Advanced One-Pot Synthesis of Arbidol Hydrochloride for Commercial Scale-Up
The global demand for effective antiviral agents has placed significant pressure on pharmaceutical supply chains to deliver high-quality active pharmaceutical ingredients with consistent reliability and economic efficiency. In response to these industry challenges, recent intellectual property developments, specifically patent CN115232055A, have introduced a transformative synthesis method for Arbidol Hydrochloride that addresses critical bottlenecks in traditional manufacturing. This technical breakthrough shifts the paradigm from multi-step, solvent-intensive procedures to a streamlined one-pot preparation method that utilizes a water-ethanol mixed solvent system. By leveraging solid raw materials such as paraformaldehyde and dimethylamine hydrochloride instead of their volatile liquid counterparts, the process not only enhances operational safety but also drastically simplifies post-treatment workflows. For R&D directors and procurement specialists evaluating long-term sourcing strategies, understanding the mechanistic advantages of this green chemistry approach is essential for securing a competitive edge in the antiviral market. This report analyzes the technical feasibility and commercial implications of adopting this novel route for the commercial scale-up of complex pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional manufacturing routes for Arbidol Hydrochloride have long been plagued by inherent safety hazards and inefficient resource utilization that drive up operational expenditures. The legacy process relies heavily on formalin solutions, which are classified as carcinogenic, and aqueous dimethylamine, both of which are highly volatile and prone to leakage, posing severe risks to personnel health and environmental compliance. Furthermore, the conventional method employs glacial acetic acid as both a solvent and an acid catalyst, necessitating a large volume of this corrosive chemical throughout the production cycle. Following the reaction, the process requires a complex alkalization step using significant amounts of alkali to neutralize the reaction liquid and adjust the pH to form the free alkali base. This is followed by extraction using dichloromethane and ethyl acetate, distillation, and subsequent crystallization with acetone, creating a massive burden on waste treatment facilities and increasing the overall carbon footprint of the manufacturing site.
The Novel Approach
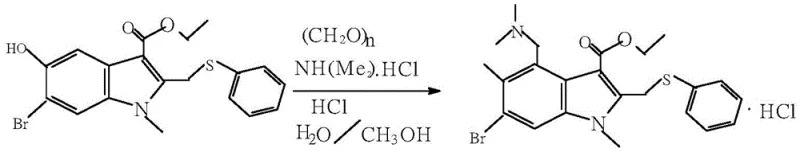
In stark contrast to the cumbersome legacy workflows, the novel approach detailed in the patent data introduces a unified one-pot preparation method that fundamentally restructures the synthesis logic for improved efficiency. By replacing hazardous liquid reagents with solid paraformaldehyde and dimethylamine hydrochloride, the new protocol eliminates the risk of volatile gas leakage at the source, thereby creating a safer working environment for plant operators. The substitution of glacial acetic acid with a water-ethanol mixed solvent system not only reduces the consumption of aggressive organic chemicals but also enables the direct cooling and crystallization of the product after the Mannich reaction is complete. This innovation removes the need for multiple extraction, distillation, and refining steps that characterize the old process, effectively collapsing a three-step operation into a single continuous workflow. Consequently, the discharge of three wastes is greatly reduced, and the potential safety hazards associated with handling large volumes of flammable and toxic solvents are effectively eliminated from the production line.
Mechanistic Insights into One-Pot Mannich Reaction
The core of this technological advancement lies in the precise optimization of the Mannich reaction conditions within a hydro-alcoholic medium, which facilitates a cleaner transformation of the indole precursor. The process utilizes concentrated hydrochloric acid with a mass fraction greater than 30% as a highly active acid catalyst, which promotes the generation of the reactive iminium ion intermediate from the solid amine and formaldehyde sources. Maintaining the reaction temperature initially at 35-40°C for 3 hours allows for controlled initiation of the aminomethylation, preventing runaway exotherms that could lead to byproduct formation. Subsequently, slowly heating the mixture to 65-70°C for 6 hours ensures complete conversion of the starting material, driving the equilibrium towards the desired Arbidol structure without degrading the sensitive indole ring system. This thermal profile is critical for maximizing yield while minimizing the formation of polymeric impurities often associated with uncontrolled formaldehyde reactions in acidic media.
Impurity control in this new synthesis route is achieved through the strategic selection of the crystallization solvent system, which inherently excludes many non-polar side products. Unlike the old process where the product exists as an acetate salt requiring neutralization and extraction, the new method directly yields the hydrochloride salt upon cooling the reaction mixture to 0-5°C. The use of a 1:1 mass ratio of water to ethanol creates a solvent environment where the target Arbidol Hydrochloride has low solubility at low temperatures, promoting the precipitation of high-purity crystals while leaving soluble impurities in the mother liquor. This direct crystallization mechanism bypasses the need for chromatographic purification or repeated recrystallization steps, ensuring that the final technical index reaches a purity of 99.5% or higher. The elimination of dichloromethane and ethyl acetate extraction also prevents the introduction of halogenated solvent residues, further enhancing the safety profile of the final API for pharmaceutical formulation.
How to Synthesize Arbidol Hydrochloride Efficiently
Implementing this synthesis route requires strict adherence to the specified feeding ratios and thermal controls to replicate the high yields reported in the patent literature. The process begins with the simultaneous charging of water, ethanol, dimethylamine hydrochloride, paraformaldehyde, concentrated hydrochloric acid, and the indole ester precursor into a standard reaction kettle equipped with efficient stirring and temperature control systems. Once the solids are fully dissolved and the mixture is homogenous, the temperature must be carefully managed to ensure the reaction proceeds through the intended kinetic pathway without generating thermal hotspots. Detailed standardized synthesis steps see the guide below.
- Charge the reactor with water, ethanol, dimethylamine hydrochloride, paraformaldehyde, concentrated hydrochloric acid, and the indole precursor.
- Control temperature at 35-40°C for 3 hours, then heat to 65-70°C for 6 hours to complete the Mannich reaction.
- Cool the mixture to 0-5°C for crystallization, centrifuge, wash with 50% ethanol, and dry to obtain the final product.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this novel synthesis method offers tangible economic benefits that extend beyond simple raw material cost savings. The patent data explicitly highlights a reduction in production costs from approximately 2000 yuan per kilogram to about 1400 yuan per kilogram, representing a substantial margin improvement that can be leveraged in contract negotiations. This cost reduction in pharmaceutical intermediates manufacturing is driven primarily by the elimination of expensive organic solvents like glacial acetic acid and acetone, as well as the reduction in energy consumption associated with distillation and drying operations. Furthermore, the simplification of the workflow reduces the labor hours required per batch, allowing facilities to increase throughput without expanding physical infrastructure or headcount. These factors combine to create a more resilient supply chain capable of absorbing market fluctuations in raw material pricing while maintaining competitive delivery terms for downstream partners.
- Cost Reduction in Manufacturing: The economic advantage of this process is anchored in the drastic simplification of the material bill and the removal of costly unit operations. By avoiding the use of glacial acetic acid, ethyl acetate, and acetone, the facility saves significantly on solvent procurement and recovery costs, which are often volatile components of the operating budget. Additionally, the yield improvement from 75% to 90% means that less raw material is wasted per kilogram of finished product, directly lowering the cost of goods sold. The elimination of complex post-treatment steps such as alkalization and multiple extractions further reduces utility consumption and waste disposal fees, contributing to a leaner and more profitable production model that aligns with modern cost-reduction strategies.
- Enhanced Supply Chain Reliability: Operational reliability is significantly bolstered by the shorter production cycle, which the patent notes is reduced from 4 days to just 2 days. This acceleration in manufacturing velocity allows for faster turnover of inventory and more responsive fulfillment of purchase orders, reducing the lead time for high-purity pharmaceutical intermediates. The use of stable solid reagents like paraformaldehyde and dimethylamine hydrochloride also mitigates the supply risks associated with transporting and storing hazardous volatile liquids, ensuring that production schedules are not disrupted by regulatory delays or safety incidents. Consequently, suppliers adopting this technology can offer more consistent delivery windows and maintain higher safety stock levels without incurring excessive holding costs or safety liabilities.
- Scalability and Environmental Compliance: From a sustainability perspective, the process achieves zero discharge of organic solvents, as the water-ethanol mixture can be recycled and adjusted for repeated use in subsequent batches. This closed-loop solvent management system greatly reduces the volume of hazardous waste requiring treatment, ensuring compliance with increasingly stringent environmental regulations regarding VOC emissions and wastewater quality. The inherent safety of the one-pot design, which eliminates high-risk extraction and distillation steps, makes the process easier to scale from pilot plants to 100 MT annual commercial production without requiring exponential increases in safety engineering controls. This scalability ensures that the supply chain can grow to meet global demand surges while maintaining a minimal environmental footprint.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this green synthesis technology for Arbidol Hydrochloride. These insights are derived directly from the comparative data provided in the patent documentation, focusing on the practical implications for industrial adoption. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this route into their existing manufacturing portfolios.
Q: How does the new process improve safety compared to traditional methods?
A: The new process replaces carcinogenic formalin solution and volatile dimethylamine aqueous solution with solid paraformaldehyde and dimethylamine hydrochloride, significantly reducing leakage risks and occupational health hazards.
Q: What are the yield and purity improvements in this synthesis method?
A: According to patent data, the yield increases from approximately 75% to 90%, and product purity improves from 99% to over 99.5%, reducing the need for extensive refining.
Q: Does this method reduce environmental waste discharge?
A: Yes, by eliminating glacial acetic acid, ethyl acetate, and acetone, and utilizing a recyclable water-ethanol system, the process achieves zero organic solvent discharge and greatly reduces three-waste emission.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arbidol Hydrochloride Supplier
As the pharmaceutical industry continues to prioritize green chemistry and cost-efficient manufacturing, partnering with a technically proficient CDMO is vital for securing a stable supply of critical antiviral ingredients. NINGBO INNO PHARMCHEM possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovative processes like the one described in CN115232055A can be successfully translated from the laboratory to the factory floor. Our commitment to quality is underpinned by stringent purity specifications and rigorous QC labs that verify every batch against the highest international standards, guaranteeing that the Arbidol Hydrochloride we supply meets the exacting requirements of global regulatory bodies. We understand that consistency is key in API manufacturing, and our advanced facilities are designed to handle complex chemistries with precision and safety.
We invite forward-thinking pharmaceutical companies to collaborate with us to leverage these technological advancements for their product pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements and quality targets. We encourage potential partners to contact us directly to obtain specific COA data and route feasibility assessments that demonstrate how our optimized synthesis methods can enhance your supply chain resilience. Let us work together to bring safer, more affordable, and high-quality antiviral medications to the global market through superior chemical engineering and dedicated service.
