Scalable Green Synthesis of Dihydropyridine Intermediates Using Deep Eutectic Solvents
Scalable Green Synthesis of Dihydropyridine Intermediates Using Deep Eutectic Solvents
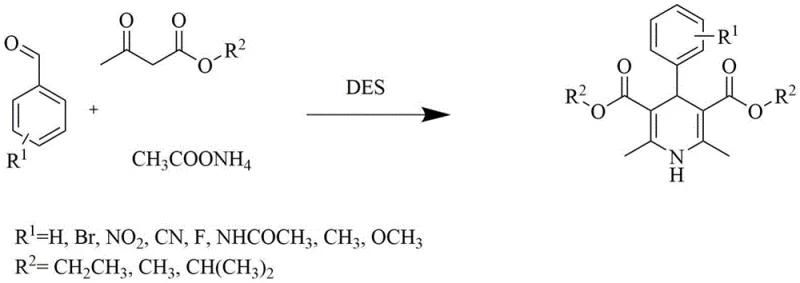
The pharmaceutical industry is constantly seeking sustainable alternatives to traditional synthetic routes, particularly for high-volume intermediates like dihydropyridines, which serve as the core scaffold for widely prescribed antihypertensive agents such as Nifedipine. A groundbreaking advancement in this domain is detailed in patent CN114213318A, which discloses a novel green synthesis process utilizing Deep Eutectic Solvents (DES) to replace volatile organic compounds and expensive ionic liquids. This technology addresses critical pain points in the manufacturing of nitrogen heterocyclic compounds by integrating reaction and separation into a streamlined workflow that minimizes environmental impact while maximizing yield. By leveraging the unique hydrogen-bonding networks of choline chloride-based solvents, this method achieves exceptional reaction selectivity and facilitates product isolation through simple precipitation. For R&D directors and procurement managers alike, this represents a paradigm shift towards more cost-effective and environmentally compliant manufacturing strategies for essential cardiovascular drug intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of dihydropyridine derivatives has relied heavily on traditional organic solvents such as methanol or ethanol, often in conjunction with acidic or basic catalysts. While these methods are well-established, they suffer from significant drawbacks that hinder efficient large-scale production. For instance, when methanol is employed as the solvent for synthesizing Nifedipine, the reaction yield is notoriously low, often hovering around 56.6%, primarily due to poor reaction selectivity and the formation of viscous byproducts that complicate downstream processing. Furthermore, the use of volatile organic solvents necessitates complex recovery systems to meet environmental regulations, adding substantial operational expenditure. Alternative approaches using ionic liquids, such as 1-butyl-3-methylimidazolium tetrafluoroborate, have shown improved yields up to 85.7%, but they introduce new challenges regarding high raw material costs and the difficulty of recycling the solvent, which often degrades or becomes contaminated, rendering the process economically unviable for bulk manufacturing.
The Novel Approach
The innovative process outlined in the patent data introduces a robust alternative by employing Deep Eutectic Solvents (DES) composed of choline chloride mixed with either ethylene glycol or glycerol. This approach fundamentally alters the reaction landscape by providing a medium that not only dissolves the reactants effectively but also actively participates in the catalytic cycle through hydrogen bonding interactions. Unlike traditional ionic liquids, these DES systems are inexpensive to prepare, chemically stable under reaction conditions, and possess low viscosity, which enhances mass transfer rates significantly. The most transformative aspect of this methodology is the solubility profile of the product; as the dihydropyridine compound forms, it precipitates out of the solution, driving the equilibrium forward and allowing for isolation via simple filtration. This eliminates the need for complex extraction or distillation steps, thereby simplifying the workflow and reducing the generation of hazardous waste streams associated with solvent disposal.
Mechanistic Insights into DES-Catalyzed Hantzsch Condensation
The efficacy of this synthesis relies on the unique physicochemical properties of the Deep Eutectic Solvent, which acts as a dual-function medium serving as both the solvent and the catalyst. The mechanism involves the formation of a extensive hydrogen bond network between the hydrogen bond donor (glycerol or ethylene glycol) and the hydrogen bond acceptor (choline chloride). This network increases the protonation degree of the reaction intermediates, effectively activating the carbonyl groups of the ethyl acetoacetate and the aldehyde towards nucleophilic attack by the ammonia source. The stabilization of transition states within this polar environment lowers the activation energy required for the multicomponent condensation, allowing the reaction to proceed efficiently at moderate temperatures ranging from 40°C to 100°C. This mild thermal requirement is crucial for preventing the decomposition of sensitive functional groups on substituted benzaldehydes, ensuring high fidelity in the final product structure.
Furthermore, the mechanism incorporates an inherent purification step driven by thermodynamics. As the dihydropyridine ring closes and the molecular weight increases, the solubility of the product in the polar DES medium decreases sharply. This phenomenon causes the product to crystallize or precipitate out of the reaction mixture as it forms, effectively removing it from the reactive environment. This continuous removal prevents reverse reactions and minimizes side reactions that typically occur when products remain in solution for extended periods. Consequently, the solid cake obtained after filtration exhibits high purity, often exceeding 99% without the need for recrystallization, while the unreacted starting materials remain dissolved in the mother liquor, ready to be recycled in subsequent batches, thus creating a closed-loop system that maximizes atom economy.
How to Synthesize Dihydropyridine Compounds Efficiently
To implement this green synthesis route effectively, manufacturers must adhere to specific stoichiometric ratios and process parameters that optimize both yield and solvent longevity. The standard protocol involves dissolving benzaldehyde derivatives, ethyl acetoacetate, and ammonium acetate in the DES medium at a molar ratio of approximately 1:2.2:1.2, ensuring an excess of the beta-keto ester to drive the condensation to completion. The reaction mixture is then heated to a controlled temperature, typically around 70°C, and stirred for a period of 3 to 20 hours depending on the electronic nature of the substituents on the aromatic ring. Following the reaction, the addition of water or ethanol can further induce precipitation, after which the solid product is collected by filtration and dried under vacuum. The detailed standardized synthesis steps see the guide below.
- Dissolve benzaldehyde derivatives, ethyl acetoacetate, and ammonium acetate in a Deep Eutectic Solvent (DES) composed of choline chloride and glycerol or ethylene glycol.
- Heat the reaction mixture to a temperature between 40°C and 100°C and maintain stirring for a duration of 3 to 20 hours to facilitate condensation.
- Upon completion, filter the resulting precipitate to isolate the high-purity product, then recover ethanol from the filtrate to recycle the DES for subsequent batches.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this DES-based technology offers compelling economic and logistical benefits that directly impact the bottom line. The primary advantage lies in the drastic simplification of the post-reaction workup; by replacing energy-intensive distillation and chromatography with simple filtration, the process significantly reduces utility consumption and labor hours. This streamlining of operations translates into a leaner manufacturing footprint, allowing facilities to increase throughput without proportional increases in capital expenditure on separation equipment. Moreover, the elimination of volatile organic solvents reduces the regulatory burden associated with VOC emissions and hazardous waste disposal, lowering compliance costs and mitigating environmental risks.
- Cost Reduction in Manufacturing: The economic model of this process is strengthened by the recyclability of the reaction medium. Unlike single-use organic solvents or expensive ionic liquids that degrade after one cycle, the DES system described can be reused for 10 to 15 cycles without a significant decrease in catalytic performance or yield. This longevity drastically reduces the recurring cost of raw materials per kilogram of product. Additionally, the high conversion rates achieved from the second cycle onwards minimize the loss of valuable starting materials, ensuring that the cost of goods sold remains competitive even for complex substituted derivatives.
- Enhanced Supply Chain Reliability: The robustness of the DES solvent system contributes to greater supply chain stability. Because the solvent components (choline chloride, glycerol, ethylene glycol) are commodity chemicals with abundant global availability, the risk of supply disruption is minimal compared to specialized ionic liquids or exotic catalysts. The simplicity of the process also reduces the dependency on highly skilled operators for complex purification tasks, making it easier to scale production across different manufacturing sites without compromising quality consistency.
- Scalability and Environmental Compliance: From a sustainability perspective, this process aligns perfectly with modern green chemistry principles, which is increasingly a prerequisite for supplying major pharmaceutical companies. The theoretical absence of waste generation, combined with the ability to recycle the solvent matrix, positions this technology as a leader in eco-friendly manufacturing. This environmental stewardship not only future-proofs the supply against tightening regulations but also enhances the brand value of the final API by associating it with a sustainable production lifecycle.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this DES-based synthesis technology. These answers are derived directly from the experimental data and beneficial effects reported in the patent literature, providing a clear understanding of the process capabilities and limitations for potential partners.
Q: What are the advantages of using Deep Eutectic Solvents (DES) over traditional ionic liquids for dihydropyridine synthesis?
A: Unlike traditional ionic liquids which can be expensive and difficult to recycle, the DES system described in patent CN114213318A utilizes low-cost components like choline chloride and glycerol. This system offers superior chemical stability, lower viscosity for better mass transfer, and allows for the solvent to be reused up to 15 times without significant loss in catalytic activity or yield.
Q: How does this process ensure high product purity without complex purification steps?
A: The process leverages the unique solubility properties of the reaction medium. As the dihydropyridine product forms, it precipitates out of the DES solution due to low solubility, while unreacted starting materials and byproducts remain dissolved. This allows for the isolation of high-purity solids (>99%) through simple filtration, eliminating the need for energy-intensive distillation or chromatography.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the method is specifically designed for industrial scalability. It operates at moderate temperatures (40-100°C), avoids the generation of theoretical waste, and simplifies post-treatment to basic filtration. The ability to recycle the solvent system multiple times significantly reduces raw material costs and waste disposal burdens, making it highly viable for commercial scale-up.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Dihydropyridine Supplier
At NINGBO INNO PHARMCHEM, we recognize the transformative potential of green synthesis technologies like the DES-mediated route for dihydropyridines in reshaping the pharmaceutical supply chain. As a dedicated CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative laboratory processes are seamlessly translated into robust industrial operations. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch meets the highest international standards, guaranteeing that your downstream synthesis remains uninterrupted by impurity profiles.
We invite you to collaborate with us to leverage this advanced technology for your next project. Our technical team is prepared to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements, demonstrating how this green route can optimize your budget. Please contact our technical procurement team today to request specific COA data and route feasibility assessments, and let us help you secure a sustainable and cost-effective supply of high-purity dihydropyridine intermediates.
