Advanced Manufacturing of Altrenogest: A Technical Breakthrough for Veterinary Pharmaceutical Intermediates
The pharmaceutical landscape for veterinary hormones is constantly evolving, driven by the need for higher purity active ingredients and more sustainable manufacturing processes. Patent CN109232700B, published in mid-2020, introduces a significant methodological advancement in the synthesis of altrenogest, a critical progestogen used extensively in livestock management for estrus synchronization. This technical disclosure moves away from the hazardous and inefficient protocols of the past, offering a streamlined three-step pathway that begins with estra-4,9-diene-3,17-dione. By leveraging modern organometallic techniques, specifically the use of Lithium Diisopropylamide (LDA) for regioselective enolization, the inventors have established a route that not only enhances chemical yield but also drastically improves the impurity profile of the final active pharmaceutical ingredient (API). For industry stakeholders, this represents a pivotal shift towards greener chemistry without compromising the rigorous quality standards required for veterinary drug registration.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of altrenogest has been plagued by significant technical and environmental hurdles that hinder scalable manufacturing. Early methodologies, such as those disclosed in Netherlands patent NL6517141, relied heavily on benzene as a primary solvent for Grignard reactions. Benzene is a known human carcinogen with strict regulatory limits on residual levels in pharmaceutical products, necessitating complex and costly removal processes that often degrade overall yield. Furthermore, alternative routes described in French patent FRM5183 utilized expensive oxidized trenbolone derivatives as starting materials, creating a supply chain bottleneck due to the high cost and limited availability of these precursors. Other existing Chinese patents, like CN106243178A, attempted to mitigate these issues but resulted in multi-step sequences with difficult-to-control parameters, leading to inconsistent product quality and substantial generation of hazardous waste that complicated industrial compliance.
The Novel Approach
The methodology outlined in CN109232700B addresses these legacy issues through a clever redesign of the synthetic logic, focusing on selectivity and reagent availability. Instead of relying on toxic aromatic solvents or scarce starting materials, this novel approach utilizes readily available estra-4,9-diene-3,17-dione and employs a silyl protection strategy that is both robust and scalable. The process replaces the hazardous benzene with anhydrous tetrahydrofuran (THF), a solvent that offers superior solubility for organometallic intermediates while being easier to recover and recycle. By integrating the deprotection and double-bond rearrangement into a single oxidative step using tetrachloro-p-benzoquinone, the total number of unit operations is reduced, which directly correlates to lower energy consumption and reduced equipment footprint in a commercial plant setting.
Mechanistic Insights into LDA-Mediated Enol Silylation and Oxidative Rearrangement
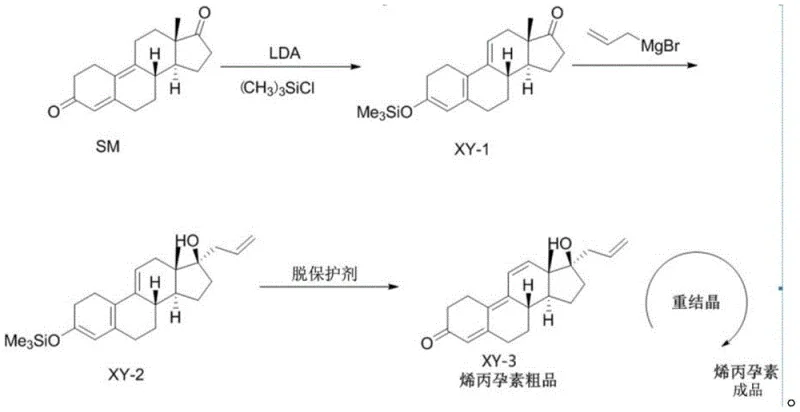
The cornerstone of this synthesis lies in the precise control of stereochemistry and regiochemistry during the initial functionalization of the steroid backbone. In the first step, the use of LDA at cryogenic temperatures (specifically between -40°C and -45°C) ensures the kinetic formation of the lithium enolate at the C3 position, avoiding thermodynamic equilibration that could lead to unwanted isomers. The subsequent addition of trimethylchlorosilane ((CH3)3SiCl) traps this enolate to form the stable enol silyl ether intermediate XY-1. This protection is crucial because it masks the C3 ketone, preventing it from participating in side reactions during the subsequent nucleophilic attack, thereby directing the Grignard reagent exclusively to the C17 position. This level of chemoselectivity is vital for maintaining high purity, as it prevents the formation of bis-addition byproducts that are notoriously difficult to separate from the target molecule.
Following the Grignard addition of allyl magnesium bromide to generate intermediate XY-2, the process employs a sophisticated oxidative deprotection mechanism. The use of tetrachloro-p-benzoquinone serves a dual purpose: it acts as a Lewis acid to facilitate the hydrolysis of the silyl ether and simultaneously functions as an oxidant to drive the migration of the double bond from the 4,9-position to the desired 4,6-diene system characteristic of altrenogest. This tandem transformation is mechanistically elegant, as it converts a potentially unstable intermediate directly into the conjugated dienone system required for biological activity. The byproduct of this reaction, phenol derivatives, are water-soluble and can be easily removed during the aqueous workup, ensuring that the crude product entering the crystallization stage is already of high quality, minimizing the load on the final purification steps.
How to Synthesize Altrenogest Efficiently
Implementing this synthesis requires strict adherence to temperature controls and stoichiometric ratios to maximize the benefits of the patented route. The process is divided into three distinct stages: the formation of the silyl enol ether, the Grignard alkylation, and the final oxidative rearrangement. Each stage has been optimized in the patent examples to demonstrate reproducibility on a kilogram scale, providing a clear roadmap for technology transfer. Operators must ensure that the initial enolization is conducted under strictly anhydrous conditions to prevent the quenching of the LDA base, which would otherwise lead to incomplete conversion and lower yields. The detailed standardized synthesis steps see the guide below for specific operational parameters.
- React estra-4,9-diene-3,17-dione with LDA and trimethylchlorosilane in THF at low temperature to form the enol silyl ether intermediate XY-1.
- Perform a Grignard reaction on intermediate XY-1 using allyl magnesium bromide to introduce the allyl group, yielding intermediate XY-2.
- Treat intermediate XY-2 with tetrachloro-p-benzoquinone to effect double bond rearrangement and deprotection, followed by recrystallization to obtain pure altrenogest.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this synthesis route offers tangible strategic benefits beyond mere chemical elegance. The shift away from benzene and expensive oxidized precursors fundamentally alters the cost structure of altrenogest manufacturing. By utilizing commodity chemicals like THF, TMSCl, and allyl magnesium bromide, the reliance on specialized, high-cost reagents is minimized. This substitution not only lowers the direct material cost but also mitigates the risk of supply disruptions associated with niche chemical suppliers. Furthermore, the simplified workup procedures, which rely on standard aqueous extractions and filtrations rather than complex chromatographic separations, reduce the consumption of silica gel and organic solvents, contributing to significant operational expenditure savings.
- Cost Reduction in Manufacturing: The elimination of toxic benzene removes the need for specialized containment systems and expensive hazardous waste disposal services, which are major cost drivers in traditional steroid synthesis. Additionally, the high regioselectivity of the LDA-mediated step reduces the formation of hard-to-remove impurities, meaning less material is lost during the final recrystallization. This improvement in mass balance means that for every ton of starting material purchased, a greater proportion is converted into saleable high-purity product, effectively lowering the cost of goods sold (COGS) without requiring capital investment in new reactor hardware.
- Enhanced Supply Chain Reliability: The starting material, estra-4,9-diene-3,17-dione, is a well-established intermediate in the steroid industry with a mature global supply base. Unlike the proprietary or semi-synthetic precursors required by older French patents, this raw material is available from multiple vendors, preventing single-source dependency. The reagents used throughout the sequence, including LDA and tetrachloro-p-benzoquinone, are bulk industrial chemicals with stable pricing and consistent availability. This robustness ensures that production schedules can be maintained even during periods of market volatility, securing continuity of supply for downstream veterinary formulation clients.
- Scalability and Environmental Compliance: The process is designed with scale-up in mind, utilizing reaction conditions that are easily managed in standard glass-lined or stainless steel reactors. The absence of highly exothermic or dangerous reactions reduces the safety engineering controls required for large-scale batches. From an environmental perspective, the replacement of benzene with THF and the generation of water-soluble byproducts align with modern Green Chemistry principles. This facilitates easier regulatory approval in jurisdictions with strict environmental mandates, allowing manufacturers to operate with a smaller environmental footprint and reduced liability regarding emissions and effluent treatment.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and validation of this synthesis pathway. These answers are derived directly from the experimental data and comparative examples provided in the patent documentation, offering clarity on performance metrics and operational nuances. Understanding these details is essential for R&D teams evaluating the feasibility of adopting this technology for commercial production.
Q: How does this new synthesis route improve upon traditional methods regarding solvent toxicity?
A: Unlike prior art such as Netherlands patent NL6517141 which utilizes highly toxic benzene as a solvent, this optimized process employs anhydrous tetrahydrofuran (THF) and dichloromethane, significantly reducing environmental hazards and operator safety risks while simplifying waste treatment protocols.
Q: What represents the primary cost advantage of using LDA and TMSCl in the first step?
A: The use of Lithium Diisopropylamide (LDA) and trimethylchlorosilane allows for highly regioselective protection of the 3-carbonyl group under mild conditions. This specificity minimizes side reactions and impurity formation, thereby reducing the burden on downstream purification and increasing overall mass yield compared to less selective acidic protection methods.
Q: Why is tetrachloro-p-benzoquinone preferred for the final deprotection step?
A: Tetrachloro-p-benzoquinone acts as a dual-function reagent that facilitates both the removal of the trimethylsilyl protecting group and the necessary double bond rearrangement in a single operational step. This convergence eliminates the need for separate reduction-dehydrogenation steps found in older patents, streamlining the workflow and improving final product purity to over 99%.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Altrenogest Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition from laboratory patent to commercial reality requires a partner with deep technical expertise and robust infrastructure. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of the CN109232700B process are fully realized in a GMP-compliant environment. We maintain stringent purity specifications and operate rigorous QC labs equipped with advanced analytical instrumentation to verify that every batch of altrenogest meets the 99.6% purity benchmark demonstrated in the patent examples, guaranteeing efficacy and safety for end-user livestock applications.
We invite global pharmaceutical partners to collaborate with us to leverage this advanced synthesis technology for your veterinary product pipelines. By engaging with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that optimize both your supply chain resilience and your bottom line.
