Advanced Rhodium-Catalyzed Hydroformylation for Scalable TCD-Dialdehyde Production
The chemical industry continuously seeks robust methodologies for synthesizing complex intermediates that balance high purity with economic viability. Patent CN100400490C introduces a transformative approach to the preparation of tricyclodecanedialdehyde (TCD-dialdehyde), a critical precursor for high-performance polyamides and epoxy curing agents. This innovation addresses the longstanding challenge of catalyst deactivation and expensive metal loss inherent in traditional hydroformylation processes. By employing a specific multi-zone temperature strategy with unmodified rhodium catalysts, the technology enables efficient conversion of dicyclopentadiene under superatmospheric pressures. For R&D directors and procurement specialists, this represents a significant opportunity to optimize the supply chain for high-purity pharmaceutical intermediates and specialty polymer additives. The following analysis dissects the technical merits and commercial implications of this patented route.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the hydroformylation of dicyclopentadiene has been plagued by thermodynamic and kinetic hurdles that compromise process economics. Conventional methods often rely on ligand-modified rhodium catalysts, such as those using triphenylphosphine, to stabilize the metal center. However, these ligands possess high boiling points that closely match the product, making separation via distillation energetically prohibitive and leading to significant thermal degradation of the sensitive dialdehyde product. Alternatively, processes using unmodified cobalt or rhodium catalysts at single high temperatures suffer from the reverse Diels-Alder reaction, where the starting material decomposes into cyclopentadiene. This decomposition product forms catalytically inactive complexes with the rhodium, necessitating excessively high metal loadings to maintain reaction rates, which drastically inflates raw material costs and creates severe issues with precious metal recovery.
The Novel Approach
The methodology disclosed in CN100400490C circumvents these pitfalls through a sophisticated spatial and thermal management of the reaction environment. Instead of a single isothermal condition, the process utilizes at least two distinct reaction zones. The first zone operates at a moderate temperature range of 80 to 120°C, sufficient to hydroformylate the more reactive, strained double bonds of the dicyclopentadiene without triggering significant reverse Diels-Alder decomposition. The effluent is then passed to a subsequent zone heated to 120 to 150°C, accelerating the conversion of the remaining less reactive double bonds. This stepwise heating profile allows for the use of unmodified rhodium catalysts at remarkably low concentrations, typically between 2 to 20 ppm, thereby eliminating ligand separation costs and minimizing precious metal inventory risks while maintaining high space-time yields.
Mechanistic Insights into Stepwise Temperature Hydroformylation
The core innovation lies in the kinetic control of the competing reactions within the reactor system. Dicyclopentadiene exists as an equilibrium mixture of endo and exo isomers, both containing strained norbornene-type double bonds. At elevated temperatures typically required for full conversion, the retro-Diels-Alder reaction becomes dominant, releasing monomeric cyclopentadiene. This monomer acts as a potent poison for unmodified rhodium carbonyl catalysts, forming stable, inactive pi-complexes that remove the metal from the catalytic cycle. By initiating the reaction in a lower temperature zone (80-120°C), the process selectively targets the highly strained double bonds first. Once these are converted to aldehyde groups, the resulting monoaldehyde intermediate is no longer susceptible to the retro-Diels-Alder fragmentation. This structural stabilization permits the temperature to be safely raised in the second zone to drive the second hydroformylation step to completion without the risk of generating the catalyst-poisoning cyclopentadiene species.
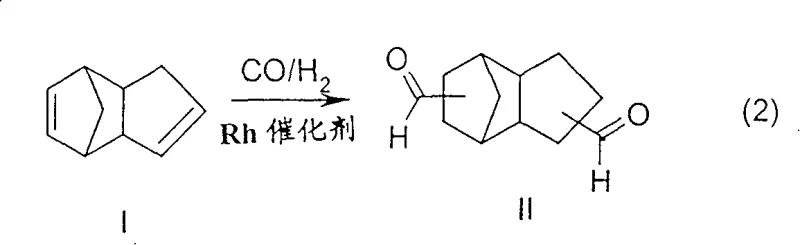
Furthermore, the use of unmodified rhodium catalysts, specifically species like HRh(CO)4 generated in situ from precursors such as rhodium acetylacetonate, offers distinct mechanistic advantages regarding downstream processing. Unlike phosphine-modified systems, the absence of bulky organic ligands means the catalyst remains fully soluble and can be efficiently removed using ion exchange resins directly from the reaction mixture, even under residual pressure. This capability prevents the thermal decomposition of the rhodium carbonyls into metallic rhodium sludge, a common failure mode in high-temperature distillation setups. The ability to recover the catalyst quantitatively via ion exchange not only secures the valuable rhodium inventory but also ensures the final TCD-dialdehyde product meets stringent purity specifications required for downstream hydrogenation to TCD-diol or reductive amination to TCD-diamine.
How to Synthesize Tricyclodecanedialdehyde Efficiently
Implementing this synthesis requires precise control over reactor zoning and pressure management to replicate the patented efficiency. The process begins with dissolving the rhodium precursor in the dicyclopentadiene feed, optionally with a solvent like pentanol or toluene, to achieve the target metal concentration. The mixture is then pressurized with synthesis gas (CO/H2) to between 200 and 350 bar. Critical to success is the thermal gradient; the reaction stream must reside in the initial lower-temperature zone long enough to consume the reactive diene character before entering the higher-temperature finishing zone. Detailed standardized synthesis steps see the guide below.
- Prepare a reaction mixture containing dicyclopentadiene and an unmodified rhodium catalyst (2-20 ppm Rh) in a suitable solvent or bulk.
- Conduct hydroformylation in a first reaction zone at 80-120°C and 200-350 bar pressure to convert the strained double bonds.
- Transfer the mixture to a subsequent reaction zone heated to 120-150°C to complete the conversion to TCD-dialdehyde while preventing catalyst deactivation.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the shift to this ligand-free, stepwise hydroformylation process translates directly into enhanced operational resilience and cost structure optimization. The elimination of expensive phosphine ligands removes a complex purification bottleneck, allowing for simpler distillation protocols that consume less energy and reduce the thermal load on the product. This simplification significantly lowers the barrier for commercial scale-up of complex intermediates, as the process is compatible with standard high-pressure reactor configurations like bubble columns or loop reactors without requiring exotic separation equipment. Consequently, manufacturers can achieve substantial cost savings in fine chemical manufacturing by reducing both utility consumption and the capital expenditure associated with downstream purification trains.
- Cost Reduction in Manufacturing: The ability to operate with rhodium concentrations as low as 2 to 20 ppm represents a drastic reduction in precious metal working capital compared to prior art methods requiring hundreds of ppm. Since the catalyst is not deactivated by cyclopentadiene due to the temperature staging, there is no need to overcharge the reactor with expensive metal to compensate for loss. Furthermore, the absence of high-boiling ligands eliminates the need for high-vacuum, high-energy distillation columns, leading to significantly reduced utility costs and improved overall process economics without compromising yield.
- Enhanced Supply Chain Reliability: By mitigating the risk of catalyst deactivation, the process ensures consistent reaction rates and predictable batch cycles, which is crucial for maintaining reliable pharmaceutical intermediates supplier status. The robustness of the unmodified rhodium system against thermal degradation means fewer unplanned shutdowns for reactor cleaning or catalyst replenishment. Additionally, the compatibility with continuous flow operations allows for a steady output of TCD-dialdehyde, smoothing out inventory fluctuations and ensuring a dependable supply of this critical building block for downstream polymer and resin production.
- Scalability and Environmental Compliance: The process design inherently supports green chemistry principles by minimizing waste generation associated with ligand disposal and metal recovery. The efficient recovery of rhodium via ion exchange resins prevents heavy metal contamination in waste streams, aligning with increasingly strict environmental regulations. Moreover, the modular nature of the multi-zone reactor setup facilitates easy capacity expansion, enabling producers to respond agilely to market demand surges for TCD-derivatives while maintaining a low environmental footprint through optimized atom economy and energy usage.
Frequently Asked Questions (FAQ)
The following questions address common technical inquiries regarding the implementation and benefits of this hydroformylation technology. These insights are derived directly from the experimental data and claims within the patent documentation, providing clarity on reaction conditions and catalyst management. Understanding these nuances is essential for technical teams evaluating the feasibility of adopting this route for large-scale production.
Q: How does this process prevent rhodium catalyst loss compared to conventional methods?
A: By utilizing a stepwise temperature profile, the process minimizes the reverse Diels-Alder reaction that generates cyclopentadiene, which typically forms inactive complexes with unmodified rhodium catalysts.
Q: What are the advantages of using unmodified rhodium catalysts in this synthesis?
A: Unmodified catalysts eliminate the need for high-boiling phosphine ligands, significantly simplifying product separation via distillation and reducing energy consumption and thermal stress on the product.
Q: Can this hydroformylation method be scaled for industrial production?
A: Yes, the patent explicitly supports continuous operation in various reactor types such as bubble columns or loop reactors, facilitating commercial scale-up from pilot to multi-ton annual production.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Tricyclodecanedialdehyde Supplier
At NINGBO INNO PHARMCHEM, we recognize the strategic importance of efficient intermediate synthesis in the global chemical value chain. Our technical team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the stepwise rhodium hydroformylation process are translated into robust manufacturing realities. We adhere to stringent purity specifications and operate rigorous QC labs to guarantee that every batch of TCD-dialdehyde meets the exacting standards required for high-performance polymer and pharmaceutical applications. Our commitment to quality ensures that your downstream processes, whether hydrogenation or amination, proceed with maximum efficiency and minimal impurity burden.
We invite you to collaborate with us to leverage these advanced synthetic capabilities for your specific project needs. Contact our technical procurement team today to request a Customized Cost-Saving Analysis tailored to your volume requirements. We are prepared to provide specific COA data and comprehensive route feasibility assessments to demonstrate how our optimized manufacturing processes can enhance your supply chain stability and reduce total landed costs. Let us be your partner in driving innovation and efficiency in the production of next-generation chemical intermediates.
