Advanced Synthetic Route for Eplerenone Intermediates Enhancing Commercial Scalability and Purity
The pharmaceutical landscape for cardiovascular therapeutics continues to evolve, with Eplerenone standing out as a critical selective aldosterone antagonist for managing hypertension and congestive heart failure. Patent CN1321128C introduces a pivotal advancement in the preparation of pregna-4-ene-7,21-dicarboxylic acid derivatives, specifically targeting the efficient synthesis of this high-value active pharmaceutical ingredient. Unlike traditional methodologies that rely heavily on late-stage modifications or biological interventions, this disclosed technology leverages a strategic early-stage elimination of the 11-hydroxyl group to streamline the entire synthetic trajectory. For global procurement leaders and technical directors, understanding this shift is essential, as it represents a move towards more predictable, chemically driven manufacturing paradigms that reduce dependency on variable biological systems. The innovation lies not just in the molecule itself, but in the architectural redesign of the synthesis pathway to favor industrial scalability and purity control from the outset.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Eplerenone and its key intermediates has been dominated by routes originating from Canrenone or Hydrocortisone, which inherently carry significant process burdens. As illustrated in the prior art pathways, these conventional methods often necessitate microbial transformation steps to introduce oxygenation at the 11-position, a process notorious for its low conversion rates and extensive downstream purification requirements. 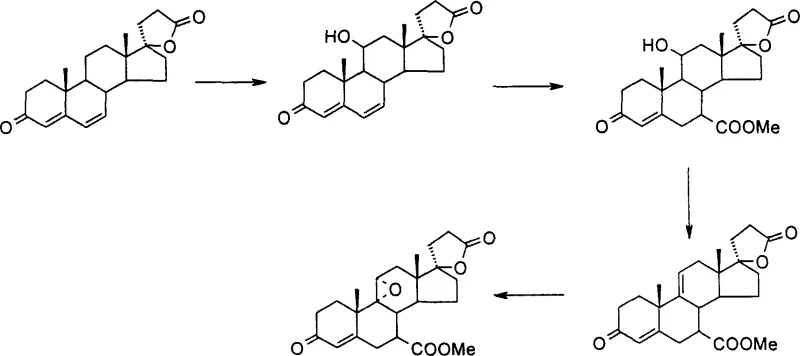 . Furthermore, performing the elimination of the 11-hydroxyl group in the final stages of synthesis, as seen in patents like WO9721720, creates a bottleneck where valuable advanced intermediates are risked in low-yielding dehydration reactions. This late-stage complexity amplifies the cost of goods sold and introduces substantial supply chain fragility, as any failure in the biological step compromises the entire batch of high-value material. The reliance on specific microbial strains also limits the ability to rapidly scale production in standard chemical reactors, creating a disconnect between R&D potential and commercial reality.
. Furthermore, performing the elimination of the 11-hydroxyl group in the final stages of synthesis, as seen in patents like WO9721720, creates a bottleneck where valuable advanced intermediates are risked in low-yielding dehydration reactions. This late-stage complexity amplifies the cost of goods sold and introduces substantial supply chain fragility, as any failure in the biological step compromises the entire batch of high-value material. The reliance on specific microbial strains also limits the ability to rapidly scale production in standard chemical reactors, creating a disconnect between R&D potential and commercial reality.
The Novel Approach
In stark contrast, the methodology outlined in CN1321128C fundamentally reorders the synthetic logic by prioritizing the elimination of the 11-hydroxyl group immediately after sourcing the starting material. By utilizing 11α-hydroxy-4-ene-androst-3,17-dione as the foundational building block, the process converts the problematic hydroxyl moiety into a double bond at the very beginning, establishing the 4,9-diene system early. This strategic pivot eliminates the need for any microbial fermentation, replacing it with robust chemical reagents such as p-toluenesulfonic acid in toluene under reflux conditions. The subsequent construction of the 17-position lactone ring and the 7-alpha ester functionality proceeds on a stabilized diene framework, which offers superior chemical handling properties. This approach not only simplifies the operational workflow but also aligns perfectly with the needs of a reliable pharmaceutical intermediates supplier seeking to minimize process variability and maximize throughput in a GMP-compliant environment.
Mechanistic Insights into Early-Stage Elimination and Lactone Formation
The core chemical innovation resides in the acid-catalyzed dehydration mechanism that transforms the 11α-hydroxyl group into the 9(11)-double bond precursor. When 11α-hydroxy-4-ene-androst-3,17-dione is treated with p-toluenesulfonic acid in toluene, the reaction proceeds through a carbocation intermediate that is stabilized by the adjacent steroid skeleton, facilitating the loss of water to form the conjugated diene system. This step is critical because it removes a polar, reactive handle early, preventing side reactions in subsequent nucleophilic additions. Following this, the introduction of the 17-alpha side chain utilizes a Corey-Chaykovsky type epoxidation followed by ring opening with diethyl malonate. The precise control of temperature, dropping from 70°C during epoxide formation to -5°C during reagent addition, is paramount to ensuring regioselectivity and preventing degradation of the sensitive enone system. This level of thermal management demonstrates a deep understanding of steroid reactivity, ensuring that the resulting 17β-hydroxy-3-oxo-17α-pregna-4,9-diene-21-carboxylic acid-γ-lactone is formed with high stereochemical integrity.
Impurity control is further enhanced by the sequential oxidation and functionalization steps employed to install the 7-alpha methyl ester. The use of DDQ (2,3-dichloro-5,6-dicyano-p-benzoquinone) for the dehydrogenation of the B-ring allows for the selective formation of the 4,6,9-triene system without over-oxidizing other sensitive sites on the steroid nucleus. Subsequent conjugate addition of acetone cyanohydrin followed by hydrolysis and esterification ensures that the 7-position is functionalized with the correct alpha-stereochemistry. By avoiding the random oxidation patterns often seen in microbial routes, this chemical pathway produces a much cleaner impurity profile. For R&D directors, this means fewer unknown peaks in HPLC chromatograms and a significantly reduced burden on analytical teams during method validation. The rigorous washing protocols described, involving sodium sulfite and bicarbonate solutions, further ensure that residual oxidants and acidic byproducts are completely removed, safeguarding the stability of the final crystalline product.
How to Synthesize Eplerenone Efficiently
The synthesis of this complex steroid intermediate requires precise adherence to the patented sequence to ensure optimal yield and purity. The process begins with the dehydration of the starting androstenedione derivative, followed by a multi-step sequence involving epoxidation, lactonization, and selective oxidation. Each stage demands strict control over reaction parameters such as temperature, pH, and stoichiometry to prevent the formation of isomeric byproducts. The detailed standardized synthesis steps below outline the critical operational windows required to replicate this high-efficiency pathway successfully in a pilot or production plant setting.
- Eliminate the 11-alpha-hydroxyl group from 11α-hydroxy-4-ene-androst-3,17-dione using p-toluenesulfonic acid in toluene to form the 4,9-diene system.
- Perform epoxidation at the 17-position using trimethylsulfonium iodide and sodium hydride, followed by ring opening with diethyl malonate to establish the lactone precursor.
- Introduce the 7-alpha functionality via DDQ oxidation and subsequent cyanohydrin addition, concluding with final epoxidation to yield the target stereochemistry.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition from microbial to purely chemical synthesis represents a significant de-risking of the supply base. The elimination of fermentation steps removes the long lead times associated with cell culture maintenance and biomass processing, allowing for much more responsive manufacturing schedules. This agility is crucial in the volatile pharmaceutical market where demand for cardiovascular medications can fluctuate rapidly. By adopting a route that relies on commodity chemicals and standard reactor vessels, manufacturers can leverage existing capacity more effectively, avoiding the capital expenditure required for specialized biotech infrastructure. This structural efficiency translates directly into a more resilient supply chain capable of withstanding external shocks and maintaining continuous delivery schedules for critical API intermediates.
- Cost Reduction in Manufacturing: The economic benefits of this process are derived primarily from the simplification of the unit operations and the removal of expensive biological processing costs. By eliminating the need for sterile fermentation tanks and the associated downstream separation of biomass, the overall overhead per kilogram of product is drastically reduced. Furthermore, the higher chemical yields achieved through the optimized elimination and functionalization steps mean that less raw material is wasted, improving the atom economy of the entire process. These factors combine to create a leaner cost structure that allows for competitive pricing without compromising on the quality standards required for pharmaceutical grade materials.
- Enhanced Supply Chain Reliability: Reliability is bolstered by the use of readily available starting materials such as 11α-hydroxy-androstenedione, which is a common feedstock in the steroid industry. Unlike specialized microbial strains that may have single-source vulnerabilities, the chemical reagents used in this pathway are sourced from a broad global market, reducing the risk of supply disruption. The robustness of the chemical reactions also means that batch failure rates are minimized, ensuring a consistent flow of material to downstream API manufacturers. This predictability allows supply chain planners to maintain lower safety stock levels while still meeting service level agreements, optimizing working capital across the value chain.
- Scalability and Environmental Compliance: Scaling this process from laboratory to commercial tonnage is straightforward due to the absence of biological constraints that often hinder scale-up. The reaction conditions, involving standard organic solvents and manageable temperatures, are easily replicated in large-scale glass-lined or stainless steel reactors. Additionally, the waste streams generated are primarily chemical in nature and can be treated using established effluent treatment protocols, avoiding the complex bio-hazardous waste disposal issues associated with fermentation broths. This environmental compatibility facilitates smoother regulatory approvals and supports the sustainability goals of modern pharmaceutical manufacturing facilities.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis route. They are derived from the specific technological differentiators found in the patent documentation, focusing on the practical implications for manufacturing and quality assurance. Understanding these details is vital for stakeholders evaluating the feasibility of integrating this intermediate into their broader production portfolios.
Q: Why is the elimination of the 11-hydroxyl group performed early in this synthesis?
A: Performing the elimination of the 11-hydroxyl group at the beginning of the synthesis, rather than at the end as seen in conventional Canrenone-based routes, significantly simplifies the reaction sequence. This strategic modification avoids the need for complex microbial transformation steps which often suffer from low yields and batch-to-batch variability, thereby enhancing the overall robustness of the manufacturing process.
Q: What are the primary advantages of this route over microbial transformation methods?
A: The primary advantage lies in the complete avoidance of biological fermentation steps. Microbial transformations, while specific, introduce challenges related to sterility, long fermentation cycles, and difficult downstream processing. By utilizing a purely chemical synthesis pathway starting from readily available 11α-hydroxy-androstenedione, the process ensures faster turnaround times, easier scale-up, and more consistent impurity profiles suitable for stringent regulatory requirements.
Q: How does this method impact the cost structure of Eplerenone production?
A: This method impacts the cost structure positively by reducing the number of unit operations and eliminating the high overhead associated with biotechnological facilities. The use of common chemical reagents and standard reactor setups allows for better utilization of existing fine chemical infrastructure. Furthermore, the improved operational simplicity reduces labor intensity and energy consumption per kilogram of produced intermediate, leading to substantial long-term economic benefits.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Eplerenone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of robust synthetic pathways in securing the global supply of life-saving cardiovascular medications. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical advantages of patent CN1321128C are fully realized in practice. We operate with stringent purity specifications and maintain rigorous QC labs to verify that every batch of Eplerenone intermediate meets the exacting standards required by international regulatory bodies. Our commitment to technical excellence ensures that the complex stereochemistry and impurity profiles of this molecule are managed with the highest level of precision.
We invite forward-thinking procurement leaders to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic potential of switching to this non-microbial pathway. We encourage you to contact us today to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a seamless transition to a more efficient and reliable sourcing strategy for your pharmaceutical intermediates.
