Advanced Milrinone Manufacturing: Technical Breakthroughs and Commercial Scalability Analysis
The cardiovascular therapeutic landscape relies heavily on the consistent supply of high-quality phosphodiesterase inhibitors, with Milrinone standing out as a critical agent for treating refractory heart failure. Recent intellectual property developments, specifically patent CN111377857B, have introduced a transformative approach to synthesizing this vital compound, addressing long-standing inefficiencies in prior art. This technical disclosure outlines a convergent synthetic strategy that utilizes 1-(4-pyridyl)-2-acetone and 2-cyano-3-dimethylamino acrylamide as key starting materials. By shifting away from toxic linear pathways, this method offers a robust framework for producing high-purity Milrinone suitable for stringent pharmaceutical applications. The structural integrity of the final molecule is paramount for its efficacy as a PDE III inhibitor, ensuring optimal calcium ion transport modulation within cardiac muscle cells. 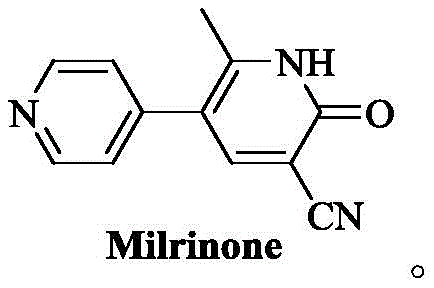
For R&D Directors evaluating process feasibility, the elimination of hazardous reagents represents a significant advancement in green chemistry principles within API manufacturing. The traditional reliance on malononitrile or potassium cyanide has historically complicated waste management and increased regulatory compliance burdens. This new methodology operates under mild alkaline conditions with a pH range of 12 to 14, utilizing common inorganic bases like sodium hydroxide. Such parameters simplify the engineering controls required for reactor design, thereby reducing capital expenditure barriers for technology adoption. Furthermore, the reaction proceeds efficiently at reflux temperatures between 60°C and 85°C, negating the need for energy-intensive cryogenic systems often demanded by alternative coupling strategies. This thermal profile not only enhances safety but also aligns with sustainability goals by lowering the overall carbon footprint of the production facility.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the industrial production of Milrinone has been plagued by complex multi-step sequences that compromise both economic efficiency and operational safety. Early linear synthetic routes, as depicted in various prior art documents, often necessitate the use of highly toxic malononitrile in the final cyclization steps. 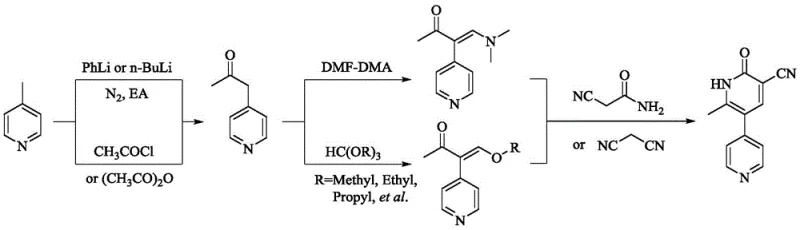
These conventional pathways frequently suffer from low atom economy, resulting in substantial quantities of chemical waste that require costly disposal protocols. Additionally, the crude products obtained from these older methods often exhibit undesirable red coloration, indicating the presence of difficult-to-remove conjugated impurities. Achieving injection-grade purity from such crude materials typically demands multiple recrystallization cycles, which drastically reduces the overall yield to approximately 38% in some convergent variations. The reliance on hazardous solvents like dichloroethane in certain bromination steps further exacerbates environmental concerns and worker safety risks. Consequently, procurement teams face volatile pricing structures driven by the high cost of waste treatment and the low efficiency of raw material conversion.
The Novel Approach
In stark contrast, the methodology disclosed in CN111377857B streamlines the synthesis into a more direct and manageable process that significantly mitigates these historical pain points. By employing 2-cyano-3-dimethylamino acrylamide as a building block, the reaction avoids the generation of toxic byproducts associated with cyanide sources. 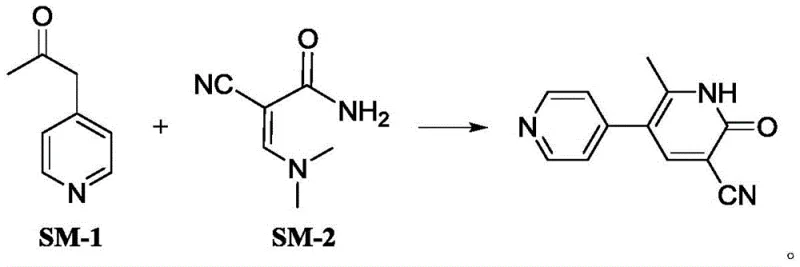
This innovative route facilitates a cleaner reaction profile where the target molecule precipitates as white crystals, simplifying the downstream purification process immensely. The patent data indicates that yields can reach approximately 90%, representing a substantial improvement over the 70% benchmark of previous generations. This increase in efficiency directly translates to better resource utilization, allowing manufacturers to produce more active pharmaceutical ingredient per batch without expanding facility footprints. Moreover, the use of ethanol as a preferred solvent aligns with modern solvent selection guides, offering a safer and more environmentally benign alternative to chlorinated hydrocarbons. For supply chain heads, this reliability in process performance ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed purification steps.
Mechanistic Insights into Alkaline-Catalyzed Condensation
Understanding the mechanistic underpinnings of this synthesis is crucial for R&D teams aiming to replicate or scale the process effectively. The reaction proceeds through a base-catalyzed condensation where the alkaline environment activates the methylene group of the acetone derivative. The hydroxide ions abstract a proton to generate a nucleophilic enolate species, which subsequently attacks the electron-deficient double bond of the acrylamide component. This Michael-type addition is followed by an intramolecular cyclization that constructs the pyridone ring system characteristic of Milrinone. The precise control of pH between 12 and 14 is critical; insufficient alkalinity fails to drive the equilibrium towards product formation, while excessive base may lead to hydrolysis of the nitrile group. The thermal energy provided at 75-80°C ensures that the activation energy barrier for cyclization is overcome without degrading the sensitive bipyridine scaffold.
Impurity control is inherently built into this mechanism due to the specificity of the reactants and the simplicity of the workup procedure. Unlike routes involving transition metal catalysts, there is no risk of heavy metal contamination requiring specialized scavenging resins. The dimethylamine byproduct generated during the reaction is easily removed during the aqueous workup or remains soluble in the mother liquor. The final adjustment of pH to 6-7 using acetic acid triggers the precipitation of the product in its neutral form, leaving ionic impurities in the solution. Subsequent pulping with hot water at 40-50°C further washes away residual salts and organic soluble impurities. This rigorous purification capability ensures that the final API intermediate meets the stringent specifications required for parenteral formulations, addressing the R&D Director's concern for impurity profiles.
How to Synthesize Milrinone Efficiently
Implementing this synthesis route requires adherence to specific operational parameters to maximize yield and purity outcomes. The process begins with the precise weighing of 1-(4-pyridyl)-2-acetone and 2-cyano-3-dimethylamino acrylamide in a molar ratio ranging from 1:1.2 to 1:1.8. These solids are dissolved in ethanol, creating a homogeneous solution that facilitates efficient molecular collision. The addition of the base must be controlled to maintain the target pH without causing localized exotherms that could degrade the reactants. Reaction progress is monitored via thin-layer chromatography to determine the optimal endpoint, preventing over-reaction which might lead to byproduct formation.
- Prepare the reaction mixture by combining 1-(4-pyridyl)-2-acetone and 2-cyano-3-dimethylamino acrylamide in an organic solvent such as ethanol.
- Adjust the pH to an alkaline environment (pH 12-14) using sodium hydroxide and heat the mixture to reflux temperature (60-85°C).
- Upon completion, adjust pH to neutral with acid, crystallize the product, and purify via hot water pulping to achieve high purity.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this technological shift offers profound benefits for procurement managers focused on cost reduction in pharmaceutical intermediates manufacturing. The elimination of expensive and hazardous reagents like potassium cyanide removes a significant line item from the raw material budget. Furthermore, the simplified purification process reduces the consumption of solvents and filtration media, leading to substantial cost savings in operational expenditures. The high yield achieved minimizes the amount of starting material required per kilogram of finished product, directly improving the gross margin profile of the manufacturing campaign. For supply chain heads, the robustness of this method ensures reducing lead time for high-purity pharmaceutical intermediates by minimizing batch failures and reprocessing needs. The use of commodity chemicals like ethanol and sodium hydroxide enhances supply security, as these materials are readily available globally compared to specialized organometallic catalysts.
- Cost Reduction in Manufacturing: The process architecture eliminates the need for costly transition metal catalysts and complex protection-deprotection sequences found in Suzuki coupling routes. By removing these expensive steps, the overall cost of goods sold is significantly lowered, allowing for more competitive pricing strategies in the global market. The reduction in waste treatment costs associated with non-toxic reagents further contributes to the economic viability of the project. Additionally, the high conversion rate means less raw material is wasted, optimizing the return on investment for every procurement dollar spent on feedstock.
- Enhanced Supply Chain Reliability: Relying on stable, commercially available starting materials mitigates the risk of supply disruptions caused by the scarcity of specialized precursors. The mild reaction conditions reduce the strain on equipment, lowering maintenance frequency and extending the lifecycle of production assets. This operational stability translates to more predictable delivery schedules for downstream customers, fostering stronger long-term partnerships. The ability to scale from laboratory to commercial production without fundamental process changes ensures a seamless transition during technology transfer phases.
- Scalability and Environmental Compliance: The absence of halogenated solvents and heavy metals simplifies the regulatory approval process for new manufacturing sites. Waste streams are easier to treat and dispose of, ensuring compliance with increasingly strict environmental regulations across different jurisdictions. This eco-friendly profile enhances the brand reputation of the manufacturer as a responsible partner in the pharmaceutical value chain. The process is inherently designed for commercial scale-up of complex heterocycles, allowing for flexible production volumes to meet fluctuating market demand without compromising quality standards.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. These insights are derived directly from the experimental data and comparative analysis provided in the patent documentation. Understanding these details helps stakeholders make informed decisions about adopting this method for their own production lines.
Q: How does the new synthesis method improve product purity compared to traditional routes?
A: The novel convergent route avoids the formation of colored impurities common in malononitrile-based methods, achieving purity levels exceeding 99.9% through simple recrystallization and water pulping.
Q: What are the safety advantages of avoiding potassium cyanide in Milrinone production?
A: Eliminating highly toxic reagents like potassium cyanide and malononitrile significantly reduces hazardous waste treatment costs and improves overall operational safety for industrial scale-up.
Q: Is this synthesis route suitable for large-scale commercial manufacturing?
A: Yes, the process utilizes common solvents like ethanol and mild reaction conditions (60-85°C), making it highly adaptable for commercial scale-up of complex pharmaceutical intermediates without requiring cryogenic equipment.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of adopting advanced synthesis technologies to maintain competitiveness in the global pharmaceutical market. Our team of expert chemists possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that innovations like the CN111377857B route are implemented with precision. We maintain stringent purity specifications and operate rigorous QC labs to guarantee that every batch of Milrinone meets the highest international standards. Our commitment to quality assurance means that clients can rely on us for consistent supply of high-purity pharmaceutical intermediates that facilitate the development of life-saving cardiovascular medications.
We invite potential partners to engage with our technical procurement team to discuss how this optimized synthesis route can benefit your specific supply chain requirements. By requesting a Customized Cost-Saving Analysis, you can gain a clearer understanding of the economic impact of switching to this greener, more efficient method. We encourage you to contact us to obtain specific COA data and route feasibility assessments tailored to your project needs. Together, we can drive forward the availability of essential medicines through superior chemical manufacturing excellence and collaborative innovation.
