Optimizing Arbidol Hydrochloride Production: A Technical Breakthrough for Scalable API Manufacturing
Optimizing Arbidol Hydrochloride Production: A Technical Breakthrough for Scalable API Manufacturing
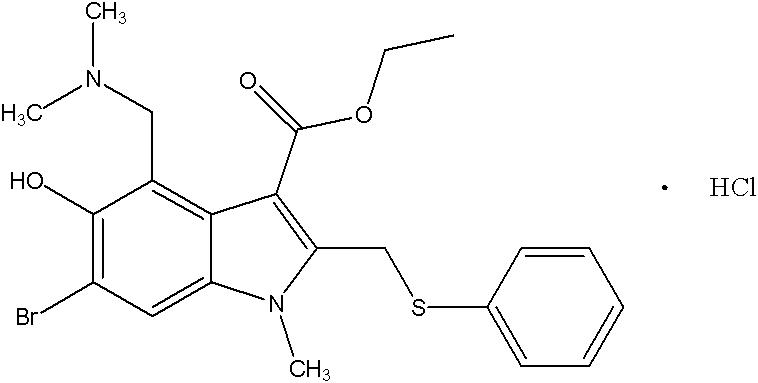
The pharmaceutical industry continuously seeks robust synthetic pathways that balance high purity with economic viability, particularly for antiviral agents like Arbidol Hydrochloride. Patent CN102351778A introduces a transformative preparation method that addresses the longstanding inefficiencies of traditional indole synthesis. This technical insight report analyzes the novel route disclosed in the patent, which utilizes 3-iodo-4-nitrophenol as a key starting material to achieve an overall reaction yield exceeding 30%. By replacing the conventional Nenitzescu reaction with a more efficient iron-mediated cyclization, this methodology offers a compelling value proposition for a reliable API intermediate supplier seeking to optimize their manufacturing portfolio. The process is characterized by mild reaction conditions, typically maintaining temperatures below 100°C, and eliminates the need for hazardous high-temperature and high-pressure operations, marking a significant advancement in process safety and operational simplicity.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Arbidol Hydrochloride has relied heavily on the Nenitzescu reaction, a classic method for constructing the indole ring system. However, extensive industrial data indicates that this approach suffers from inherent limitations that hinder large-scale commercial viability. As detailed in prior art such as Chinese patent CN1687033A, the Nenitzescu reaction typically yields only about 60% at the cyclization step, dragging the total overall yield down to approximately 22.9%. Furthermore, alternative routes described in literature, such as those utilizing 5-hydroxy-1,2-dimethyl indole-3-ethyl formate, face severe supply chain bottlenecks due to the difficulty in sourcing these specialized precursors. Other variations reported in academic journals have demonstrated even lower efficiencies, with total recovery rates plummeting to merely 11.2% due to poor selectivity and complex purification requirements. These inefficiencies not only inflate the cost of goods sold but also introduce significant variability in batch-to-batch consistency, posing a risk to the supply continuity required by global pharmaceutical manufacturers.
The Novel Approach
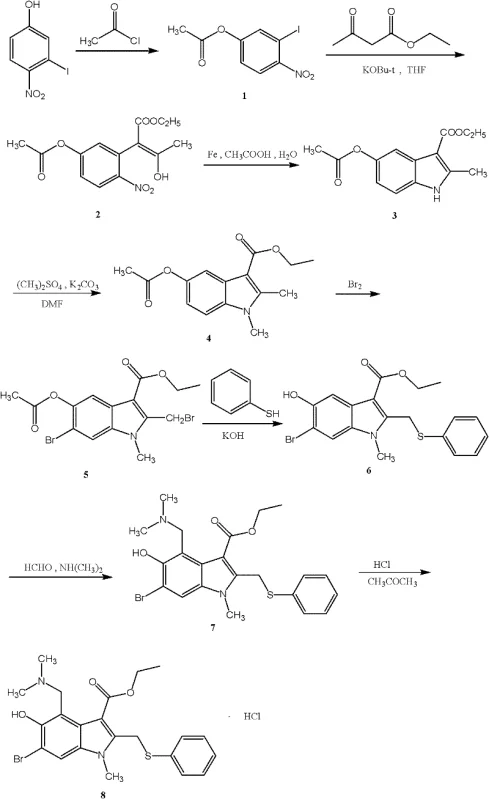
In stark contrast to these legacy methods, the invention disclosed in CN102351778A presents a streamlined eight-step synthesis that fundamentally re-engineers the construction of the indole core. The process initiates with the acetylation of 3-iodo-4-nitrophenol, followed by a substitution reaction with ethyl acetoacetate under alkaline conditions. The pivotal innovation lies in the subsequent reduction-condensation step, where iron powder and acetic acid facilitate the cyclization to form the indole ring with exceptional efficiency. This specific sequence allows for an indole synthesis yield surpassing 90%, contributing to a robust overall yield of greater than 30%. The reaction pathway avoids the use of expensive transition metal catalysts often required in cross-coupling reactions, instead leveraging inexpensive and abundant iron reagents. This strategic shift not only simplifies the reaction workup but also aligns perfectly with the goals of cost reduction in pharmaceutical manufacturing by minimizing raw material waste and energy consumption.
Mechanistic Insights into Iron-Mediated Cyclization
The core of this technological advancement is the mechanistic elegance of the iron-mediated reduction-condensation concerted reaction. In this step, the nitro group of the intermediate is reduced to an amino group in situ by iron powder in an acetic acid-water solvent system. This newly formed amine immediately undergoes a nucleophilic attack on the carbonyl carbon of the adjacent side chain, followed by dehydration to close the five-membered indole ring. The use of iron powder is particularly advantageous from a process chemistry perspective because it acts as a heterogeneous reagent that is easily removed by filtration, eliminating the need for complex chromatographic purification to remove metal residues. The reaction conditions are remarkably gentle, proceeding effectively at temperatures around 80°C, which prevents the thermal degradation of sensitive functional groups such as the ester moiety. This high level of chemoselectivity ensures that the bromine atom, introduced in later steps or present in the scaffold, remains intact until the specific halogenation stage, thereby preserving the structural integrity required for subsequent functionalization.
Furthermore, the control of impurities is inherently superior in this route due to the high regioselectivity of the cyclization. Traditional methods often generate isomeric byproducts that are structurally similar to the target molecule, making them notoriously difficult to separate. The specific electronic environment created by the iodine and acetoxy groups in the precursor directs the cyclization exclusively to the desired position. Following the ring closure, the N-methylation using dimethyl sulfate and the subsequent dibromination with bromine in carbon tetrachloride proceed with high fidelity. The final Mannich reaction, introducing the dimethylaminomethyl group, is conducted in aqueous dimethylamine and formaldehyde, ensuring complete conversion without over-alkylation. This precise control over the reaction trajectory results in a crude product of high purity, which can be further refined through simple recrystallization techniques using solvent systems like acetone-ethanol, ensuring the final Arbidol Hydrochloride meets stringent pharmaceutical standards.
How to Synthesize Arbidol Hydrochloride Efficiently
The synthesis of Arbidol Hydrochloride via this patented route involves a logical sequence of protection, cyclization, and functionalization steps designed for maximum throughput. The process begins with the protection of the phenolic hydroxyl group, followed by the critical iron-mediated ring closure that defines the efficiency of the entire pathway. Operators must pay close attention to the stoichiometry of the iron powder and the temperature profile during the cyclization to ensure optimal yield. The subsequent steps involving methylation and bromination require careful handling of reagents but benefit from the robustness of the intermediate scaffold. For a detailed breakdown of the specific reaction parameters, solvent choices, and workup procedures, please refer to the standardized synthesis guide below which outlines the critical process parameters derived from the patent embodiments.
- Perform hydroxyl protection on 3-iodo-4-nitrophenol using acetyl chloride and a base catalyst to obtain Compound I.
- Execute a substitution reaction with ethyl acetoacetate under alkaline conditions to form Compound II.
- Conduct a reduction-condensation concerted reaction using iron powder and acetic acid to synthesize the indole ring (Compound III).
- Proceed with N-methylation, dibromination, thiophenol substitution, and Mannich reaction to finalize the structure before acidification.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthetic route offers tangible strategic benefits that extend beyond mere chemical yield. The shift away from the Nenitzescu reaction eliminates the dependency on scarce and expensive quinone-based starting materials, replacing them with commodity chemicals like 3-iodo-4-nitrophenol and ethyl acetoacetate. This substitution drastically simplifies the sourcing strategy, reducing the risk of supply disruptions caused by the limited availability of niche precursors. Moreover, the elimination of high-pressure and extreme low-temperature steps reduces the capital expenditure required for specialized reactor equipment, allowing for production in standard glass-lined or stainless steel vessels commonly found in multipurpose chemical plants. This flexibility enhances the agility of the supply chain, enabling faster scale-up from pilot batches to commercial tonnage without the need for significant infrastructure investment.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the substantial increase in overall yield and the utilization of low-cost reagents. By achieving an overall yield of over 30%, compared to the sub-20% yields of legacy methods, the amount of raw material required per kilogram of final product is significantly decreased. Additionally, the use of iron powder as a reducing agent is far more economical than the palladium or rhodium catalysts often employed in modern cross-coupling alternatives. The simplified purification process, which relies on crystallization rather than column chromatography, further reduces solvent consumption and waste disposal costs. These factors combine to create a leaner cost structure, allowing for competitive pricing in the global market while maintaining healthy margins for the manufacturer.
- Enhanced Supply Chain Reliability: The reliance on widely available bulk chemicals ensures a stable and continuous supply of raw materials. 3-iodo-4-nitrophenol and ethyl acetoacetate are produced by numerous chemical suppliers globally, mitigating the risk of single-source dependency. The robustness of the reaction conditions means that production is less susceptible to minor fluctuations in utility supplies, such as cooling water temperature or steam pressure, which can often halt more sensitive processes. This reliability translates into shorter lead times for high-purity pharmaceutical intermediates, as production schedules can be maintained with greater predictability. For downstream pharmaceutical companies, this means a more secure supply of the active ingredient, crucial for meeting regulatory commitments and market demand for antiviral medications.
- Scalability and Environmental Compliance: From an environmental and scalability perspective, this route is exceptionally well-suited for industrial expansion. The absence of heavy metal catalysts simplifies the wastewater treatment process, as there is no need for expensive metal scavenging or complex effluent processing to meet strict environmental discharge limits. The reaction solvents, primarily alcohols, acetone, and esters, are readily recoverable and recyclable, supporting green chemistry initiatives. The mild thermal profile of the synthesis allows for safe operation at large scales, minimizing the risk of thermal runaway incidents. This safety margin facilitates the commercial scale-up of complex pharmaceutical intermediates from laboratory benchtop quantities to multi-ton annual production capacities with minimal technical risk, ensuring long-term sustainability for the manufacturing operation.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Arbidol Hydrochloride using this optimized methodology. These answers are derived directly from the experimental data and process descriptions found in the patent literature, providing clarity on the feasibility and advantages of the route. Understanding these details is essential for technical teams evaluating the potential for technology transfer or contract manufacturing partnerships.
Q: What is the primary advantage of the iron-mediated cyclization method for Arbidol HCl?
A: The primary advantage is the significantly improved overall yield, reaching over 30%, compared to traditional Nenitzescu methods which often yield below 20%. Additionally, the reaction conditions are milder, avoiding extreme temperatures and hazardous high-pressure operations.
Q: Are the raw materials for this synthesis route readily available?
A: Yes, the process utilizes 3-iodo-4-nitrophenol as the starting material, which is commercially accessible and easier to source than the specialized indole derivatives required in older synthetic pathways, thereby enhancing supply chain stability.
Q: How does this method impact purification and separation processes?
A: The method simplifies separation and purification due to the high selectivity of the iron-mediated cyclization and the specific recrystallization steps employed, such as using acetone-alcohol-water mixtures, resulting in a high-purity final product suitable for pharmaceutical applications.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Arbidol Hydrochloride Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthesis routes in the modern pharmaceutical landscape. Our team of expert chemists has thoroughly analyzed the methodology presented in CN102351778A and possesses the extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. We are committed to delivering high-purity Arbidol Hydrochloride that adheres to stringent purity specifications, utilizing our rigorous QC labs to ensure every batch meets international regulatory standards. Our facility is equipped to handle the specific unit operations required for this synthesis, including safe handling of bromination reagents and efficient crystallization processes, guaranteeing a consistent supply of this vital antiviral intermediate.
We invite potential partners to engage with our technical procurement team to discuss how this optimized route can benefit your specific supply chain needs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the economic advantages of switching to this manufacturing protocol. We encourage you to contact us to obtain specific COA data for our pilot batches and to discuss route feasibility assessments tailored to your project timelines. Let us collaborate to bring high-quality, cost-effective antiviral solutions to the market efficiently.
