Advanced Two-Zone Hydroformylation for High-Purity Tricyclodecanedialdehyde Production
Advanced Two-Zone Hydroformylation for High-Purity Tricyclodecanedialdehyde Production
The chemical industry continuously seeks robust methodologies for synthesizing complex intermediates that serve as the backbone for high-performance polymers and pharmaceutical agents. Patent CN100400490C introduces a groundbreaking advancement in the preparation of tricyclodecanedialdehyde, a critical precursor for TCD-diol and TCD-diamine derivatives. This technology addresses long-standing challenges in hydroformylation processes, specifically focusing on the stability of rhodium catalysts and the suppression of unwanted side reactions during the conversion of dicyclopentadiene. By implementing a sophisticated multi-zone reaction strategy, the process achieves high space-time yields while operating with remarkably low concentrations of precious metal catalysts. For R&D directors and procurement specialists alike, this innovation represents a pivotal shift towards more sustainable and economically viable manufacturing protocols for fine chemical intermediates. The ability to produce high-purity tricyclodecanedialdehyde without the burden of complex ligand separation systems opens new avenues for cost-effective scale-up in the global supply chain.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional hydroformylation processes for converting dicyclopentadiene into tricyclodecanedialdehyde have historically been plagued by significant thermodynamic and kinetic hurdles. A primary issue arises from the reversible nature of the Diels-Alder reaction; under the elevated temperatures typically required for hydroformylation, dicyclopentadiene tends to undergo a retro-Diels-Alder decomposition, reverting to monomeric cyclopentadiene. This monomer is detrimental because it forms stable, catalytically inactive complexes with rhodium, effectively poisoning the catalyst and necessitating the use of excessive amounts of this precious metal to maintain reaction rates. Furthermore, conventional methods often rely on ligand-modified catalysts to stabilize the rhodium, but these ligands possess high boiling points that make their separation from the product via distillation energetically prohibitive and technically difficult. The result is a process characterized by high operational costs, significant rhodium losses due to thermal decomposition, and complex downstream purification requirements that hinder commercial scalability.
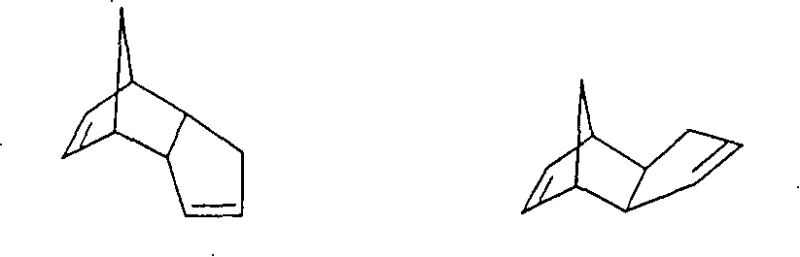
The Novel Approach
The methodology disclosed in patent CN100400490C circumvents these intrinsic limitations through a clever manipulation of reaction conditions across distinct thermal zones. Instead of subjecting the reaction mixture to a single, uniform high temperature, the process utilizes a sequential temperature gradient. In the initial reaction zone, the temperature is maintained between 80°C and 120°C, a range sufficiently low to suppress the retro-Diels-Alder decomposition of the starting material while still allowing the hydroformylation of the more reactive, strained double bond. Once this initial conversion stabilizes the ring system, the mixture is transferred to a subsequent zone where the temperature is raised to between 120°C and 150°C. This strategic increase accelerates the hydroformylation of the remaining, less reactive double bond without risking the formation of catalyst-poisoning cyclopentadiene. This approach allows for the use of unmodified rhodium catalysts at very low concentrations, drastically reducing raw material costs and simplifying the entire production workflow for reliable fine chemical intermediates supplier operations.
Mechanistic Insights into Rhodium-Catalyzed Hydroformylation
The core of this technological breakthrough lies in the precise control of the catalytic cycle using unmodified rhodium species, likely HRh(CO)4, generated in situ from precursors like Rh(CO)2(acac). In standard single-zone processes, the equilibrium between dicyclopentadiene and cyclopentadiene shifts towards the monomer at high temperatures, leading to rapid catalyst deactivation. However, by initiating the reaction at the lower temperature range of 80°C to 120°C, the process kinetically favors the addition of syngas (CO/H2) across the strained double bond of the cyclopentadienyl ring. This initial hydroformylation event effectively locks the ring structure, preventing the reverse dissociation into monomeric cyclopentadiene. Consequently, the reaction mixture entering the second zone contains predominantly mono-aldehyde intermediates that are thermally stable against retro-Diels-Alder cleavage. This mechanistic pathway ensures that the rhodium catalyst remains in its active carbonyl form throughout the process, minimizing the risk of precipitation as metallic rhodium and ensuring consistent catalytic turnover numbers essential for cost reduction in pharmaceutical intermediates manufacturing.
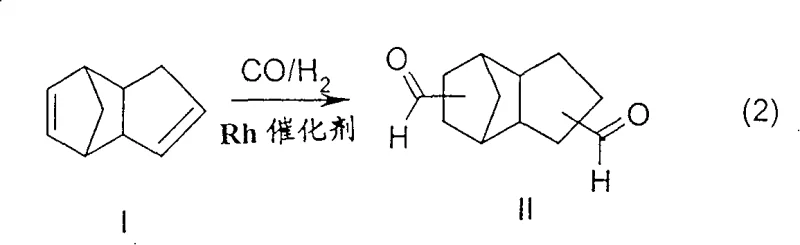
Furthermore, the absence of bulky phosphine or phosphite ligands plays a crucial role in the purity profile of the final product. Ligand-free rhodium catalysts are not only more active per atom of metal but also eliminate the formation of high-boiling byproducts that complicate purification. The reaction proceeds under superatmospheric pressures ranging from 200 to 350 bar, which further drives the equilibrium towards the desired aldehyde products and maintains the solubility of the gaseous reactants in the liquid phase. The resulting tricyclodecanedialdehyde is obtained as a mixture of regioisomers and stereoisomers, reflecting the endo/exo nature of the starting dicyclopentadiene, yet the process ensures high conversion rates with minimal formation of heavy condensation products. This level of control over the impurity spectrum is vital for downstream applications where high-purity tricyclodecanedialdehyde is required for the synthesis of specialized polyols and curing agents.
How to Synthesize Tricyclodecanedialdehyde Efficiently
Implementing this advanced hydroformylation protocol requires careful attention to reactor design and thermal management to fully realize its efficiency benefits. The process is ideally suited for continuous operation, where the reaction mixture flows sequentially through reactors configured to maintain the specific temperature gradients described in the patent. Operators must ensure that the transition between the first and second reaction zones is seamless to prevent any thermal shock that could induce unwanted side reactions. The use of standard industrial reactors, such as stirred tanks or bubble columns, is feasible, provided they can withstand the high pressures of up to 350 bar. Following the reaction, the separation of the rhodium catalyst is achieved efficiently using ion exchange resins, a step that is facilitated by the absence of interfering ligands. Detailed standardized synthesis steps see the guide below for specific operational parameters regarding flow rates and residence times.
- Prepare the reaction medium by dissolving an unmodified rhodium catalyst precursor, such as Rh(CO)2(acac), into dicyclopentadiene or a suitable solvent to achieve a rhodium concentration between 2 and 20 weight ppm.
- Introduce the mixture into the first reaction zone maintained at a temperature between 80°C and 120°C under a CO/H2 pressure of 200 to 350 bar to selectively hydroformylate the strained double bond.
- Transfer the effluent directly to a subsequent reaction zone heated to 120°C to 150°C to complete the hydroformylation of the remaining double bond, followed by catalyst separation via ion exchange.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the adoption of this two-zone hydroformylation technology offers transformative benefits for supply chain stability and overall manufacturing economics. By eliminating the need for expensive phosphorus-based ligands, the process removes a significant cost driver associated with both raw material procurement and waste disposal. The simplified downstream processing, which avoids energy-intensive distillation steps required to separate products from high-boiling ligands, translates directly into substantial cost savings in fine chemical manufacturing. Moreover, the ability to operate with extremely low rhodium concentrations—typically between 2 and 20 weight ppm—drastically reduces the capital tied up in precious metal inventory and minimizes the financial risk associated with metal loss during production cycles. These factors combine to create a more resilient supply chain capable of delivering consistent volumes of high-quality intermediates without the volatility associated with complex ligand-based chemistries.
- Cost Reduction in Manufacturing: The elimination of ligand modification steps and the subsequent ease of product separation significantly lower the operational expenditure per kilogram of product. Without the need to recover or dispose of high-boiling phosphine ligands, energy consumption during the purification phase is drastically reduced. Additionally, the high catalytic activity of the unmodified rhodium species allows for lower metal loading, which directly decreases the raw material cost component. This streamlined approach ensures that the production of tricyclodecanedialdehyde remains economically competitive even when fluctuating market prices for precious metals occur, providing a stable cost structure for long-term contracts.
- Enhanced Supply Chain Reliability: The robustness of the unmodified rhodium catalyst system against deactivation ensures consistent production rates and minimizes unplanned downtime caused by catalyst regeneration or replacement. Since the process suppresses the formation of catalyst-poisoning cyclopentadiene, the reactor runs can be extended, leading to improved asset utilization and more predictable delivery schedules. This reliability is crucial for downstream manufacturers who depend on a steady supply of TCD-dialdehyde for their own production of polyols and diamines, thereby strengthening the entire value chain against disruptions common in specialty chemical logistics.
- Scalability and Environmental Compliance: The process is inherently scalable, utilizing standard high-pressure reactor technologies that are widely available in the fine chemical industry. The reduction in rhodium usage and the absence of toxic phosphorus ligands simplify waste stream management, making it easier to comply with stringent environmental regulations regarding heavy metal discharge and organic waste. The ability to recover rhodium quantitatively from ion exchange resins further enhances the sustainability profile of the process, aligning with modern green chemistry principles and reducing the environmental footprint of commercial scale-up of complex polymer additives.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this hydroformylation technology. Understanding these details is essential for engineering teams evaluating the feasibility of integrating this process into existing facilities. The answers provided are derived directly from the technical specifications and experimental data contained within the patent documentation, ensuring accuracy and relevance for decision-makers. These insights cover catalyst handling, reaction safety, and product quality assurance, providing a comprehensive overview for stakeholders involved in the procurement and production of high-value chemical intermediates.
Q: Why is a two-zone temperature profile critical for TCD-dialdehyde synthesis?
A: A two-zone profile prevents the retro-Diels-Alder reaction. The lower temperature in the first zone (80-120°C) stabilizes the dicyclopentadiene ring against decomposition into cyclopentadiene, which would otherwise deactivate the rhodium catalyst, while the higher temperature in the second zone accelerates the reaction of the less reactive double bond.
Q: What are the advantages of using unmodified rhodium catalysts in this process?
A: Using unmodified rhodium catalysts eliminates the need for high-boiling phosphine or phosphite ligands. This significantly simplifies downstream processing, allowing for easier separation of the product via distillation without the high energy consumption associated with separating products from heavy ligands.
Q: How does this process impact the economic feasibility of downstream TCD-diol production?
A: By minimizing rhodium loss and avoiding expensive ligand removal steps, the overall cost of the TCD-dialdehyde intermediate is reduced. Since TCD-dialdehyde is the key precursor for TCD-diol and TCD-diamine, these upstream efficiencies directly translate to substantial cost savings in the manufacturing of the final polyol and diamine derivatives.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricyclodecanedialdehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of efficient and scalable synthetic routes for high-value intermediates like tricyclodecanedialdehyde. As a leading CDMO partner, we possess extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that our clients receive materials that meet the most stringent purity specifications. Our rigorous QC labs are equipped to analyze complex isomeric mixtures and trace metal residues, guaranteeing that every batch of TCD-dialdehyde delivered adheres to the highest industry standards. We understand that the transition from laboratory innovation to industrial reality requires not just chemical expertise but also a deep commitment to quality assurance and regulatory compliance, which forms the cornerstone of our service offering to global pharmaceutical and polymer manufacturers.
We invite you to engage with our technical procurement team to discuss how this advanced hydroformylation technology can be tailored to your specific production needs. By requesting a Customized Cost-Saving Analysis, you can gain a clear understanding of the potential economic benefits of switching to this ligand-free process. We encourage you to contact us today to obtain specific COA data and route feasibility assessments that will demonstrate our capability to support your supply chain with reliable, high-quality tricyclodecanedialdehyde. Let us collaborate to optimize your manufacturing processes and secure a competitive advantage in the global market for fine chemical intermediates.
