Advanced Synthesis of (2S)-Indoline-2-Carboxylic Acid for Commercial Scale-Up
The pharmaceutical industry continuously seeks robust and cost-effective pathways for producing chiral intermediates essential for antihypertensive medications. Patent CN112375028A introduces a groundbreaking method for synthesizing (2S)-indoline-2-carboxylic acid, a critical building block for Perindopril, by leveraging a novel resolution strategy starting from indole-2-carboxylic acid. This technical breakthrough addresses long-standing challenges in chiral isomer resolution, offering a pathway that significantly enhances both economic viability and operational efficiency for large-scale manufacturing. By replacing expensive starting materials with more accessible precursors and integrating a recycling loop for unwanted isomers, this technology represents a paradigm shift in how fine chemical manufacturers approach the production of complex amino acid derivatives. The method not only ensures high optical purity but also streamlines the supply chain by reducing dependency on scarce raw materials, making it an attractive option for global procurement strategies focused on sustainability and cost control.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
Traditional synthetic routes for producing (S)-indoline-2-carboxylic acid have historically relied on direct resolution of racemic indoline-2-carboxylic acid or biocatalytic hydrolysis, both of which present significant industrial bottlenecks. Direct resolution often suffers from low efficiency and requires multiple recrystallization steps to achieve the necessary optical purity, leading to substantial material loss and increased waste generation. Furthermore, biocatalytic methods, while environmentally friendly in theory, frequently encounter issues with slow reaction rates and low yields when performed in aqueous environments, limiting their scalability for high-volume commercial production. The reliance on indoline-2-carboxylic acid as a starting material in prior art also imposes a heavy financial burden, as this precursor is inherently more expensive and less stable than its indole counterpart. These cumulative inefficiencies result in higher production costs and longer lead times, creating vulnerabilities in the supply chain for downstream API manufacturers who require consistent, high-quality intermediates.
The Novel Approach
The innovative method disclosed in the patent overcomes these deficiencies by utilizing indole-2-carboxylic acid as the primary raw material, which is not only more cost-effective but also allows for a protective group strategy that facilitates smoother downstream processing. By initially acetylating the indole nitrogen, the electronic properties of the molecule are modified to favor subsequent catalytic hydrogenation under milder conditions, avoiding the harsh pressures typically required for direct reduction. This strategic modification prevents catalyst poisoning and ensures a high conversion rate during the hydrogenation step, which is critical for maintaining process throughput. Additionally, the implementation of a mixed solvent system for resolution enhances the selectivity of the chiral separation, allowing for the isolation of the target (2S) isomer with superior purity in fewer steps. This holistic approach transforms a previously linear and wasteful process into a cyclic, efficient operation that maximizes resource utilization.
Mechanistic Insights into Acetylation and Chiral Resolution
The core of this synthesis lies in the precise manipulation of functional groups to control reactivity and stereochemistry throughout the reaction sequence. The process begins with the acetylation of racemic indole-2-carboxylic acid using acetic anhydride, which generates an N-acetyl protected intermediate that is crucial for the success of the subsequent hydrogenation. This protection step reduces the electron-donating capability of the nitrogen atom, thereby preventing potential side reactions and stabilizing the molecule against oxidative degradation during the catalytic phase. Following protection, the molecule undergoes catalytic hydrogenation using a palladium on carbon catalyst under controlled pressure and temperature, effectively saturating the pyrrole ring to form the indoline skeleton without affecting the carboxylic acid moiety. The careful control of reaction parameters during this stage is essential to prevent over-reduction or racemization, ensuring that the structural integrity of the intermediate is preserved for the final resolution step.
Following the formation of the racemic indoline derivative, the process employs a sophisticated chiral resolution technique using (R)-alpha-methylbenzylamine to separate the enantiomers. The interaction between the chiral amine and the racemic acid forms diastereomeric salts with different solubility profiles in a specific acetone-water mixed solvent system, allowing the desired (2S) isomer to crystallize out selectively. This crystallization is highly sensitive to solvent composition and temperature, requiring precise optimization to maximize the recovery of the target enantiomer while minimizing the co-precipitation of the unwanted (2R) form. The mother liquor, enriched with the (2R) isomer, is not discarded but instead subjected to a racemization process involving base treatment at elevated temperatures, which converts it back into the racemic mixture for re-entry into the resolution cycle. 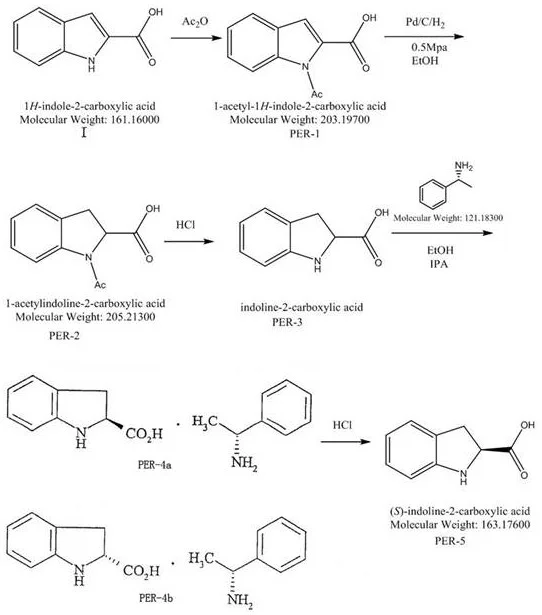
How to Synthesize (2S)-Indoline-2-Carboxylic Acid Efficiently
Implementing this synthesis route requires strict adherence to the specified reaction conditions and purification protocols to ensure consistent product quality and yield. The process involves a sequence of acetylation, hydrogenation, hydrolysis, and resolution steps, each demanding precise control over temperature, pressure, and stoichiometry to avoid impurities. Operators must pay particular attention to the hydrogenation phase, ensuring that the catalyst is properly activated and that the pressure is maintained within the optimal range to achieve complete conversion without degrading the product. For a comprehensive understanding of the operational parameters and safety considerations, the detailed standardized synthesis steps are provided in the guide below.
- React racemic indole-2-carboxylic acid with acetic anhydride to form N-acetylindole-2-carboxylic acid.
- Perform catalytic hydrogenation using Pd/C under pressure to obtain N-acetylindoline-2-carboxylic acid.
- Hydrolyze the acetyl group and resolve the racemate using (R)-alpha-methylbenzylamine to isolate the (2S) isomer.
Commercial Advantages for Procurement and Supply Chain Teams
From a commercial perspective, this patented methodology offers profound advantages that directly address the key concerns of procurement managers and supply chain directors regarding cost stability and material availability. By shifting the raw material base to indole-2-carboxylic acid, manufacturers can leverage a more abundant and less volatile market supply, significantly mitigating the risk of price spikes associated with specialized fine chemicals. The elimination of complex biocatalytic steps and the reduction in purification cycles translate to a streamlined production workflow that lowers operational expenditures and energy consumption. Furthermore, the ability to recycle the unwanted isomer back into the process creates a closed-loop system that drastically reduces waste disposal costs and environmental impact, aligning with modern green chemistry initiatives. These factors combine to create a supply chain that is not only more cost-efficient but also more resilient against external market fluctuations.
- Cost Reduction in Manufacturing: The substitution of expensive indoline-2-carboxylic acid with the more economical indole-2-carboxylic acid results in a fundamental decrease in raw material expenditure, which is a primary driver of overall production costs. The protective group strategy employed in this route simplifies the hydrogenation conditions, reducing the need for high-pressure equipment and specialized catalysts that typically incur high maintenance and replacement costs. Additionally, the high efficiency of the resolution step minimizes material loss, ensuring that a greater proportion of the input mass is converted into saleable product, thereby improving the overall cost-per-kilogram metric. These cumulative savings allow for a more competitive pricing structure without compromising on the quality or purity specifications required by regulatory bodies.
- Enhanced Supply Chain Reliability: Utilizing a starting material that is widely available in the bulk chemical market reduces dependency on niche suppliers who may face production disruptions or capacity constraints. The robustness of the chemical synthesis steps, as opposed to sensitive enzymatic processes, ensures that production can be maintained consistently across different seasons and geographic locations without significant variability in yield. The integrated racemization loop further secures supply continuity by maximizing the utility of every batch of raw material, effectively buffering against potential shortages of the chiral resolving agent. This reliability is crucial for long-term supply agreements where consistent delivery schedules are paramount for the downstream formulation of finished pharmaceutical products.
- Scalability and Environmental Compliance: The process is designed with scalability in mind, utilizing standard unit operations such as hydrogenation and crystallization that are easily transferable from pilot scale to multi-ton commercial production. The reduction in solvent usage and the ability to recycle byproducts significantly lower the volume of hazardous waste generated, simplifying compliance with increasingly stringent environmental regulations. The avoidance of heavy metal contaminants, often associated with less refined catalytic processes, reduces the burden on downstream purification and analytical testing, accelerating the time to market for new drug applications. This alignment with sustainable manufacturing practices enhances the corporate social responsibility profile of the supply chain, appealing to stakeholders who prioritize eco-friendly production methods.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the synthesis and supply of (2S)-indoline-2-carboxylic acid, based on the detailed specifications outlined in the patent literature. These insights are intended to clarify the operational benefits and quality assurances associated with this advanced manufacturing route. Understanding these details is essential for technical teams evaluating the feasibility of integrating this intermediate into their existing production pipelines.
Q: What is the primary advantage of using indole-2-carboxylic acid over indoline-2-carboxylic acid?
A: Indole-2-carboxylic acid is significantly cheaper and more readily available than indoline-2-carboxylic acid, leading to substantial raw material cost reductions without compromising final product quality.
Q: How is high optical purity achieved in this synthesis route?
A: High optical purity (>99.5% ee) is achieved through diastereomeric salt formation using (R)-alpha-methylbenzylamine in a specific acetone-water mixed solvent system, followed by recrystallization.
Q: Is the unwanted (2R) isomer wasted in this process?
A: No, the process includes a racemization step where the mother liquor containing the (2R) isomer is treated with base and heat to regenerate racemic indoline-2-carboxylic acid for reuse, maximizing overall yield.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable (2S)-Indoline-2-Carboxylic Acid Supplier
At NINGBO INNO PHARMCHEM, we recognize the critical importance of high-quality intermediates in the development and manufacture of life-saving cardiovascular medications. Our team possesses extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the rigorous demands of global pharmaceutical clients. We are committed to delivering (2S)-indoline-2-carboxylic acid with stringent purity specifications and consistent batch-to-batch reproducibility, supported by our rigorous QC labs that employ state-of-the-art analytical instrumentation. Our expertise in chiral resolution and catalytic hydrogenation allows us to optimize this specific patent route for maximum efficiency, providing our partners with a secure and cost-effective source of this vital building block.
We invite procurement leaders and R&D directors to collaborate with us to explore how this optimized synthesis can enhance your supply chain resilience and reduce overall manufacturing costs. By requesting a Customized Cost-Saving Analysis, you can gain deeper insights into the specific economic benefits tailored to your volume requirements. We encourage you to contact our technical procurement team to索取 specific COA data and route feasibility assessments, ensuring that our capabilities align perfectly with your project timelines and quality standards.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
