Advanced Manufacturing of Altrenogest: A High-Yield Route for Veterinary Pharmaceutical Intermediates
Advanced Manufacturing of Altrenogest: A High-Yield Route for Veterinary Pharmaceutical Intermediates
The pharmaceutical and veterinary industries are constantly seeking robust, scalable, and cost-effective synthetic routes for high-value active ingredients. Patent CN110950920A introduces a groundbreaking preparation method for Tetraethenone, widely known as Altrenogest, a potent synthetic steroidal progestogen used extensively in animal husbandry. This novel methodology addresses critical bottlenecks found in traditional synthesis pathways, offering a streamlined three-step process that begins with a 3-protected estra-diene-17-ketone substrate. By leveraging a strategic combination of Grignard addition, controlled hydrolysis, and oxidative dehydrogenation, this technology delivers a final product with exceptional purity levels exceeding 99% and a total mass yield surpassing 75%. For R&D directors and procurement specialists, this represents a significant opportunity to optimize supply chains for veterinary drug intermediates, ensuring consistent quality while mitigating the risks associated with complex multi-step steroid syntheses.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Altrenogest has been plagued by inefficient routes that rely on expensive starting materials or harsh reaction conditions that compromise yield and purity. One prevalent prior art method utilizes Trenbolone as a raw material, requiring oxime formation to protect the carbonyl at the 3-position, followed by oxidation and Grignard reaction. This approach is fundamentally flawed due to the high cost of the starting material and the poor yield associated with the protection and deprotection cycles. Furthermore, alternative routes starting from estra-4,9-diene-3,17-diketone often necessitate harsh ketal protection conditions and difficult dehydrogenation steps, leading to low overall yields and significant competitive disadvantages. As illustrated in the reaction schemes of previous patents, such as the route depicted below, these conventional methods often struggle with incomplete reactions and the formation of complex byproduct mixtures that are difficult to purify.
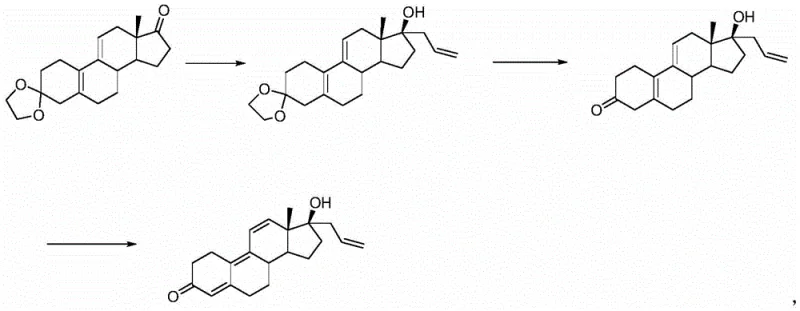
The Novel Approach
In stark contrast to these legacy methods, the technology disclosed in CN110950920A employs a highly efficient strategy starting from readily available and inexpensive 3-protected estra-Δ5,10, Δ9,11-dien-17-one. This innovative pathway simplifies the synthetic sequence by utilizing a stable protected intermediate that withstands the rigorous conditions of Grignard addition without degradation. The subsequent hydrolysis step is meticulously controlled to ensure complete removal of the protecting group without inducing unwanted double bond migration, a common failure point in older technologies. Finally, the dehydrogenation step utilizes specific quinone oxidants to cleanly establish the conjugated tetraene system. The general reaction scheme for this superior process is outlined below, demonstrating the logical flow from protected ketone to the final high-purity API intermediate.
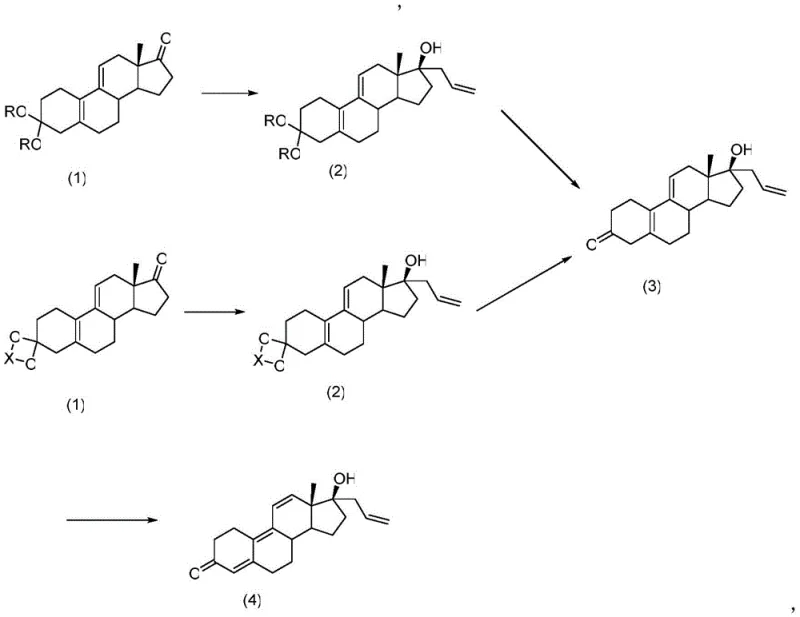
Mechanistic Insights into Grignard Addition and Oxidative Dehydrogenation
The core of this synthetic breakthrough lies in the precise execution of the Grignard reaction at the C17 position of the steroid nucleus. By dissolving the 3-protected substrate in organic solvents such as tetrahydrofuran or methyltetrahydrofuran and maintaining temperatures between -10°C and 20°C, the process ensures high stereoselectivity for the 17β-hydroxy-17α-propenyl configuration. The use of allyl magnesium bromide or chloride allows for the efficient introduction of the allyl side chain, which is crucial for the biological activity of the final progestogen. Following this, the hydrolysis step employs mild acidic conditions using acetic acid, sulfuric acid, or hydrochloric acid to cleave the 3-position ketal or acetal protecting groups. This step is critical; unlike prior art where harsh conditions led to isomerization, this method maintains the integrity of the Δ5,10 and Δ9,11 double bond systems while exposing the 3-ketone functionality necessary for the final transformation.
The final dehydrogenation step is equally sophisticated, utilizing oxidizing reagents such as tetrachlorobenzoquinone (chloranil) or 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ). These reagents are selected for their ability to abstract hydrogen atoms and introduce the necessary unsaturation to form the tetraene system without over-oxidizing the sensitive steroid backbone. The reaction is conducted in dichloromethane at temperatures ranging from 0°C to 40°C, ensuring that the reaction proceeds to completion with minimal byproduct formation. This mechanistic precision results in a crude product that is exceptionally clean, facilitating easy purification through simple crystallization from ethanol. For technical teams, this implies a robust process window where minor deviations in temperature or stoichiometry do not catastrophically impact the final quality, thereby enhancing the reliability of the manufacturing process.
How to Synthesize Altrenogest Efficiently
The synthesis of Altrenogest via this patented route involves three distinct operational stages that can be seamlessly integrated into existing steroid manufacturing facilities. The process begins with the preparation of the Grignard adduct, followed by an aqueous workup and acid-mediated deprotection, and concludes with oxidative aromatization of the A-ring and B-ring systems. Each step has been optimized for scalability, utilizing common industrial solvents and reagents that are easily sourced globally. The detailed standardized synthesis steps, including specific molar ratios, solvent volumes, and temperature profiles, are provided in the technical guide below to assist process engineers in replicating this high-efficiency protocol.
- Perform a Grignard reaction on 3-protected estra-diene-17-ketone using allyl magnesium halide at controlled low temperatures to form the 17-alpha-allyl intermediate.
- Execute an acidic hydrolysis step to remove the 3-position protecting group, yielding the 3-keto intermediate in dichloromethane solution.
- Conduct oxidative dehydrogenation using quinone-based oxidants like DDQ or chloranil to establish the final conjugated tetraene system.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits in terms of cost structure and supply security. The primary advantage stems from the utilization of cheap and stable raw materials, specifically the 3-protected estra-diene-17-ketone, which is far more economical than the specialized precursors required by older methods. This shift in raw material sourcing drastically reduces the direct material costs associated with production. Furthermore, the high total mass yield of over 75% means that less starting material is required to produce the same amount of final product, compounding the cost savings. The simplicity of the purification process, which relies on standard crystallization rather than complex chromatography, further lowers operational expenditures by reducing solvent consumption and processing time.
- Cost Reduction in Manufacturing: The elimination of expensive starting materials like Trenbolone and the reduction in the number of synthetic steps directly translate to substantial cost savings. By avoiding low-yield protection and deprotection cycles that characterize legacy routes, manufacturers can achieve a significantly lower cost of goods sold (COGS). The use of common reagents such as allyl magnesium halides and quinone oxidants ensures that the process remains economically viable even at fluctuating market prices for fine chemicals.
- Enhanced Supply Chain Reliability: Sourcing stability is a critical concern for long-term production contracts. Since the key starting materials for this route are bulk commodities in the pharmaceutical industry, the risk of supply disruption is minimized. The robustness of the reaction conditions, which tolerate a wide range of temperatures and solvent grades, ensures that production can continue uninterrupted even if specific reagent grades vary slightly. This reliability is essential for maintaining continuous supply to downstream veterinary drug formulators.
- Scalability and Environmental Compliance: The process is designed with industrial scale-up in mind, utilizing solvents like dichloromethane and ethanol that are easily recovered and recycled in modern facilities. The high purity of the intermediate products reduces the burden on waste treatment systems, as fewer toxic byproducts are generated compared to harsher oxidative methods. This alignment with green chemistry principles facilitates easier regulatory approval and supports the sustainability goals of modern chemical enterprises.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Altrenogest synthesis technology. These answers are derived directly from the experimental data and claims presented in patent CN110950920A, providing a factual basis for decision-making.
Q: What is the total yield and purity of the Altrenogest synthesis described in CN110950920A?
A: The patented method achieves a total mass yield higher than 75% and a final product purity exceeding 99%, ensuring high efficiency for industrial production.
Q: Why is the 3-position protection strategy critical in this synthesis?
A: Protecting the 3-carbonyl group prevents unwanted side reactions during the Grignard addition at the 17-position and allows for controlled deprotection later, minimizing byproduct formation.
Q: What oxidizing agents are suitable for the final dehydrogenation step?
A: The process utilizes tetrachlorobenzoquinone (chloranil) or 2,3-dichloro-5,6-dicyano-p-benzoquinone (DDQ) to effectively introduce the required double bonds without degrading the steroid skeleton.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Altrenogest Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient synthetic routes like the one described in CN110950920A for securing the global supply of veterinary pharmaceuticals. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless and risk-free. We are committed to delivering high-purity Altrenogest intermediates that meet stringent purity specifications, supported by our rigorous QC labs and state-of-the-art analytical capabilities. By partnering with us, you gain access to a supply chain that is not only cost-competitive but also technically superior and fully compliant with international quality standards.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can be integrated into your supply chain. Request a Customized Cost-Saving Analysis today to quantify the potential economic benefits for your specific operation. Our experts are ready to provide specific COA data and route feasibility assessments to demonstrate how we can become your trusted partner in the production of high-value steroid intermediates.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
