Industrial Scale Production of Pharmaceutical Grade Melphalan and Salts via Optimized pH Control
Introduction to Advanced Melphalan Manufacturing Technology
The landscape of oncology drug manufacturing is constantly evolving, driven by the need for higher purity standards and more cost-effective production methodologies. A significant breakthrough in this domain is detailed in patent CN102030671B, which outlines a robust preparing method for pharmaceutical grade Melphalan, as well as its hydrochloride and dihydrochloride salts. This technology represents a paradigm shift from traditional laboratory-scale syntheses to industrial-grade processes capable of handling technical grade raw materials while delivering exceptional purity. The core innovation lies in a sophisticated four-step protocol that integrates chlorination, hydrolysis, precise pH-controlled purification, and lyophilization. By leveraging this methodology, manufacturers can overcome the historical limitations associated with producing alkylating agents, ensuring a reliable supply of high-quality intermediates for the global pharmaceutical market.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the production of Melphalan and its salts has been fraught with challenges related to raw material costs and purification efficiency. Prior art, such as United States Patents US3032584 and US3032585, disclosed production techniques that were often restricted to high-purity, expensive starting materials to achieve acceptable quality. These conventional methods frequently struggled to effectively remove structurally similar impurities, such as monohydroxy melphalan, without resorting to costly and time-consuming chromatographic separations. Furthermore, the inability to utilize cheaper, technical grade precursors meant that the overall cost of goods sold (COGS) remained prohibitively high for large-scale commercial applications. The reliance on rigid synthetic pathways often resulted in lower yields and inconsistent batch-to-batch quality, posing significant risks for supply chain stability in the competitive oncology sector.
The Novel Approach
In stark contrast, the novel approach described in the patent data introduces a flexible and economically viable synthesis route. This method ingeniously utilizes 4-[bis(2-hydroxyethyl)-amino]-N-phthaloyl-L-phenylalanine ethyl ester as a starting material, which can be sourced as a technical grade commodity. The breakthrough is anchored in the strategic manipulation of pH values during the hydrolysis and purification stages. By adjusting the pH to a narrow window of 0.5 to 3, the process selectively precipitates the desired product while leaving impurities in solution. This eliminates the need for complex purification equipment and allows for the direct production of pharmaceutical grade material through simple filtration and lyophilization. The result is a streamlined process that drastically simplifies the manufacturing workflow while maintaining rigorous quality standards suitable for clinical applications.
Mechanistic Insights into pH-Controlled Purification and Chlorination
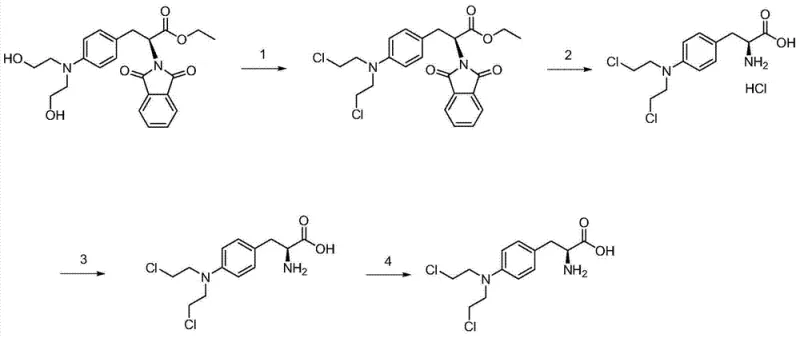
The chemical elegance of this synthesis lies in the precise control of reaction conditions to drive selectivity. The initial step involves a chlorination reaction where the hydroxyl groups of the starting material are converted into chloroethyl moieties using a chlorinating agent such as phosphorus oxychloride. This transformation is critical as it installs the reactive nitrogen mustard functionality essential for the drug's biological activity. Following chlorination, the phthaloyl protecting group is removed via acid hydrolysis, typically using hydrochloric acid under reflux conditions. This step liberates the amino group and generates the hydrochloride salt in situ. The subsequent purification mechanism is the heart of this technology; it exploits the differential solubility profiles of Melphalan salts versus their hydroxy-analogs at specific acidic pH levels. By maintaining the environment within the 0.5 to 3 pH range, the target molecule crystallizes out with high fidelity, effectively rejecting impurities that remain soluble in the mother liquor.
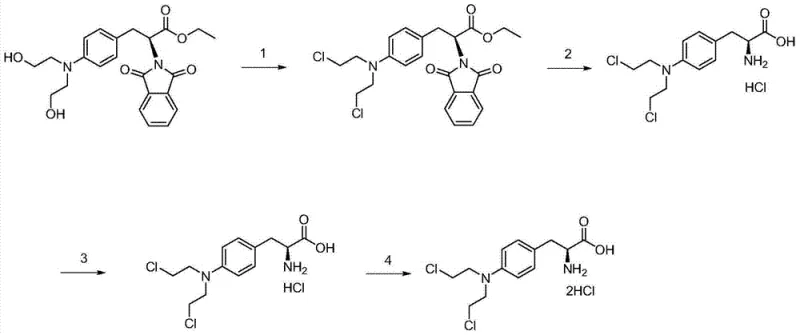
Furthermore, the process incorporates a decolorization step using activated carbon, which adsorbs organic by-products and colored impurities generated during the harsh acid reflux. This ensures the final product meets the stringent visual and chemical specifications required for injectable oncology drugs. The versatility of the mechanism allows for the production of different salt forms by simply altering the final pH adjustment and salification conditions. For instance, shifting the final pH to a neutral range (3.5-7) allows for the isolation of the free base, while further treatment with hydrochloric acid yields the dihydrochloride salt. This mechanistic flexibility provides a robust platform for manufacturing various forms of the active pharmaceutical ingredient (API) from a common intermediate, optimizing asset utilization in a multi-product facility.
How to Synthesize Pharmaceutical Grade Melphalan Efficiently
The synthesis of Melphalan derivatives via this patented route is designed for operational simplicity and scalability. The process begins with the chlorination of the protected phenylalanine derivative in a solvent like toluene, followed by an aqueous workup to isolate the chlorinated intermediate. This intermediate is then subjected to hydrolysis and salt formation, where precise temperature control and reflux times are maintained to ensure complete deprotection. The critical purification phase involves dissolving the crude solid, treating with activated carbon, and carefully adjusting the pH to induce crystallization. Detailed standardized operating procedures for each step, including specific reagent ratios and filtration protocols, are essential for replicating the high purity demonstrated in the patent examples. For a comprehensive guide on executing these steps with exact parameters, please refer to the technical documentation below.
- Chlorination Reaction: React 4-[bis(2-hydroxyethyl)-amino]-N-phthaloyl-L-phenylalanine ethyl ester with phosphorus oxychloride in toluene under reflux to convert hydroxyl groups to chloroethyl groups.
- Hydrolysis and Salt Formation: Hydrolyze the chlorinated intermediate using hydrochloric acid under reflux, followed by pH adjustment to 0.5-3 to precipitate the crude hydrochloride salt.
- Purification: Dissolve the crude solid in acid, treat with activated carbon for decolorization, and precisely adjust pH to 0.5-3 again to crystallize the purified product, removing impurities like monohydroxy melphalan.
- Final Processing: Perform a secondary salification with hydrochloric acid (for dihydrochloride) or neutralization (for free base), followed by lyophilization to obtain the final pharmaceutical grade solid.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this manufacturing technology offers profound advantages for procurement managers and supply chain directors seeking to optimize their API sourcing strategies. The ability to utilize technical grade raw materials directly translates into significant cost reductions in Melphalan manufacturing, as the premium associated with high-purity starting reagents is eliminated. This raw material flexibility creates a buffer against market volatility, ensuring that production costs remain stable even when supply chains for fine chemicals fluctuate. Moreover, the simplified four-step process reduces the number of unit operations required, leading to lower energy consumption and reduced labor hours per kilogram of output. These efficiencies compound to offer a highly competitive pricing structure for bulk purchasers without compromising on the quality of the final therapeutic agent.
- Cost Reduction in Manufacturing: The economic model of this process is fundamentally superior due to the elimination of expensive purification technologies. By relying on pH-controlled crystallization rather than chromatography, the process avoids the high capital and operational expenditures associated with resin columns and solvent recovery systems. The use of common reagents like phosphorus oxychloride and hydrochloric acid further drives down variable costs. Additionally, the high yield and purity achieved in the initial crystallization steps minimize the need for reprocessing or recycling, thereby maximizing material throughput and reducing waste disposal costs. This lean manufacturing approach ensures that the final cost of goods is optimized for maximum margin potential.
- Enhanced Supply Chain Reliability: Supply chain resilience is significantly bolstered by the robustness of this synthetic route. Since the process tolerates technical grade inputs, manufacturers are not constrained by the limited availability of ultra-high purity precursors, which can often be a bottleneck in the supply chain. The use of standard chemical reactors and filtration equipment means that production can be easily scaled or shifted between different facilities without requiring specialized infrastructure. This flexibility reduces lead time for high-purity pharmaceutical intermediates, allowing for faster response to market demand surges. Furthermore, the stability of the intermediates and the simplicity of the storage requirements contribute to a more secure and predictable supply continuity for downstream drug product manufacturers.
- Scalability and Environmental Compliance: The environmental footprint of this process is minimized through efficient atom economy and reduced solvent usage. The reliance on aqueous workups and simple organic solvents like toluene, which can be readily recovered and recycled, aligns with modern green chemistry principles. The absence of heavy metal catalysts or exotic reagents simplifies waste treatment and regulatory compliance, reducing the administrative burden on environmental health and safety teams. Scalability is inherent in the design, as the reaction conditions (reflux, stirring, filtration) are easily translated from pilot plant to commercial scale (100 kgs to 100 MT). This ensures that the technology can support the growing global demand for Melphalan-based therapies without encountering the typical teething problems associated with process scale-up.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the production of Melphalan and its salts. These insights are derived directly from the patented methodology and are intended to clarify the capabilities and benefits of this advanced synthesis route. Understanding these details is crucial for stakeholders evaluating the feasibility of integrating this technology into their existing supply chains. For more specific technical data or custom synthesis requests, our team is available to provide detailed assessments.
Q: How does this process ensure pharmaceutical grade purity from technical grade raw materials?
A: The process utilizes a unique multi-stage pH control strategy. By strictly maintaining the pH between 0.5 and 3 during the hydrolysis and purification steps, the solubility differences between the target Melphalan salt and impurities (such as monohydroxy melphalan) are exploited. This allows for the precipitation of the high-purity product while keeping impurities in the filtrate, achieving purity levels exceeding 99%.
Q: What are the key advantages of this synthesis route for large-scale manufacturing?
A: The primary advantage is the ability to use inexpensive, technical grade starting materials without compromising final quality. Additionally, the process relies on standard unit operations such as reflux, filtration, and lyophilization, which are easily scalable in existing chemical infrastructure. The elimination of complex chromatographic purification steps significantly reduces production costs and cycle times.
Q: Can this method produce different salt forms of Melphalan?
A: Yes, the platform technology is highly versatile. By modifying the final step, the same intermediate can be converted into Melphalan Dihydrochloride, Melphalan Hydrochloride, or free base Melphalan. For the dihydrochloride, a secondary salification is performed; for the free base, a neutralization step to pH 3.5-7 is applied before lyophilization.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melphalan Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of securing a stable and high-quality supply of oncology intermediates like Melphalan. Our expertise aligns perfectly with the advanced manufacturing principles outlined in recent patent innovations, allowing us to deliver products that meet the most stringent pharmaceutical standards. We possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that your project needs are met with precision and reliability. Our state-of-the-art facilities are equipped with rigorous QC labs and adhere to stringent purity specifications, guaranteeing that every batch of Melphalan or its salts delivered to you is free from critical impurities and ready for formulation.
We invite you to collaborate with us to leverage these technological advancements for your drug development pipeline. Our technical procurement team is ready to assist you with a Customized Cost-Saving Analysis tailored to your specific volume requirements. We encourage you to contact us today to request specific COA data and route feasibility assessments, ensuring that your supply chain is built on a foundation of scientific excellence and commercial viability. Let us be your partner in bringing life-saving therapies to patients faster and more efficiently.
