Scalable Synthesis of Imidazopyridine Derivatives for Advanced Antihypertensive Drug Manufacturing
Scalable Synthesis of Imidazopyridine Derivatives for Advanced Antihypertensive Drug Manufacturing
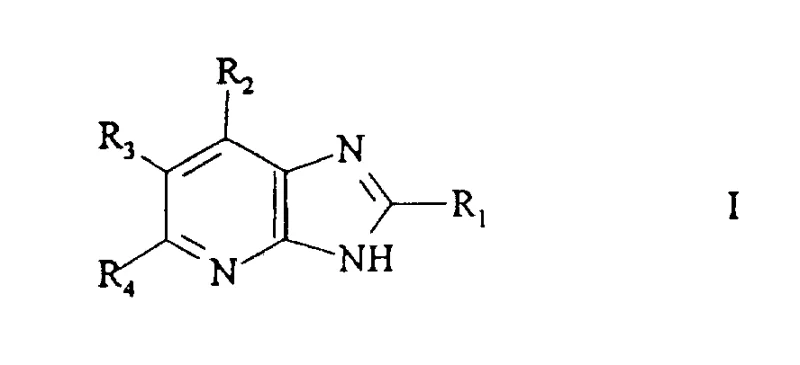
The development of efficient synthetic routes for heterocyclic compounds remains a cornerstone of modern pharmaceutical process chemistry, particularly for cardiovascular therapeutics. Patent CN110609A discloses a robust and versatile methodology for the preparation of imidazopyridine derivatives, which serve as critical intermediates in the synthesis of Angiotensin-II receptor antagonists. These compounds are pivotal in the management of hypertension and related cardiovascular disorders. The disclosed technology addresses significant bottlenecks associated with traditional synthetic pathways, specifically the challenges inherent in direct nitration strategies. By leveraging a condensation-cyclization strategy involving imidic acid alkyl esters and 1,3-dicarbonyl compounds, this process offers a streamlined alternative that enhances both chemical efficiency and operational safety. For global supply chain stakeholders, understanding the nuances of this chemistry is essential for securing reliable sources of high-value API intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of substituted imidazopyridines has relied heavily on the functionalization of pre-existing pyridine rings. A common retrospective approach involves the nitration of aminopyridines to introduce nitrogen functionality at specific positions, followed by reduction and cyclization. However, as noted in the background art of the patent, the direct nitration of corresponding aminopyridines is fraught with difficulties. Regioselectivity is often poor, leading to complex mixtures of isomers that are difficult and costly to separate. Furthermore, the harsh conditions required for nitration, typically involving strong mixed acids, pose significant safety hazards and environmental burdens due to the generation of acidic waste streams. The subsequent reduction steps often require stoichiometric amounts of reducing agents or precious metal catalysts, further inflating the cost of goods sold (COGS) and complicating the purification profile. These factors collectively render conventional nitration-based routes less attractive for large-scale commercial manufacturing where purity and cost-efficiency are paramount.
The Novel Approach
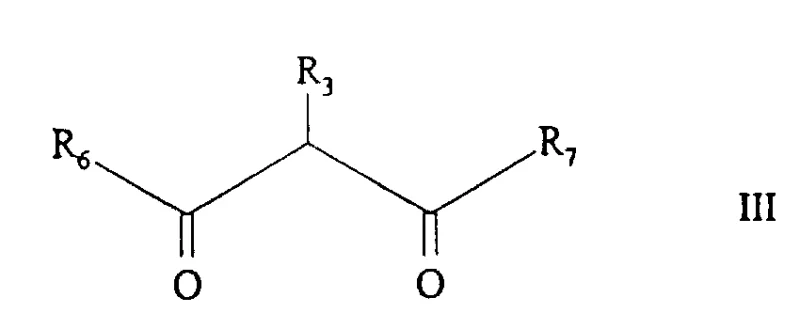
In stark contrast to the destructive functionalization of existing rings, the method described in CN110609A employs a constructive, building-block approach. The core innovation lies in the cyclization reaction of an imidic acid alkyl ester (Formula II) with aminoacetonitrile and a 1,3-dicarbonyl compound (Formula III). This strategy allows for the assembly of the imidazopyridine skeleton from simple, commercially available starting materials such as nitriles, alcohols, and beta-diketones. The reaction proceeds under relatively mild thermal conditions, typically ranging from 50°C to the reflux temperature of the solvent, avoiding the extreme temperatures and pressures often seen in older methodologies. By utilizing aminoacetonitrile as a two-carbon synthon containing the necessary nitrogen atoms, the process effectively bypasses the need for difficult nitration steps entirely. This not only simplifies the synthetic sequence but also dramatically improves the overall atom economy and scalability of the operation.
Mechanistic Insights into Imidate-Mediated Cyclization
The mechanistic pathway of this transformation is a fascinating example of nucleophilic attack and subsequent intramolecular cyclization. The process initiates with the formation of the imidic acid alkyl ester (Formula II), generated in situ or separately by reacting a nitrile with an alcohol in the presence of a hydrogen halide, typically dry HCl gas. This activation of the nitrile carbon makes it highly electrophilic. In the subsequent step, aminoacetonitrile, acting as a bidentate nucleophile, attacks the activated imidate carbon. Simultaneously or sequentially, the enol form of the 1,3-dicarbonyl compound participates in the condensation. The presence of a base, such as sodium hydroxide, sodium ethylate, or triethylamine, is crucial to deprotonate the aminoacetonitrile salt and facilitate the nucleophilic attack. The reaction medium can be quite flexible; while the 1,3-dicarbonyl compound can sometimes act as the solvent itself, aromatic hydrocarbons like toluene are frequently employed to allow for higher reaction temperatures and easier water removal via azeotropic distillation. This drives the equilibrium towards the formation of the fused heterocyclic ring system.
From an impurity control perspective, this mechanism offers distinct advantages over radical-based or electrophilic aromatic substitution routes. Because the ring is built from discrete fragments, the potential for random poly-substitution on an aromatic ring is eliminated. The primary impurities likely arise from incomplete conversion of the starting materials or hydrolysis of the imidate intermediate. However, the patent data indicates that standard workup procedures involving pH adjustment and organic extraction are highly effective at removing these byproducts. For instance, adjusting the aqueous phase to acidic pH (e.g., pH 1.3) allows for the separation of basic impurities, while subsequent basification (pH 8.4) facilitates the extraction of the neutral product into organic solvents like ethyl acetate. This level of control over the purification profile is critical for meeting the stringent purity specifications required for pharmaceutical intermediates, ensuring that downstream coupling reactions proceed without interference from trace contaminants.
How to Synthesize Imidazopyridine Derivatives Efficiently
Implementing this synthesis at a commercial scale requires careful attention to the preparation of the imidate intermediate and the control of the cyclization exotherm. The patent provides detailed embodiments, such as the preparation of 2-ethyl-5,7-dimethyl-3H-imidazo(4,5-b)pyridine, which serves as an excellent template for process optimization. The initial step involves bubbling HCl gas into a mixture of propionitrile and methanol at 0°C, a procedure that requires corrosion-resistant equipment but yields the imidate hydrochloride in high efficiency (reported up to 93%). The subsequent cyclization involves heating the mixture of imidate, aminoacetonitrile, and diketone in toluene, gradually raising the temperature to remove methanol and water before refluxing. Detailed standardized synthetic steps see the guide below.
- Preparation of Imidic Acid Alkyl Ester: React a nitrile (e.g., propionitrile) with an alcohol (e.g., methanol) in the presence of hydrogen halide (HCl gas) at low temperature (0°C) to form the imidate hydrochloride salt.
- Cyclization Reaction: Combine the imidic acid alkyl ester with aminoacetonitrile (or its salt) and a 1,3-dicarbonyl compound (e.g., methyl ethyl diketone) in a solvent like toluene.
- Workup and Purification: Heat the mixture to reflux (50-130°C), neutralize with base, extract with organic solvents (ethyl acetate), and purify via recrystallization or chromatography.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the shift from nitration-based routes to this condensation methodology represents a significant opportunity for risk mitigation and cost optimization. The reliance on commodity chemicals rather than specialized, hazard-classified reagents fundamentally alters the supply risk profile. Traditional nitration requires strict controls on nitric and sulfuric acids, which are subject to fluctuating regulatory landscapes and transportation restrictions. In contrast, the reagents for this process—nitriles, simple alcohols, and beta-diketones—are produced in massive volumes for various industrial applications, ensuring a stable and competitive supply market. This abundance translates directly into better pricing power and reduced vulnerability to supply shocks. Furthermore, the simplified purification workflow reduces the consumption of auxiliary materials such as chromatography media or exotic solvents, contributing to a leaner manufacturing cost structure.
- Cost Reduction in Manufacturing: The elimination of the nitration and subsequent reduction steps removes the need for expensive catalysts and rigorous safety infrastructure associated with energetic materials. By utilizing a convergent synthesis where the ring is constructed from cheaper building blocks, the overall cost of goods is significantly lowered. The process avoids the use of precious metals often required for hydrogenation steps in alternative routes, replacing them with inexpensive inorganic bases like sodium hydroxide or alkoxides. Additionally, the high yields reported in the examples suggest that raw material utilization is efficient, minimizing waste disposal costs and maximizing output per batch.
- Enhanced Supply Chain Reliability: The raw materials identified in this patent, such as propionitrile, methyl ethyl diketone, and aminoacetonitrile, are widely available from multiple global suppliers. This multi-sourcing capability is vital for maintaining business continuity and preventing production stoppages due to single-source failures. The robustness of the reaction conditions, which tolerate a variety of solvents including toluene and alcohols, further enhances flexibility. If a specific solvent faces supply constraints, the process can often be adapted to use alternatives without compromising yield, providing a buffer against logistical disruptions in the chemical supply chain.
- Scalability and Environmental Compliance: From an environmental, health, and safety (EHS) perspective, this route is markedly superior. It avoids the generation of large volumes of acidic wastewater typical of nitration processes. The workup involves standard liquid-liquid extractions and crystallizations, which are easily scalable from kilogram to multi-ton quantities. The ability to perform the reaction in solvents like toluene, which can be recovered and recycled efficiently, aligns with green chemistry principles and helps facilities meet increasingly strict environmental discharge regulations. This ease of scale-up ensures that the transition from pilot plant to commercial production is smooth, reducing the time-to-market for new drug formulations.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this imidazopyridine synthesis technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing a factual basis for process evaluation. Understanding these details is crucial for technical teams assessing the feasibility of adopting this route for their specific API manufacturing needs.
Q: What are the key advantages of this imidazopyridine synthesis route over traditional nitration methods?
A: The primary advantage is the avoidance of difficult regioselective nitration reactions on aminopyridine rings, which are often low-yielding or impossible. This patent describes a constructive approach building the ring system from simple nitriles and 1,3-dicarbonyls, significantly improving feasibility.
Q: What types of solvents are suitable for the cyclization step?
A: The process is robust regarding solvent choice. Suitable options include lower aliphatic alcohols (methanol, ethanol), halohydrocarbons (methylene dichloride), ethers (dioxane), and aromatic hydrocarbons (toluene, xylene). Toluene is often preferred for higher temperature reflux.
Q: Can this method be adapted for different substituents on the imidazopyridine ring?
A: Yes, the method is highly versatile. By varying the starting nitrile (for R1), the 1,3-dicarbonyl compound (for R3, R6, R7), and reaction conditions, a wide range of derivatives including alkyl, cycloalkyl, aryl, and heterocyclic substitutions can be achieved.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Imidazopyridine Derivative Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of robust intermediate synthesis in the pharmaceutical value chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We are committed to delivering high-purity imidazopyridine derivatives that meet stringent purity specifications, supported by our rigorous QC labs equipped with state-of-the-art analytical instrumentation. Our facility is designed to handle the specific solvent systems and thermal profiles required by this condensation chemistry safely and efficiently.
We invite potential partners to engage with our technical procurement team to discuss how this patented technology can be integrated into your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic benefits specific to your volume requirements. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project timelines, ensuring a partnership built on transparency and technical excellence.
