exceeding 99%. 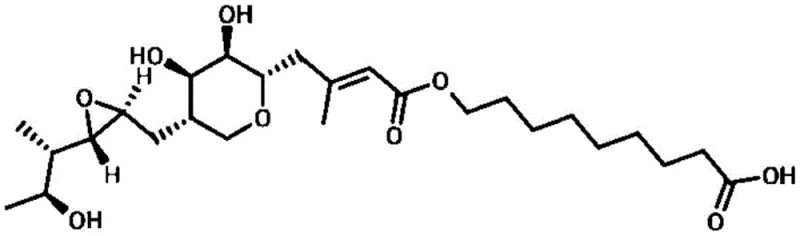 For R&D directors and process engineers, understanding the structural nuances of Mupirocin (C26H44O9), as depicted above, is crucial because its carboxylic acid and ester functionalities dictate the pH-sensitive behavior exploited in this new method. By shifting away from volatile organic compounds, this technology not only addresses stringent environmental regulations but also offers a pathway to drastically reduce operational expenditures associated with solvent procurement and waste management.
For R&D directors and process engineers, understanding the structural nuances of Mupirocin (C26H44O9), as depicted above, is crucial because its carboxylic acid and ester functionalities dictate the pH-sensitive behavior exploited in this new method. By shifting away from volatile organic compounds, this technology not only addresses stringent environmental regulations but also offers a pathway to drastically reduce operational expenditures associated with solvent procurement and waste management.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the isolation of Mupirocin from fermentation broths has been a chemically intensive process fraught with inefficiencies and safety concerns. Traditional protocols typically involve a multi-step sequence including membrane filtration of the broth, followed by liquid-liquid extraction using substantial volumes of organic solvents such as ethyl acetate or acetone. These solvents are necessary to partition the antibiotic from the aqueous fermentation matrix, but their usage introduces severe bottlenecks in manufacturing. The reliance on organic extraction necessitates complex solvent recovery systems to mitigate cost and environmental impact, yet even with recovery, there is always a risk of residual solvent contamination in the final Active Pharmaceutical Ingredient (API). Furthermore, the subsequent desorption steps from resin columns in older methods often require organic modifiers, which complicates the workflow and increases the fire hazard rating of the production facility. From a supply chain perspective, the volatility of organic solvent prices and the logistical burden of handling hazardous chemicals create unnecessary instability in the production schedule.
The Novel Approach
In stark contrast, the methodology outlined in CN110606844B eliminates the need for organic solvents entirely during the critical purification phases, marking a paradigm shift towards green chemistry in antibiotic manufacturing. This innovative route begins with the direct dissolution of crude Mupirocin in water, followed by a strategic adjustment of pH to the neutral range of 6.0 to 7.0 and the addition of inorganic salts. This preparation allows for direct loading onto medium-polarity macroporous resin columns, where the target molecule is selectively retained while impurities are washed away. The desorption is achieved not with harsh organics, but with a mild ammonium bicarbonate solution, leveraging the pH-dependent solubility of the molecule to release it from the resin matrix. Following chromatography, the process employs nanofiltration for concentration rather than energy-intensive thermal evaporation, preserving the integrity of the thermolabile antibiotic. This streamlined, aqueous-based workflow significantly simplifies the plant infrastructure requirements and removes the safety liabilities associated with flammable solvent storage and handling.
Mechanistic Insights into Aqueous Macroporous Resin Chromatography
The core scientific breakthrough of this patent lies in the sophisticated manipulation of solubility parameters within an entirely aqueous environment. Mupirocin possesses a carboxylic acid group that renders it pH-sensitive; it is poorly soluble in acidic water but highly soluble under alkaline conditions. The inventors discovered that by adding specific salts, such as sodium chloride at concentrations of 2-3 wt%, to the aqueous feed at pH 6.0-7.0, they could induce a 'salting-out' effect. This phenomenon effectively decreases the solubility of the Mupirocin in the bulk liquid, thereby driving its adsorption onto the hydrophobic pockets of the medium-polarity macroporous resin (specifically HZ-806). This mechanism ensures high loading capacity without the need for organic co-solvents to force adsorption. Furthermore, the choice of a medium-polarity resin is critical; it provides the optimal balance of hydrophobic interaction to retain the molecule during the wash phase while allowing for gentle elution when the chemical environment is altered.
Impurity control is meticulously managed through this pH-swing mechanism and the integration of nanofiltration. During the desorption phase, a dilute ammonium bicarbonate solution (0.01-0.03 wt%) is introduced. The bicarbonate ions raise the local pH, converting the Mupirocin into its more soluble salt form, which weakens its interaction with the resin and facilitates elution. This selective desorption leaves behind many non-ionizable or differently charged impurities on the column. Subsequently, the eluate undergoes nanofiltration using membranes with a molecular weight cut-off (MWCO) of 200-300 Daltons. This step is pivotal for removing small molecular weight salts and residual impurities while concentrating the Mupirocin to 40-60 g/L. Finally, re-acidifying the concentrate to pH 3.0-4.0 triggers crystallization. This acidic crystallization is superior to simple cooling crystallization because it specifically targets the isoelectric point behavior of the molecule, precipitating high-purity crystals while leaving UV-inactive impurities in the mother liquor, ensuring the final product meets the rigorous >99% purity specification.
How to Synthesize Mupirocin Efficiently
Implementing this purification protocol requires precise control over unit operations to maximize yield and purity. The process transforms crude fermentation outputs into pharmaceutical-grade material through a sequence of dissolution, chromatographic separation, membrane concentration, and controlled crystallization. Operators must strictly adhere to the pH windows defined in the patent, particularly the loading pH of 6.0-7.0 and the crystallization pH of 3.0-4.0, as deviations can lead to significant yield losses or purity failures. The following guide outlines the standardized operational framework derived from the patent examples, serving as a foundational reference for process validation and scale-up activities.
- Dissolve crude Mupirocin in water, add salt (NaCl), and adjust pH to 6.0-7.0 for loading onto medium-polarity macroporous resin.
- Desorb the target compound using a dilute ammonium bicarbonate solution (0.01-0.03 wt%) to obtain a qualified eluate.
- Concentrate the eluate via nanofiltration (MWCO 200-300), adjust pH to 3.0-4.0 for crystallization, and vacuum dry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this solvent-free purification technology offers profound strategic advantages that extend far beyond simple technical metrics. The most immediate impact is the drastic reduction in raw material costs associated with the elimination of high-volume organic solvents like ethyl acetate and acetone. In traditional manufacturing, the cost of purchasing, recovering, and disposing of these solvents constitutes a significant portion of the Cost of Goods Sold (COGS). By replacing these with inexpensive inorganic salts and water, the variable cost per kilogram of produced Mupirocin is significantly lowered. Additionally, the removal of flammable solvents reduces the insurance premiums and safety compliance costs associated with running a Class I hazardous location facility. This creates a more resilient cost structure that is less susceptible to fluctuations in the petrochemical market, providing long-term price stability for downstream buyers.
- Cost Reduction in Manufacturing: The economic benefits of this process are driven by the complete removal of organic solvent extraction and desorption steps. Without the need for large-scale solvent recovery distillation columns, the capital expenditure (CAPEX) for new facilities is reduced, and the operational expenditure (OPEX) for existing plants is minimized through lower energy consumption. The use of nanofiltration for concentration is inherently more energy-efficient than thermal evaporation, further driving down utility costs. Moreover, the simplified waste stream, consisting primarily of aqueous saline solutions rather than hazardous organic waste, drastically reduces the fees associated with environmental compliance and hazardous waste disposal, leading to substantial overall cost savings in antibiotic manufacturing.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the logistics of hazardous material transport and the regulatory hurdles surrounding solvent usage. By adopting an aqueous-based process, manufacturers mitigate the risk of production stoppages due to solvent supply shortages or transportation bans. The raw materials required for this method—salts, acids, bases, and water—are commodity chemicals with robust and stable global supply chains. This ensures that production schedules remain uninterrupted even during periods of market volatility. Furthermore, the simplified process flow reduces the number of potential failure points in the manufacturing line, enhancing the overall reliability of delivery timelines for high-purity Mupirocin intermediates.
- Scalability and Environmental Compliance: As regulatory bodies worldwide tighten restrictions on Volatile Organic Compound (VOC) emissions, this technology positions manufacturers as leaders in environmental stewardship. The process generates minimal hazardous waste, aligning perfectly with Green Chemistry principles and facilitating easier permitting for capacity expansions. The unit operations employed, such as macroporous resin chromatography and plate-and-frame filtration, are well-understood and easily scalable from pilot to commercial tonnage without the complex engineering challenges posed by large-scale solvent handling. This scalability ensures that suppliers can rapidly respond to surges in market demand for dermatological antibiotics without compromising on quality or safety standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced purification technology. These insights are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on how this method compares to legacy systems. Understanding these details is essential for technical teams evaluating the feasibility of adopting this greener manufacturing route.
Q: Why is the solvent-free method superior to traditional ethyl acetate extraction?
A: Traditional methods rely on large volumes of organic solvents like ethyl acetate or acetone, which pose significant safety hazards, environmental disposal costs, and residual solvent risks. The patented solvent-free method utilizes aqueous chromatography and nanofiltration, eliminating these risks while achieving purity greater than 99%.
Q: What is the role of salt and pH adjustment in this purification process?
A: Adding salt (such as NaCl) reduces the solubility of Mupirocin in the aqueous phase, enhancing its adsorption onto the macroporous resin. Adjusting the pH to 6.0-7.0 optimizes this interaction, while subsequent pH shifts facilitate efficient desorption and crystallization without organic modifiers.
Q: Can this process be scaled for industrial antibiotic manufacturing?
A: Yes, the process is explicitly designed for large-scale production. It replaces complex solvent recovery systems with robust unit operations like plate-and-frame filtration, resin chromatography, and nanofiltration, which are standard and easily scalable in industrial pharmaceutical facilities.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Mupirocin Supplier
The technological advancements described in patent CN110606844B highlight the evolving complexity of antibiotic production, requiring partners who possess both deep chemical expertise and robust manufacturing capabilities. NINGBO INNO PHARMCHEM stands at the forefront of this evolution, offering comprehensive CDMO services tailored to the specific needs of the pharmaceutical industry. Our facilities are equipped to handle diverse synthesis and purification pathways, ranging from early-stage process development to full-scale commercial production. We boast extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your supply needs are met with consistency and precision. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that utilize state-of-the-art analytical instrumentation to verify every batch against the highest international standards.
We invite global partners to collaborate with us to leverage these cutting-edge purification technologies for your supply chain. By working with our technical procurement team, you can access a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to solvent-free manufacturing processes for your specific portfolio. We encourage you to contact us today to request specific COA data and route feasibility assessments, allowing us to demonstrate how our advanced capabilities can drive efficiency and reliability in your Mupirocin supply chain.
