Advanced Water-Based Purification Technology for High-Purity Milrinone Production
Advanced Water-Based Purification Technology for High-Purity Milrinone Production
The pharmaceutical industry constantly seeks robust manufacturing pathways that balance high purity with economic efficiency, particularly for critical cardiovascular medications like Milrinone. A pivotal advancement in this domain is detailed in patent CN103664773A, which introduces a novel preparing and refining method that fundamentally shifts the purification paradigm from hazardous organic solvents to an aqueous system. This technological breakthrough addresses long-standing challenges in the production of Milrinone, specifically targeting the removal of stubborn impurities and residual solvents that have historically plagued the quality of injectable-grade products. By leveraging the unique solubility characteristics of Milrinone hydrochloride salts, this method not only enhances the chemical profile of the final API but also streamlines the operational workflow, offering a compelling value proposition for both research and development teams and supply chain executives looking for reliable milrinone supplier partnerships.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Historically, the synthesis and purification of Milrinone have relied heavily on organic solvents such as Dimethylformamide (DMF), methanol, and ethanol for recrystallization processes. While these solvents are effective at dissolving the crude material, they introduce significant downstream complications that impact both product quality and operational safety. A primary concern is the tenacity of DMF residues; due to its high boiling point and strong solvating power, DMF is notoriously difficult to completely remove from the crystal lattice of the final product, often leading to solvent residue levels that exceed the strict limits required for injectable formulations. Furthermore, conventional methods often struggle to effectively eliminate specific related substances, such as Impurity A (1,6-dihydro-2-methyl-6-oxo-(3,4'-bis-pyridines)-5-acid amides), resulting in a final product that may appear yellow or faint yellow rather than the desired white crystalline powder. These quality deficits necessitate additional, costly processing steps and can lead to batch rejections, creating volatility in the supply of high-purity milrinone.
The Novel Approach
In stark contrast to these legacy techniques, the methodology disclosed in patent CN103664773A employs water as the primary medium for purification, capitalizing on the pH-dependent solubility of Milrinone. The process involves dissolving the crude product in water under acidic conditions (pH 1-4), followed by treatment with activated carbon to adsorb colored impurities and organic contaminants. This aqueous approach effectively bypasses the issues associated with high-boiling organic solvents, ensuring that the final product is free from toxic residues like DMF. The innovation lies in the precise control of temperature and pH during the crystallization phase, where the solution is cooled to between 0°C and 5°C to precipitate the pure compound. This shift not only simplifies the isolation procedure but also dramatically improves the visual and chemical quality of the API, yielding a white powder with impurity levels well below 0.1%, thereby meeting the rigorous specifications of the USP29 standard for injectable drugs.
Mechanistic Insights into Condensation and Aqueous Recrystallization
The core synthetic transformation involves the condensation reaction between ethoxy methylene malononitrile and 1-(4-pyridyl)-2-acetone in an absolute ethanol environment. This reaction is kinetically controlled, requiring a dropwise addition of the ketone solution to the nitrile solution at temperatures ranging from 20°C to 60°C, followed by a heating phase up to 75°C-85°C to drive the cyclization to completion. The mechanistic pathway facilitates the formation of the pyridone ring structure characteristic of Milrinone, but the true sophistication of this patent lies in the subsequent workup. By exploiting the basic nitrogen in the pyridine ring, the process converts the neutral Milrinone molecule into a water-soluble hydrochloride salt upon acidification. This conversion is critical because it allows the molecule to enter the aqueous phase while leaving many non-polar organic impurities behind or adsorbed onto the activated carbon surface.
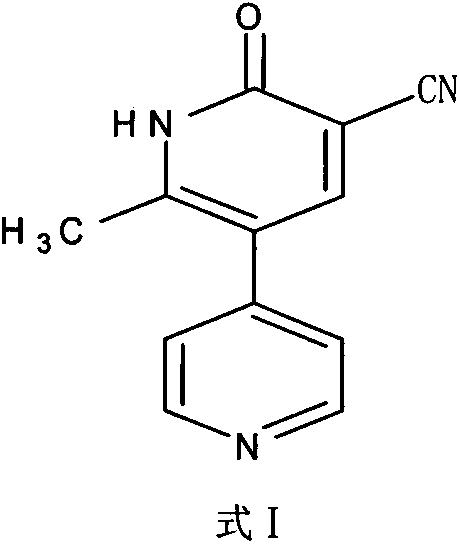
Once the impurities are sequestered, the pH is adjusted, or the temperature is lowered to reverse the solubility equilibrium, causing the pure Milrinone to crash out of the solution as a crystalline solid. This "salt-out" and "free-base" precipitation strategy is far more selective than simple organic recrystallization, as it relies on ionic interactions and hydrogen bonding networks that are highly specific to the target molecule. Consequently, the method achieves a remarkable suppression of Impurity A, reducing its concentration to as low as 0.02%, while keeping total impurities under 0.3%. For R&D directors, this level of control over the impurity profile is essential for ensuring the safety and efficacy of the final cardiovascular medication, minimizing the risk of adverse reactions associated with unknown degradants.
How to Synthesize Milrinone Efficiently
The implementation of this synthesis route requires precise adherence to the thermal and stoichiometric parameters outlined in the patent to ensure optimal yield and purity. The process begins with the preparation of the reaction mixture in absolute ethanol, maintaining a specific mass ratio of ethoxy methylene malononitrile to 1-(4-pyridyl)-2-acetone, typically around 1.5:1 to 3:1, to drive the equilibrium forward. Following the reaction, the crude solid is subjected to the multi-stage aqueous purification protocol involving acid dissolution, carbon treatment, and controlled cooling crystallization. For a comprehensive understanding of the operational specifics, including exact stirring times and filtration techniques, the detailed standardized synthesis steps are provided in the guide below.
- Condense ethoxy methylene malononitrile and 1-(4-pyridyl)-2-acetone in ethanol at controlled temperatures between 20°C and 85°C.
- Dissolve the crude product in water, adjust pH to 1-4 with hydrochloric acid, and treat with activated carbon to remove impurities.
- Filter the solution, cool to 0-5°C to crystallize the pure Milrinone, and dry under vacuum to obtain the final high-purity product.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, the transition to this water-based purification technology offers profound benefits that extend beyond mere technical compliance, directly impacting the bottom line and supply chain resilience. The most immediate advantage is the drastic reduction in raw material costs associated with solvent procurement and disposal. By replacing expensive and regulated solvents like DMF with water, manufacturers can significantly lower their variable costs per kilogram of produced API. The patent documentation explicitly notes that this substitution leads to a production cost decline of approximately 50%, a figure that represents a massive competitive advantage in the commoditized market of cardiovascular intermediates. This cost efficiency allows for more aggressive pricing strategies or higher margins, making the technology highly attractive for procurement managers focused on cost reduction in cardiovascular API manufacturing.
- Cost Reduction in Manufacturing: The elimination of DMF and the reduced reliance on large volumes of ethanol for purification translates directly into lower operational expenditures. Organic solvents require specialized storage, handling, and recovery systems, all of which add capital and operational overhead. By utilizing water, the process simplifies the infrastructure requirements, reduces energy consumption associated with solvent distillation, and minimizes waste disposal fees. Furthermore, the improved yield reported in the patent, with synthesis yields reaching approximately 84% and refining yields around 45.5%, ensures that more saleable product is generated from the same amount of starting material, further enhancing the economic viability of the process.
- Enhanced Supply Chain Reliability: Supply chain continuity is often threatened by the availability and regulatory status of hazardous chemicals. DMF, being a reproductive toxin, faces increasing regulatory scrutiny and usage restrictions globally. By adopting a water-based process, manufacturers mitigate the risk of supply disruptions caused by solvent shortages or sudden regulatory bans. Additionally, the simplicity of the aqueous workup reduces the complexity of the manufacturing schedule, potentially shortening the batch cycle time. This operational agility enables suppliers to respond more rapidly to market demand fluctuations, ensuring a steady flow of high-purity milrinone to downstream formulators without the bottlenecks associated with complex solvent recovery loops.
- Scalability and Environmental Compliance: As the pharmaceutical industry moves towards greener manufacturing practices, this technology positions itself as a leader in environmental stewardship. The reduction in volatile organic compound (VOC) emissions is substantial, aligning with global sustainability goals and easing the burden on environmental compliance teams. The process is inherently scalable, as demonstrated by the patent examples which utilize reactor sizes up to 150L, indicating readiness for commercial scale-up of complex pyridine derivatives. The ease of handling water compared to flammable organic solvents also improves workplace safety, reducing insurance premiums and liability risks associated with industrial chemical processing.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this advanced Milrinone manufacturing process. These insights are derived directly from the experimental data and beneficial effects described in patent CN103664773A, providing clarity on how this method compares to industry standards. Understanding these details is crucial for stakeholders evaluating the feasibility of adopting this technology for their own production lines or sourcing strategies.
Q: How does the water-based purification method improve Milrinone quality compared to traditional DMF methods?
A: Traditional methods using DMF often leave toxic solvent residues that are difficult to remove and fail injectable specifications. The patented water-based method utilizes the solubility differences of Milrinone salts to effectively remove Impurity A and solvent residues, ensuring single impurities are below 0.1% and meeting USP29 standards.
Q: What are the cost implications of switching to this aqueous purification process?
A: The patent data indicates a significant reduction in production costs, estimated at approximately 50%, primarily due to the replacement of expensive organic solvents like DMF and ethanol with water, alongside simplified operational procedures and improved yields.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process is explicitly designed for industrial suitability. It eliminates complex solvent recovery steps associated with DMF, uses readily available raw materials, and operates under manageable temperature conditions (20°C-85°C), making it highly scalable for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
The technological advancements detailed in patent CN103664773A represent a significant leap forward in the production of high-quality cardiovascular therapeutics, yet realizing this potential requires a manufacturing partner with deep technical expertise and robust infrastructure. NINGBO INNO PHARMCHEM stands at the forefront of this industry, possessing extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production. Our commitment to excellence is underpinned by stringent purity specifications and rigorous QC labs that ensure every batch of Milrinone meets the highest international standards, including USP and EP monographs. We understand that consistency is key in the pharmaceutical supply chain, and our state-of-the-art facilities are designed to deliver that reliability consistently.
We invite procurement leaders and technical directors to engage with us to explore how this optimized manufacturing route can benefit your specific supply chain needs. By partnering with us, you gain access to a Customized Cost-Saving Analysis that quantifies the potential economic benefits of switching to our superior grade of Milrinone. We encourage you to contact our technical procurement team today to request specific COA data and route feasibility assessments, ensuring that your next project is built on a foundation of quality, efficiency, and trust.
