Advanced Manufacturing of High-Purity Milrinone Intermediates for Global Pharma Supply Chains
Advanced Manufacturing of High-Purity Milrinone Intermediates for Global Pharma Supply Chains
The pharmaceutical industry continuously seeks robust synthetic pathways for critical cardiovascular agents, and the preparation method disclosed in patent CN111377858B represents a significant leap forward in the manufacturing of Milrinone. As a potent phosphodiesterase III inhibitor, Milrinone is essential for treating refractory heart failure, yet its historical synthesis has been plagued by safety hazards and low yields. This groundbreaking technology introduces a convergent synthetic strategy that utilizes 1-(4-pyridyl)-2-acetone and 2-cyano-3-ethoxy acrylamide as key starting materials. By shifting away from toxic cyanide sources and complex multi-step sequences, this method delivers a white crystalline product with exceptional purity directly from the reactor. For R&D directors and procurement specialists, this patent offers a viable pathway to secure a reliable pharmaceutical intermediate supplier capable of meeting stringent global quality standards while mitigating supply chain risks associated with hazardous reagents.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
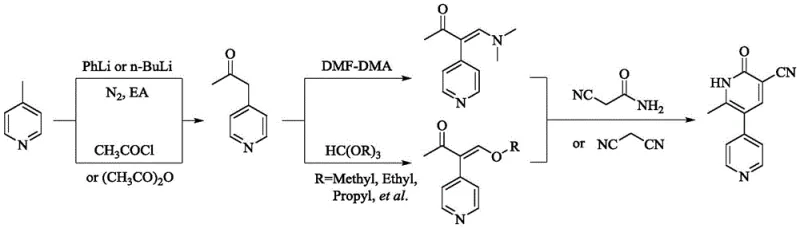
Historically, the industrial production of Milrinone has relied on linear or convergent routes that present severe operational and economic bottlenecks. Traditional linear syntheses, such as those described in earlier patents, often involve the condensation of 4-methylpyridine derivatives with highly toxic malononitrile or require the use of potassium cyanide in late-stage functionalization. These reagents not only pose extreme safety risks to personnel but also generate hazardous waste streams that complicate environmental compliance. Furthermore, alternative routes utilizing Suzuki coupling reactions necessitate the use of expensive palladium catalysts and rigorous purification steps to remove trace metals, which is unacceptable for injectable drug formulations. Other methods require cryogenic conditions down to -78°C, demanding specialized equipment and excessive energy consumption, thereby inflating the cost reduction in API manufacturing efforts. The cumulative effect of these drawbacks is a process with low overall yield, often producing red-colored crude products that require multiple, yield-eroding recrystallizations to meet pharmacopeial standards.
The Novel Approach
In stark contrast to these legacy methods, the novel approach detailed in CN111377858B streamlines the synthesis into a highly efficient, one-pot condensation and cyclization sequence. This method bypasses the need for dangerous cyanide salts by employing 2-cyano-3-ethoxy acrylamide, a safer and more stable synthon that effectively introduces the required cyano and amide functionalities simultaneously. The reaction proceeds under mild alkaline conditions in common alcohol solvents like ethanol or methanol, eliminating the need for cryogenic cooling or inert atmosphere protections typically required for organolithium reagents. This simplicity translates directly into enhanced process robustness, allowing for the direct isolation of high-purity white crystals without the extensive chromatographic purifications seen in older protocols. By integrating these improvements, manufacturers can achieve a dramatic increase in throughput and consistency, positioning this technology as the preferred choice for any entity seeking a reliable pharmaceutical intermediate supplier for cardiac therapeutics.
Mechanistic Insights into Base-Catalyzed Condensation and Cyclization
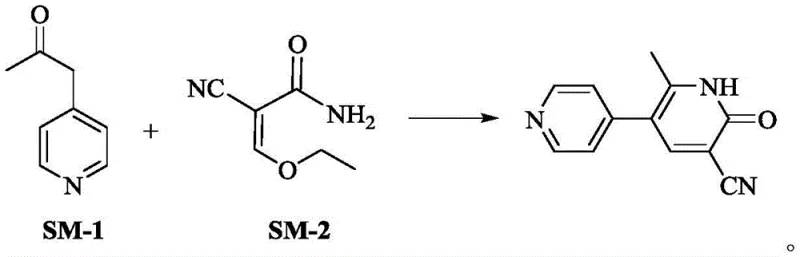
The core of this technological advancement lies in the precise control of the alkaline environment during the condensation of the ketone and the acrylamide derivative. The mechanism initiates with the deprotonation of the alpha-carbon of 1-(4-pyridyl)-2-acetone by a strong base, such as sodium hydroxide or sodium ethoxide, generating a reactive enolate species. This nucleophile then attacks the electron-deficient beta-carbon of the 2-cyano-3-ethoxy acrylamide in a Michael-type addition. Subsequent intramolecular cyclization occurs through the nucleophilic attack of the amide nitrogen onto the carbonyl carbon, followed by the elimination of ethanol to establish the aromatic pyridone ring system. The patent specifies a critical pH range of 12 to 14, which is essential to drive the equilibrium towards the product while preventing the hydrolysis of the sensitive nitrile group. Maintaining this specific alkalinity ensures that the reaction kinetics favor the formation of the desired 1,6-dihydro-2-methyl-6-oxo-[3,4'-bipyridine]-5-carbonitrile scaffold over potential side products, thereby securing the high purity observed in the final isolate.
Impurity control is another pivotal aspect of this mechanism, particularly regarding the physical appearance of the product. Conventional methods often suffer from the formation of conjugated polymeric byproducts or oxidation impurities that impart a red color to the crude material, necessitating aggressive decolorization steps. In this novel process, the use of mild reaction temperatures between 60°C and 85°C minimizes thermal degradation and polymerization pathways. Furthermore, the choice of solvent plays a crucial role; alcohols like ethanol not only solubilize the reactants effectively but also participate in the transesterification equilibrium if necessary, ensuring a clean reaction profile. The final acidification step to pH 6-7 triggers the precipitation of the product in its neutral form, effectively excluding ionic impurities and residual starting materials from the crystal lattice. This results in a product with HPLC purity consistently above 99.7%, demonstrating superior control over the杂质谱 (impurity profile) compared to prior art.
How to Synthesize Milrinone Efficiently
To implement this high-yield synthesis in a laboratory or pilot plant setting, operators must adhere to strict parameters regarding reagent stoichiometry and temperature control. The process begins by dissolving the ketone and acrylamide precursors in a lower alcohol solvent, followed by the careful addition of a base to establish the requisite alkaline environment. Heating the mixture to reflux facilitates the cyclization, after which the reaction progress is monitored via TLC to ensure complete conversion before cooling.
- Combine 1-(4-pyridyl)-2-acetone (SM-1) and 2-cyano-3-ethoxy acrylamide (SM-2) in an organic solvent such as ethanol, maintaining a molar ratio between 1: 1.2 and 1:2.0.
- Adjust the reaction mixture to a strongly alkaline environment (pH 12-14) using sodium hydroxide or alkoxides, and heat to reflux temperatures between 60°C and 85°C.
- Upon completion, cool the solution, adjust pH to 6-7 with acid to induce crystallization, and filter the resulting white solid to obtain high-purity Milrinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain heads, the adoption of this synthesis route offers transformative benefits that extend far beyond simple yield improvements. By eliminating the reliance on highly regulated and dangerous substances like potassium cyanide, facilities can significantly reduce the costs associated with hazardous material handling, storage, and waste disposal. This shift not only lowers the operational expenditure but also mitigates the risk of production stoppages due to regulatory inspections or safety incidents. Moreover, the use of commodity chemicals such as ethanol and sodium hydroxide ensures that the supply chain remains resilient against raw material shortages, as these inputs are globally available and inexpensive. The simplified workup procedure, which relies on straightforward crystallization rather than complex chromatography or multiple recrystallizations, drastically shortens the production cycle time, enabling faster turnaround for customer orders and improved inventory turnover rates.
- Cost Reduction in Manufacturing: The economic impact of this process is profound, primarily driven by the removal of expensive transition metal catalysts and the avoidance of cryogenic infrastructure. Traditional routes often require palladium catalysts for coupling reactions, which represent a significant variable cost and require costly recovery systems to meet residual metal limits. By replacing these with inexpensive inorganic bases and operating at near-ambient pressures and moderate temperatures, the energy footprint of the manufacturing process is substantially decreased. Additionally, the high crude purity means that solvent consumption for purification is minimized, leading to further savings in utility and material costs. These factors combine to create a leaner manufacturing model that enhances profit margins without compromising on the quality of the high-purity pharmaceutical intermediate.
- Enhanced Supply Chain Reliability: Supply continuity is critical for life-saving medications, and this synthetic route bolsters reliability by simplifying the bill of materials. The starting materials, 1-(4-pyridyl)-2-acetone and 2-cyano-3-ethoxy acrylamide, are stable and commercially accessible, reducing the dependency on custom synthesis of exotic intermediates. The robustness of the reaction conditions means that the process is less susceptible to batch-to-batch variability caused by minor fluctuations in temperature or moisture, a common issue with moisture-sensitive organometallic reagents. This consistency allows for more accurate demand forecasting and production planning, ensuring that downstream API manufacturers receive their materials on schedule. Consequently, partners can maintain lower safety stock levels while still guaranteeing uninterrupted supply to the market.
- Scalability and Environmental Compliance: Scaling chemical processes from the bench to commercial production often reveals hidden complexities, but this method is inherently designed for scalability. The absence of exothermic hazards associated with cyanide additions or cryogenic quenches makes the scale-up trajectory smooth and predictable. From an environmental perspective, the process aligns with green chemistry principles by utilizing safer solvents and generating less toxic waste. The elimination of heavy metals and cyanide from the waste stream simplifies effluent treatment, reducing the burden on wastewater treatment facilities and ensuring compliance with increasingly stringent environmental regulations. This sustainability profile is increasingly important for multinational corporations aiming to reduce their carbon footprint and meet corporate social responsibility goals.
Frequently Asked Questions (FAQ)
Understanding the technical nuances of this new preparation method is vital for stakeholders evaluating its integration into their supply networks. The following questions address common concerns regarding quality, safety, and scalability, drawing directly from the experimental data and technical disclosures within the patent documentation.
Q: How does this new process improve product purity compared to traditional methods?
A: Traditional methods often yield red-colored crude products requiring multiple recrystallizations. This novel base-catalyzed route produces white crystalline Milrinone directly with HPLC purity exceeding 99.7%, significantly reducing refining steps.
Q: What are the safety advantages regarding reagent selection?
A: Unlike prior art that utilizes highly toxic potassium cyanide or malononitrile, this method employs 2-cyano-3-ethoxy acrylamide, drastically improving operational safety and reducing hazardous waste disposal costs.
Q: Is this synthesis route suitable for large-scale industrial production?
A: Yes, the process operates at mild temperatures (60-85°C) without cryogenic conditions or expensive transition metal catalysts, making it highly scalable and cost-effective for commercial manufacturing.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
As the global demand for cardiovascular therapeutics continues to rise, partnering with a manufacturer that possesses both technical expertise and scalable capacity is paramount. NINGBO INNO PHARMCHEM stands at the forefront of this industry, leveraging advanced synthetic methodologies like the one described in CN111377858B to deliver superior results. Our facility is equipped with extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that we can meet the volume requirements of major pharmaceutical companies without delay. We maintain stringent purity specifications through our rigorous QC labs, utilizing state-of-the-art analytical instrumentation to verify that every batch of Milrinone meets or exceeds international pharmacopeial standards. Our commitment to quality assurance guarantees that the material you receive is safe, effective, and ready for immediate formulation into final drug products.
We invite procurement leaders and R&D teams to engage with us to explore how this optimized synthesis route can benefit your specific projects. By collaborating with our technical procurement team, you can request a Customized Cost-Saving Analysis tailored to your volume needs, demonstrating exactly how switching to our process can optimize your budget. We encourage you to contact us today to obtain specific COA data and route feasibility assessments, allowing you to make informed decisions that enhance your supply chain efficiency and product quality. Let us be your trusted partner in navigating the complexities of fine chemical manufacturing and delivering value to your organization.
