Optimizing Eplerenone Production: A Technical Analysis of the Microbial Fermentation Route
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with Eplerenone standing out as a highly selective aldosterone receptor antagonist essential for treating hypertension and heart failure. The efficiency of its production is paramount for global supply chains. Patent CN102617697A introduces a transformative methodology that shifts the synthetic paradigm from expensive precursors to a more accessible microbial fermentation pathway. This technical insight report dissects the proprietary process which begins with the bioconversion of Androstenedione (4-AD) into 9α-hydroxyandrostenedione, setting the stage for a series of precise semi-synthetic modifications. By leveraging biological catalysis for the initial functionalization, the process circumvents many of the harsh chemical conditions traditionally required, offering a greener and more economically viable pathway for industrial manufacturing.
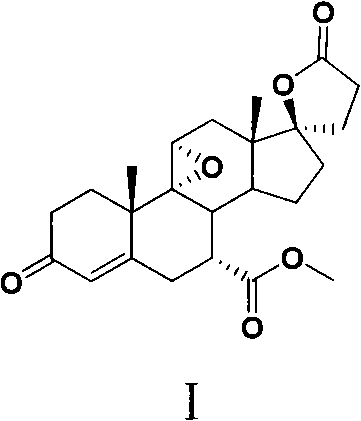
This structural complexity, characterized by the 9(11)-epoxy and 7α-methoxycarbonyl moieties, demands a synthesis strategy that balances stereochemical control with operational simplicity. The patent outlines a robust sequence involving dehydration, etherification, spiro-lactone formation, and strategic oxidation steps. For R&D directors and procurement specialists, understanding the nuances of this route is critical for evaluating potential suppliers who can deliver high-purity intermediates and APIs at a competitive cost structure without compromising on regulatory compliance or supply continuity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial synthesis of Eplerenone has relied heavily on Canrenone as the starting material, a pathway extensively documented in prior art such as US5981744. While chemically feasible, the Canrenone route presents significant economic and technical bottlenecks that hinder large-scale optimization. The primary drawback lies in the sourcing of Canrenone itself, which commands a substantially higher market price compared to other steroid precursors due to limited availability and complex upstream processing. Furthermore, the chemical transformations required to convert Canrenone into the Eplerenone scaffold involve multiple low-yielding steps, particularly the 11α-hydroxylation via microbial fermentation followed by cumbersome Michael additions and aldol condensations.
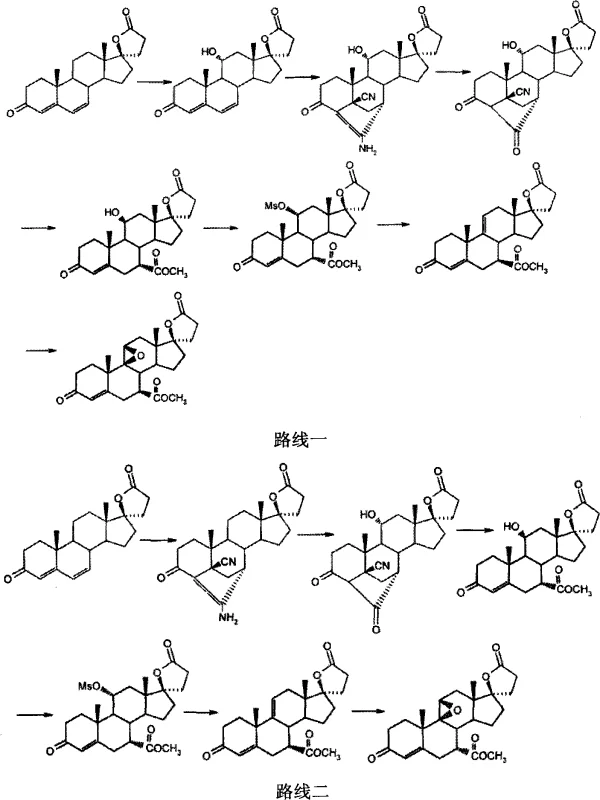
These condensation reactions often suffer from poor regioselectivity and stereoselectivity, necessitating extensive purification protocols that drive up waste generation and reduce overall throughput. The ring-opening reactions associated with these traditional pathways further exacerbate yield losses, creating a compounding effect on production costs. For supply chain managers, reliance on such a fragile synthetic sequence introduces volatility; any disruption in the supply of high-grade Canrenone or a deviation in the fermentation batch quality can halt production lines. Consequently, the industry has long sought an alternative route that decouples Eplerenone production from the constraints of the Canrenone supply chain while improving the atom economy of the synthesis.
The Novel Approach
The methodology disclosed in CN102617697A represents a strategic pivot towards utilizing Androstenedione (4-AD) as the foundational building block, a material that is not only cheaper but also produced at a massive scale globally for other steroid hormones. The innovation lies in the early-stage microbial conversion of 4-AD to 9α-hydroxyandrostenedione, effectively installing the oxygen functionality at the C9 position with high specificity. This biological step replaces several chemical oxidation/reduction cycles, streamlining the front end of the synthesis. Following this, the route employs a clever dehydration strategy using boron trifluoride complexes to establish the critical 9(11) double bond, which serves as the handle for the final epoxidation.
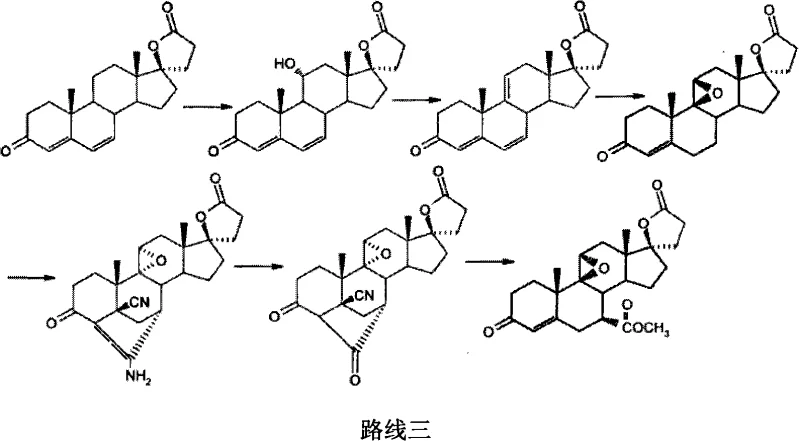
Subsequent steps involve the protection of the 3-keto group as an enol ether, followed by a sophisticated sequence to construct the C17 spiro-lactone ring. Unlike the traditional routes that struggle with the C17 side chain installation, this novel approach utilizes a condensation with ethyl malonate under controlled basic conditions, ensuring high fidelity in forming the gamma-lactone ring. The introduction of the 7α-substituent is achieved through a unique furanization followed by oxidative cleavage and esterification, a tactic that offers superior control over the stereochemistry at the C7 position compared to direct alkylation methods. This comprehensive re-engineering of the synthetic tree results in a process that is not only more cost-effective but also inherently more scalable for multi-ton production campaigns.
Mechanistic Insights into Spiro-Lactone Formation and Furan Oxidation
A deep dive into the reaction mechanisms reveals the sophistication of this synthetic design, particularly in the construction of the D-ring lactone and the installation of the 7α-ester. The formation of the spiro-lactone at C17 is a pivotal moment in the synthesis, achieved through a variation of the Corey-Chaykovsky reaction conditions. In this specific protocol, the 17-keto precursor is treated with trimethylsulfonium iodide in the presence of a strong base like sodium hydride within a DMSO/THF solvent system. This generates a sulfur ylide in situ, which attacks the carbonyl carbon to form an epoxide intermediate. This epoxide is not isolated but immediately subjected to condensation with ethyl malonate in an ethanolic solution of sodium ethylate.
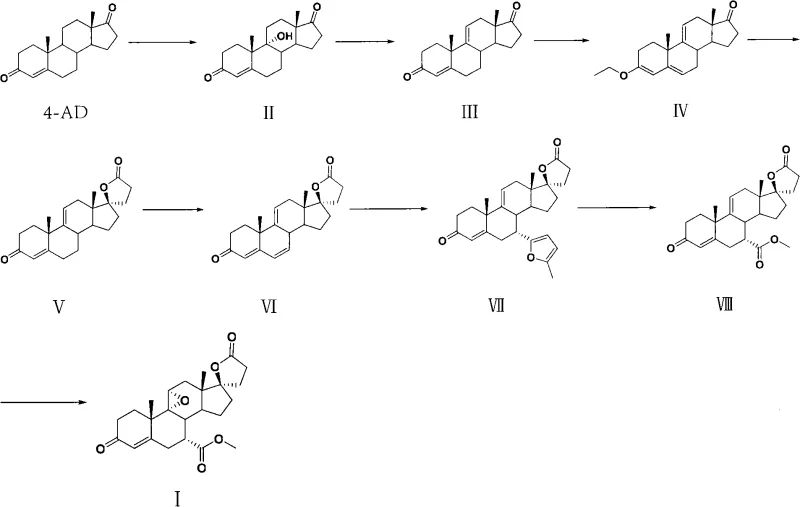
The mechanism proceeds through a nucleophilic attack by the malonate anion on the epoxide, followed by ring opening and subsequent intramolecular cyclization to close the lactone ring. The careful control of temperature, dropping from 70-80°C during ylide formation to -10-0°C during the addition of the ketone, is crucial to minimizing side reactions such as polymerization or over-alkylation. Following the lactone formation, the decarboxylation step in alkaline water/alkoxide solution cleanly removes the extra carboxyl group, leaving the desired spiro-structure intact. This sequence demonstrates high chemoselectivity, tolerating the sensitive enone system in the A-ring without requiring additional protecting groups, thereby reducing step count and waste.
Equally critical is the transformation of the 7α-furan moiety into the 7α-methoxycarbonyl group. The patent describes a two-stage oxidation process where the furan ring is first oxidized, likely forming a reactive intermediate that is susceptible to nucleophilic attack by methanol in the presence of coupling agents like EDCI and DMAP. This oxidative esterification is a delicate operation; over-oxidation can lead to ring degradation, while under-oxidation leaves the furan intact, complicating purification. The use of specific oxidants and the maintenance of low temperatures (0-5°C) during the esterification phase ensure that the methoxycarbonyl group is installed with the correct alpha-stereochemistry. Finally, the 9(11) double bond is epoxidized using benzoyl hydroperoxide in a non-polar solvent. The choice of solvent and the precise temperature window of 48-50°C are vital to achieving the alpha-epoxide configuration required for biological activity, yielding a product with purity exceeding 99.5% after recrystallization.
How to Synthesize Eplerenone Efficiently
The synthesis of Eplerenone via this microbial-semi-synthetic hybrid route requires precise adherence to reaction parameters to maximize yield and purity. The process integrates biological catalysis with advanced organic transformations, demanding a facility equipped for both fermentation and complex chemical synthesis. Operators must pay close attention to the stoichiometry of the boron trifluoride complex during dehydration and the moisture content during the spiro-lactone formation, as water can quench the reactive ylide intermediates. The following guide outlines the standardized operational framework derived from the patent examples, serving as a baseline for process engineers aiming to replicate this high-efficiency pathway.
- Convert Androstenedione (4-AD) to 9α-hydroxyandrostenedione via microbial fermentation.
- Perform dehydration using a boron trifluoride complex to introduce the 9(11) double bond.
- Execute 17-keto epoxidation, condensation with ethyl malonate, and decarboxylation to form the spiro-lactone ring.
- Introduce the 6(7) double bond, followed by furanization at the 7-alpha position.
- Oxidize the furan ring to a methoxycarbonyl group and perform final 9,11-epoxidation to yield Eplerenone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift from Canrenone to Androstenedione as the starting material represents a fundamental improvement in supply security and cost structure. The traditional reliance on Canrenone exposes buyers to price volatility and supply bottlenecks, as it is a niche intermediate with fewer global producers. In contrast, Androstenedione is a commodity steroid intermediate produced in vast quantities for the synthesis of testosterone and other hormones, ensuring a robust and diversified supply base. This abundance translates directly into reduced raw material costs and greater negotiating leverage for purchasers. Furthermore, the elimination of low-yielding Michael addition steps significantly improves the overall mass balance of the process, meaning less raw material is required to produce a kilogram of final API, driving down the variable cost of goods sold.
- Cost Reduction in Manufacturing: The economic benefits of this route are driven by the substitution of expensive starting materials and the simplification of the synthetic sequence. By utilizing 4-AD, which is significantly cheaper than Canrenone, the baseline material cost is drastically lowered. Additionally, the high-yielding microbial fermentation step replaces multiple chemical steps that typically require expensive reagents and generate substantial waste. The streamlined process reduces the consumption of solvents and auxiliary chemicals, leading to lower utility costs and waste disposal fees. The avoidance of transition metal catalysts in key steps further eliminates the need for costly metal scavenging and validation processes, contributing to a leaner manufacturing budget.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of widely available feedstocks. Since 4-AD is produced by multiple manufacturers globally using established fermentation technologies, the risk of single-source dependency is minimized. The process described is also highly amenable to scale-up; the fermentation step can be expanded to large bioreactors, and the subsequent chemical steps utilize standard unit operations like extraction, crystallization, and filtration that are common in multipurpose pharmaceutical plants. This flexibility allows manufacturers to ramp up production quickly in response to market demand spikes without requiring specialized or hard-to-source equipment, ensuring consistent delivery schedules for downstream clients.
- Scalability and Environmental Compliance: From an environmental and regulatory standpoint, this route offers distinct advantages. The microbial conversion step is inherently greener, operating under mild conditions and generating less hazardous waste compared to harsh chemical oxidations. The overall reduction in step count means fewer opportunities for solvent loss and emission, aligning with increasingly stringent environmental regulations. The process avoids the use of heavy metals and toxic reagents where possible, simplifying the environmental impact assessment and permitting process for new manufacturing sites. This 'green' profile not only reduces compliance costs but also enhances the brand value of the final product for pharmaceutical customers seeking sustainable supply chains.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Eplerenone using the patented microbial fermentation route. These insights are derived directly from the experimental data and process descriptions found in CN102617697A, providing clarity on the feasibility and advantages of this method for potential partners and licensees.
Q: What is the primary advantage of using Androstenedione over Canrenone for Eplerenone synthesis?
A: Androstenedione is significantly cheaper and more readily available than Canrenone. Furthermore, the microbial fermentation route avoids the low-yielding Michael addition and aldol condensation steps associated with Canrenone-based syntheses, leading to better overall process economics.
Q: How is the 7-alpha-methoxycarbonyl group introduced in this novel route?
A: The route utilizes a furanization strategy where a 7-alpha-(5'-methyl-2'-furyl) group is first introduced. This furan ring is subsequently oxidized and esterified to generate the critical 7-alpha-methoxycarbonyl functionality required for eplerenone activity.
Q: What are the critical reaction conditions for the final epoxidation step?
A: The final epoxidation of the 9(11) double bond is conducted using benzoyl hydroperoxide in a non-polar solvent such as methylene dichloride or toluene. The reaction is typically maintained at 48-50°C for 6-7 hours to ensure high conversion and purity greater than 99.5% after recrystallization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eplerenone Supplier
The technical superiority of the microbial fermentation route for Eplerenone synthesis underscores the importance of partnering with a manufacturer that possesses both biological and chemical expertise. NINGBO INNO PHARMCHEM stands at the forefront of this capability, combining advanced fermentation technology with state-of-the-art semi-synthetic processing. Our facilities are designed to handle the complexities of steroid chemistry, from the initial bioconversion of Androstenedione to the final crystallization of high-purity Eplerenone. We bring extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive a consistent and reliable supply of material regardless of volume requirements. Our commitment to quality is enforced through stringent purity specifications and rigorous QC labs that monitor every critical parameter of the synthesis.
We invite pharmaceutical companies and generic drug manufacturers to explore the cost and efficiency benefits of this novel supply chain. By leveraging our optimized process, you can secure a competitive edge in the cardiovascular market through reduced COGS and improved supply security. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your specific volume needs. We are ready to provide specific COA data and route feasibility assessments to demonstrate how our Eplerenone production capabilities can support your long-term strategic goals.
