Advanced Manufacturing of Melphalan Intermediates: A Strategic Route for Global Pharma Supply Chains
Advanced Manufacturing of Melphalan Intermediates: A Strategic Route for Global Pharma Supply Chains
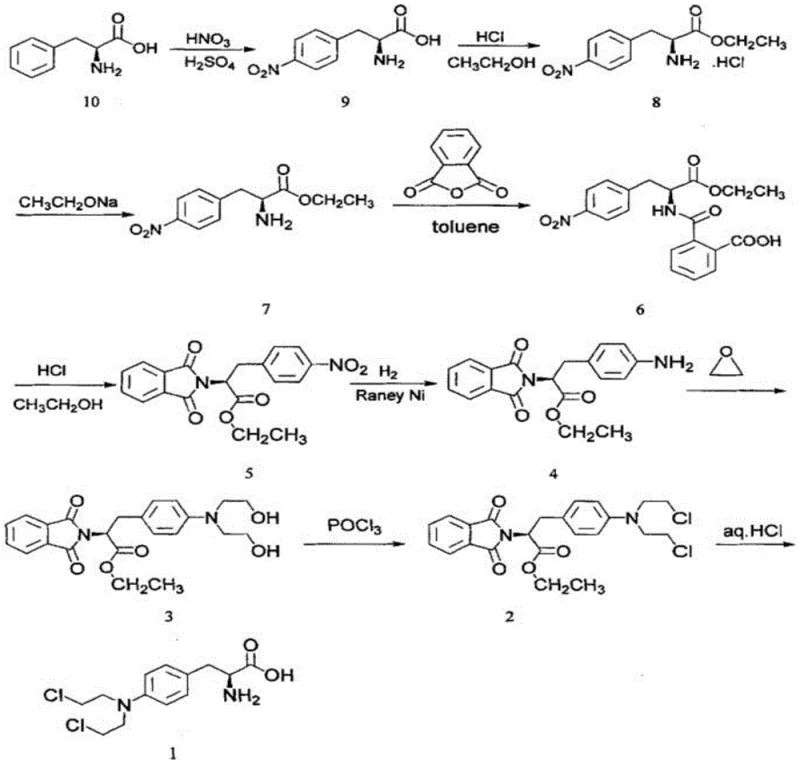
The global demand for oncology therapeutics continues to surge, driving the need for robust, scalable, and cost-effective synthesis routes for critical Active Pharmaceutical Ingredients (APIs) like Melphalan. Patent CN114315618A introduces a refined preparation method for synthesizing Melphalan that addresses longstanding challenges in yield optimization and impurity control. By utilizing L-phenylalanine as the chiral pool starting material, this methodology leverages a series of strategic transformations including nitration, esterification, acylation, Raney Ni catalytic hydrogenation, alkylation, halogenation, and deprotection. This comprehensive approach not only streamlines the production workflow but also aligns with modern Green Chemistry principles by mitigating the use of hazardous reagents found in legacy processes. For R&D directors and supply chain leaders, understanding the nuances of this pathway is essential for securing a reliable supply of high-purity pharmaceutical intermediates.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis of Melphalan has been plagued by significant technical and safety hurdles that complicate commercial scale-up. Early methodologies, such as those documented in US3032585a and related patents, often relied on the direct use of bis(2-chloroethyl)amine and elemental iodine. This approach presents severe drawbacks: bis(2-chloroethyl)amine is a known carcinogen requiring stringent containment protocols, while elemental iodine is not only expensive but acts as a strong oxidant that can cause unwanted substitution on the benzene ring at ortho and meta positions. Furthermore, traditional routes frequently struggle with the stability of ester protecting groups under strong acidic conditions, leading to premature deprotection and the generation of persistent impurities such as EP Impurity G and EP Impurity F. These impurities are notoriously difficult to remove during purification, thereby compromising the final API quality and increasing downstream processing costs significantly.
The Novel Approach
In stark contrast, the novel methodology outlined in CN114315618A offers a paradigm shift by employing L-phenylalanine as the foundational building block. This route ingeniously bypasses the need for hazardous alkylating agents in the early stages. Instead, it utilizes a phthalimide protection strategy that effectively shields the amine functionality during subsequent harsh reaction conditions. The process incorporates a highly efficient Raney Ni catalytic hydrogenation step to reduce the nitro group, avoiding the use of stoichiometric metal reducers that generate heavy metal waste. Moreover, the protocol explicitly substitutes toxic benzene solvents with chloroform or toluene during the acylation and halogenation steps without compromising reaction yields. This strategic redesign results in a concise synthesis with fewer operational steps, milder reaction conditions, and a cumulative yield of 38.8%, making it a superior candidate for industrial adoption.
Mechanistic Insights into Raney Ni-Catalyzed Hydrogenation and Phthalimide Protection
The core of this synthetic success lies in the precise orchestration of functional group transformations, particularly the reduction of the nitro group and the management of the amine protecting group. The conversion of intermediate 5 to intermediate 4 involves the reduction of the aromatic nitro group to an aniline derivative using Raney Nickel under a hydrogen atmosphere. This heterogeneous catalysis is critical because Raney Ni offers high surface area and activity, allowing the reaction to proceed efficiently in a mixture of ethyl acetate and methanol. The mechanism involves the adsorption of hydrogen and the nitro compound onto the catalyst surface, facilitating the stepwise addition of hydrogen atoms to convert the -NO2 group into -NH2. This step is pivotal as it prepares the molecule for the subsequent nucleophilic attack by ethylene oxide, and the high yield of 96% reported indicates minimal over-reduction or side reactions, ensuring a clean impurity profile for the downstream alkylation.
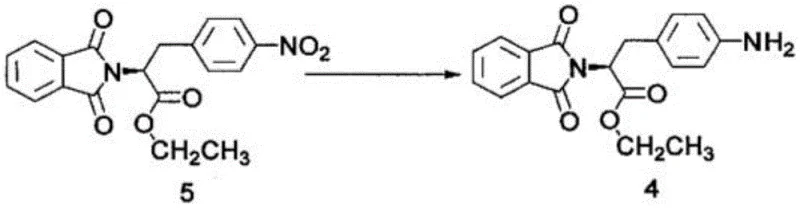
Equally important is the phthalimide protection strategy employed from intermediate 6 to 5 and maintained until the final step. The initial acylation of the free amine (intermediate 7) with phthalic anhydride forms an amic acid (intermediate 6), which subsequently undergoes thermal cyclization in the presence of acid to form the stable phthalimide ring (intermediate 5). This protecting group is robust enough to withstand the nitration and esterification conditions earlier in the sequence (if ordered differently) or the halogenation conditions later, yet it can be cleanly removed via acidic hydrolysis in the final step to reveal the primary amine of Melphalan. This orthogonality is vital for impurity control; by keeping the amine masked as a phthalimide, the process prevents the formation of poly-alkylated byproducts during the reaction with ethylene oxide and phosphorus oxychloride, thereby drastically simplifying the purification of the final drug substance.
How to Synthesize Melphalan Efficiently
Implementing this synthesis requires strict adherence to temperature controls and reagent stoichiometry to maximize the reported yields. The process begins with the careful nitration of L-phenylalanine at low temperatures (0-5°C) to prevent oxidation, followed by esterification and free-basing to prepare the substrate for protection. The subsequent acylation and cyclization steps establish the necessary nitrogen protection, setting the stage for the critical reduction and functionalization sequence. Detailed standard operating procedures regarding solvent recovery, catalyst handling, and crystallization parameters are essential for reproducibility. For a comprehensive breakdown of the standardized synthesis steps, please refer to the technical guide below.
- Nitration of L-Phenylalanine using mixed acid (HNO3/H2SO4) at 0-5°C to form 4-nitrophenylalanine.
- Protection of the amine group via acylation with phthalic anhydride followed by cyclization to form the phthalimide derivative.
- Catalytic hydrogenation of the nitro group using Raney Ni, followed by alkylation with ethylene oxide and halogenation with POCl3.
- Final acidic hydrolysis to remove the phthalimide protecting group and isolate Melphalan.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the transition to this optimized synthetic route offers tangible strategic benefits beyond mere technical feasibility. The shift away from carcinogenic reagents and toxic solvents directly translates to reduced regulatory compliance costs and lower insurance premiums associated with hazardous material handling. By utilizing commodity chemicals like L-phenylalanine and phthalic anhydride, the supply chain becomes more resilient against raw material volatility, ensuring consistent production schedules. Furthermore, the elimination of difficult-to-remove impurities reduces the burden on quality control laboratories and minimizes batch rejection rates, leading to substantial cost savings in the overall manufacturing lifecycle.
- Cost Reduction in Manufacturing: The replacement of expensive elemental iodine and carcinogenic bis(2-chloroethyl)amine with cost-effective reagents like ethylene oxide and phosphorus oxychloride significantly lowers the bill of materials. Additionally, the high efficiency of the Raney Ni catalyst, which can often be recovered and reused, reduces the consumption of precious metals or stoichiometric reducing agents. The streamlined process flow, characterized by high individual step yields such as 99% for the free-basing step and 96% for hydrogenation, minimizes material loss and maximizes throughput per batch, driving down the cost per kilogram of the final API intermediate.
- Enhanced Supply Chain Reliability: Sourcing L-phenylalanine is inherently more stable than sourcing specialized alkylating agents, as it is a bulk fermentation product available from multiple global suppliers. This diversification of the raw material base mitigates the risk of supply disruptions caused by single-source dependencies. Moreover, the robustness of the phthalimide protection group allows for greater flexibility in scheduling and storage of intermediates, as they are less prone to degradation compared to unprotected amines. This stability ensures that inventory can be managed more effectively, reducing lead times for high-purity pharmaceutical intermediates and supporting just-in-time manufacturing models.
- Scalability and Environmental Compliance: The substitution of benzene with chloroform or toluene addresses major environmental, health, and safety (EHS) concerns, facilitating easier permitting for new production lines in regions with strict environmental regulations. The aqueous workups described, such as the neutralization with sodium bicarbonate and simple filtration steps, are readily scalable from pilot plant to multi-ton commercial production without requiring exotic equipment. The reduction in hazardous waste generation, particularly the avoidance of iodine-containing waste streams, simplifies effluent treatment processes and aligns the manufacturing footprint with corporate sustainability goals.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this Melphalan synthesis route. These insights are derived directly from the patent specifications and are intended to clarify the operational advantages and chemical rationale behind the process design. Understanding these details is crucial for technical teams evaluating the feasibility of technology transfer.
Q: Why is L-Phenylalanine preferred as the starting material for Melphalan synthesis?
A: Using L-Phenylalanine ensures the preservation of chirality throughout the synthesis, yielding the biologically active levorotatory form (L-Melphalan) which possesses superior antitumor activity compared to the racemate.
Q: How does this process improve safety compared to conventional methods?
A: This route avoids the use of carcinogenic bis(2-chloroethyl)amine and elemental iodine found in older patents. Additionally, it replaces toxic benzene solvents with safer alternatives like chloroform or toluene, significantly reducing EHS risks.
Q: What is the overall yield and purity profile of this synthetic route?
A: The optimized process achieves a total yield of approximately 38.8% with high individual step yields (e.g., 96% for hydrogenation). The specific sequence minimizes the formation of difficult-to-remove impurities like EP Impurity G and F.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Melphalan Supplier
At NINGBO INNO PHARMCHEM, we recognize that the successful commercialization of oncology drugs depends on the availability of high-quality intermediates produced via validated, scalable routes. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the transition from laboratory bench to industrial reactor is seamless. We adhere to stringent purity specifications and operate rigorous QC labs equipped with state-of-the-art analytical instrumentation to guarantee that every batch of Melphalan intermediate meets the highest international standards. Our commitment to process excellence means we can deliver the consistency and reliability required by top-tier pharmaceutical manufacturers.
We invite you to collaborate with us to optimize your supply chain for Melphalan and related oncology APIs. Our technical procurement team is ready to provide a Customized Cost-Saving Analysis tailored to your specific volume requirements. Please contact us today to request specific COA data and route feasibility assessments, and let us demonstrate how our advanced manufacturing capabilities can support your long-term strategic goals.
