Advanced Catalytic Route for High-Purity Nifedipine Intermediates and Commercial Scalability
Advanced Catalytic Route for High-Purity Nifedipine Intermediates and Commercial Scalability
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic efficiency, particularly for widely prescribed cardiovascular medications like Nifedipine. Patent CN111233672B introduces a transformative methodology for synthesizing the critical nifedipine intermediate, methyl o-nitrobenzylidene acetoacetate, utilizing a sophisticated combined catalyst system. This innovation addresses long-standing challenges in the Knoevenagel condensation step, specifically targeting the reduction of genotoxic impurities and streamlining the purification process. By employing a dual-catalyst approach involving nitrogen-containing heterocyclic amines and carboxylic acids, the technology enables the direct crystallization of high-purity intermediates from the reaction matrix. This breakthrough not only enhances the chemical integrity of the final API but also offers significant operational advantages for manufacturers aiming to optimize their supply chains and reduce production costs in a competitive market.
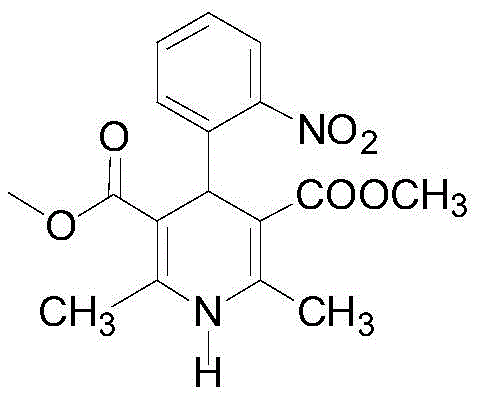
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
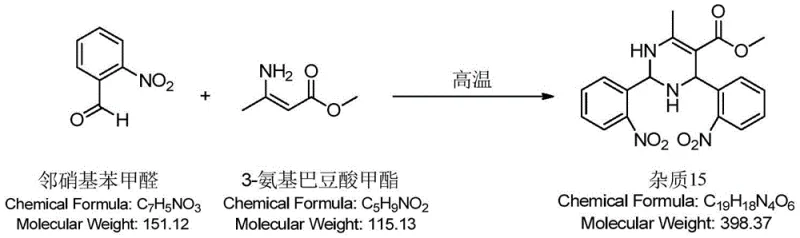
Traditional synthesis routes for nifedipine often rely on the classic Hantzsch dihydropyridine synthesis, which can be executed as a one-pot reaction or a two-step one-pot process. In the conventional one-pot method, the strong alkalinity of ammonia water frequently triggers numerous side reactions, compromising the overall yield and purity of the final product. Even when weaker bases like ammonium bicarbonate are substituted, the improvement in product purity remains marginal. Furthermore, the two-step one-pot approach, while an improvement, fails to adequately remove residual starting materials, particularly o-nitrobenzaldehyde, which is a known genotoxic impurity. Because the intermediate is not isolated before the cyclization step, unreacted aldehyde persists in the reaction mixture and can react with methyl 3-aminocrotonate to form difficult-to-remove byproducts like Impurity 15. This necessitates extensive and costly recrystallization processes to meet pharmacopoeial standards, thereby increasing manufacturing lead times and reducing overall process efficiency.
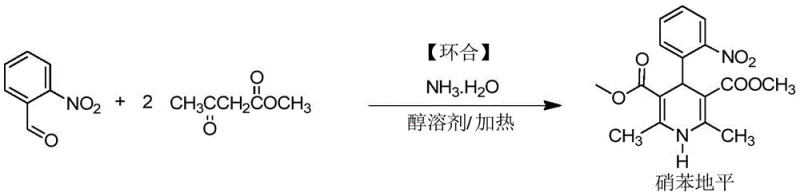
The Novel Approach
The methodology disclosed in the patent represents a paradigm shift by decoupling the condensation and cyclization steps into a 'two-step, two-pot' strategy, underpinned by a highly selective combined catalyst system. This approach facilitates the Knoevenagel condensation of o-nitrobenzaldehyde and methyl acetoacetate under mild conditions, allowing the intermediate to crystallize naturally from the alcohol solvent upon cooling. This physical separation is crucial; it ensures that unreacted raw materials, catalysts, and soluble impurities remain in the mother liquor, yielding a crude intermediate with purity exceeding 98.5% without the need for further recrystallization. The reaction proceeds efficiently at temperatures between 40°C and 60°C, avoiding the thermal stress that often degrades sensitive active methylene compounds. By isolating a high-quality intermediate before the ring-closing step, the formation of downstream genotoxic impurities is drastically minimized, ensuring a cleaner final API profile.
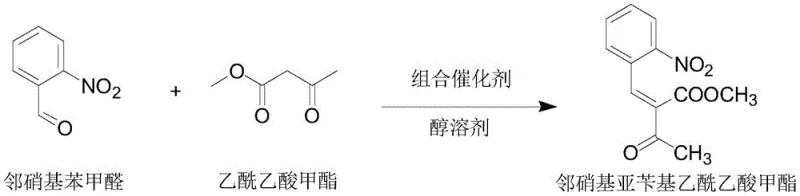
Mechanistic Insights into Combined Catalyst Synergy
The core innovation lies in the specific composition of the combined catalyst, which typically comprises a nitrogen-containing heterocyclic secondary amine, such as piperidine or its alkyl derivatives, and a nitrogen-containing heterocyclic carboxylic acid, such as substituted indole or pyridine carboxylic acids. This binary system creates a buffered reaction environment with a pH ranging from 6.15 to 8.05, which is near-neutral. This pH control is vital because the nifedipine intermediate is an active methylene compound susceptible to decomposition under strongly acidic or basic conditions. The amine component activates the methylene group of the acetoacetate, while the carboxylic acid component modulates the basicity and potentially stabilizes transition states through hydrogen bonding. Experimental data indicates that combinations involving indole carboxylic acids, particularly 2-indolecarboxylic acid or 3-indolecarboxylic acid paired with piperidine, offer superior catalytic performance compared to pyridine-based acids, likely due to better steric and electronic compatibility with the ortho-nitro substituent on the benzaldehyde ring.
A critical aspect of this mechanistic advantage is the suppression of specific genotoxic impurities, most notably Impurity 15, which arises from the cross-reaction of residual o-nitrobenzaldehyde with the aminocrotonate in subsequent steps. By ensuring high conversion of the aldehyde in the first step and physically removing the unreacted portion via crystallization, the feedstock for this side reaction is effectively eliminated. The crystallization behavior is also enhanced by the catalyst choice; the intermediate precipitates as white, lustrous crystals with uniform particle size upon cooling to below 20°C. This contrasts sharply with older methods that yielded brown-red oils requiring complex solvent exchanges and multiple recrystallizations. The ability to obtain a solid, high-purity intermediate directly simplifies the workflow and significantly lowers the risk of carrying over toxic aldehydes into the final drug substance.
How to Synthesize Methyl o-nitrobenzylidene acetoacetate Efficiently
The synthesis protocol outlined in the patent provides a clear roadmap for implementing this technology in a production setting, emphasizing precise control over stoichiometry and temperature to maximize yield and purity. The process begins with the preparation of the reaction mixture in a lower aliphatic alcohol solvent, with methanol demonstrating the highest efficacy in terms of reaction rate and product quality. The sequential addition of reagents followed by the introduction of the pre-mixed or in-situ formed catalyst system ensures homogeneity before the exothermic condensation begins. Maintaining the reaction temperature within the narrow window of 50°C to 55°C is critical; deviations towards room temperature slow the kinetics, while reflux conditions may promote degradation. Real-time monitoring via HPLC allows operators to pinpoint the reaction endpoint accurately, preventing over-reaction. Once the target peak area stabilizes, the mixture is cooled gradually to induce crystallization, followed by filtration and washing with cold solvent to remove surface impurities. This streamlined procedure eliminates the need for vacuum concentration, preserving the integrity of the thermally sensitive intermediate.
- Prepare the reaction vessel with alcohol solvent (methanol preferred) and add o-nitrobenzaldehyde and methyl acetoacetate under stirring.
- Introduce the combined catalyst system consisting of a nitrogen-containing heterocyclic secondary amine and a nitrogen-containing heterocyclic carboxylic acid.
- Maintain reaction temperature between 40-60°C until HPLC indicates completion, then cool to crystallize the pure intermediate directly from the mother liquor.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this catalytic technology translates into tangible operational efficiencies and risk mitigation strategies. The primary value proposition lies in the drastic simplification of the purification workflow. By achieving high purity (≥98.5%) directly from the reaction crystallization, manufacturers can eliminate multiple recrystallization cycles and the associated solvent consumption, leading to substantial cost reductions in raw material usage and waste disposal. The removal of the vacuum concentration step further decreases energy consumption and equipment occupancy time, enhancing the overall throughput of the production facility. Additionally, the robustness of the catalyst system ensures consistent batch-to-batch quality, reducing the variability that often plagues multi-step organic syntheses. This reliability is essential for maintaining uninterrupted supply lines to API manufacturers who operate under strict regulatory timelines.
- Cost Reduction in Manufacturing: The elimination of energy-intensive vacuum concentration and multiple recrystallization steps significantly lowers utility and solvent costs. The high molar yield and purity mean less raw material is wasted on reprocessing off-spec batches, directly improving the cost of goods sold (COGS) for the intermediate. Furthermore, the use of common, commercially available catalysts like piperidine and indole carboxylic acids avoids the expense of exotic or proprietary reagents, keeping input costs stable and predictable for long-term contracts.
- Enhanced Supply Chain Reliability: The simplified process flow reduces the number of unit operations required, which inherently lowers the probability of mechanical failure or operator error causing delays. The ability to produce a stable, solid intermediate that does not require immediate further processing allows for flexible inventory management; the intermediate can be stored and transported safely if needed, decoupling the condensation step from the cyclization step. This flexibility provides a buffer against supply chain disruptions, ensuring that API production schedules can be met even if upstream raw material deliveries face minor fluctuations.
- Scalability and Environmental Compliance: Operating at mild temperatures (50°C) and atmospheric pressure makes this process inherently safer and easier to scale from pilot plant to commercial tonnage compared to high-pressure or high-temperature alternatives. The reduction in solvent usage and the avoidance of heavy metal catalysts align with green chemistry principles, simplifying environmental permitting and wastewater treatment compliance. This eco-friendly profile is increasingly important for multinational corporations aiming to meet sustainability goals and reduce their carbon footprint across the pharmaceutical supply chain.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this combined catalyst technology for nifedipine intermediate synthesis. These insights are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on catalyst selection, impurity control, and process parameters. Understanding these nuances is essential for technical teams evaluating the feasibility of technology transfer and for procurement specialists assessing the quality assurances provided by suppliers utilizing this method.
Q: How does the combined catalyst reduce genotoxic impurities compared to traditional methods?
A: The novel catalyst system maintains a near-neutral pH environment (6.15-8.05), which suppresses the side reaction between o-nitrobenzaldehyde and methyl 3-aminocrotonate that forms Impurity 15, while facilitating the crystallization of the intermediate to separate unreacted aldehyde.
Q: What are the optimal catalyst components for this synthesis?
A: The most effective combination involves piperidine or its derivatives paired with substituted indole carboxylic acids (such as 2-indolecarboxylic acid or 3-indolecarboxylic acid), which provide superior catalytic activity and purity profiles compared to pyridine-based acids.
Q: Is this process suitable for large-scale commercial production?
A: Yes, the process operates at mild temperatures (50°C) without the need for energy-intensive vacuum concentration steps, and the product crystallizes directly from the reaction mixture, simplifying isolation and making it highly scalable for industrial API manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Nifedipine Intermediate Supplier
As the global demand for high-quality cardiovascular medications continues to rise, securing a dependable source of critical intermediates is paramount for pharmaceutical success. NINGBO INNO PHARMCHEM stands at the forefront of this sector, leveraging advanced catalytic technologies like the one described in CN111233672B to deliver superior chemical building blocks. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major API manufacturers without compromising on quality. We maintain stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instruments to verify that every batch meets or exceeds pharmacopoeial standards, particularly regarding genotoxic impurity limits.
We invite you to collaborate with us to optimize your supply chain and reduce your manufacturing costs. Our technical experts are ready to provide a Customized Cost-Saving Analysis tailored to your specific production needs, demonstrating how our advanced synthesis routes can improve your bottom line. Please contact our technical procurement team today to request specific COA data, route feasibility assessments, and samples for your evaluation. Let us be your partner in delivering safe, effective, and affordable medicines to patients worldwide through superior chemical innovation and reliable supply chain execution.
