Advanced Milrinone Synthesis: A Technical Breakthrough for Global Pharmaceutical Supply Chains
Advanced Milrinone Synthesis: A Technical Breakthrough for Global Pharmaceutical Supply Chains
The pharmaceutical industry continuously seeks robust and scalable synthetic routes for critical cardiac medications, and the recent disclosure in patent CN113493411A represents a significant leap forward in the manufacturing of Milrinone. This potent phosphodiesterase III inhibitor, essential for treating refractory heart failure and managing low-cardiac-ejection syndromes, has historically been plagued by synthesis challenges involving hazardous reagents and complex purification steps. The new methodology introduces a streamlined convergent approach that reacts 1-(4-pyridyl)-2-acetone with alpha-(N,N-dimethylamine methylene) malononitrile, effectively bypassing the safety risks and low yields associated with legacy processes. By leveraging this innovative chemistry, manufacturers can achieve off-white solid products with exceptional purity profiles, directly addressing the stringent quality requirements of global regulatory bodies. This technical evolution not only enhances the safety profile of the production facility but also establishes a foundation for more reliable supply chains capable of meeting the growing demand for high-quality cardiovascular therapeutics.
The Limitations of Conventional Methods vs. The Novel Approach
The Limitations of Conventional Methods
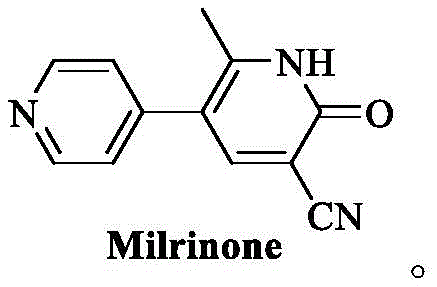
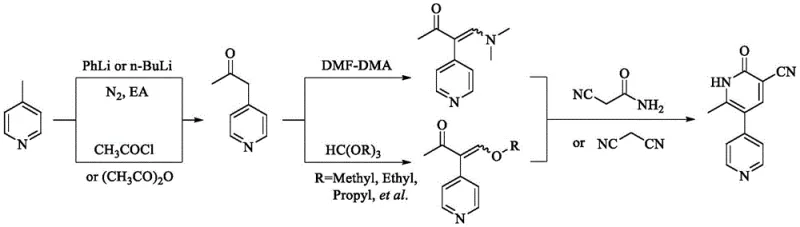
Historically, the synthesis of Milrinone has relied on two primary pathways, both of which present substantial hurdles for modern industrial application. The first pathway, often referred to as the 'linear' route, frequently employs highly reactive organolithium reagents such as n-butyllithium or phenyllithium to facilitate the initial acylation of methylpyridine. These reagents are notoriously pyrophoric, requiring strictly anhydrous conditions and inert atmospheres to prevent catastrophic combustion or explosion upon exposure to air or moisture. Furthermore, the post-treatment of these reactions is cumbersome, often necessitating multiple extractions and concentration steps that consume vast quantities of solvents and generate significant hazardous waste. The second conventional pathway involves a convergent strategy using malononitrile and trialkyl orthoformate, which, while avoiding organolithiums, introduces its own set of complications including the use of toxic malononitrile and the formation of deeply colored impurities that are difficult to remove. As illustrated in the comparative reaction schemes, these traditional methods often result in crude products with poor color and require extensive recrystallization, leading to overall yields that can drop as low as 45.5%, thereby severely impacting production efficiency and cost-effectiveness.
The Novel Approach
In stark contrast to the hazardous and inefficient legacy methods, the novel approach detailed in the patent utilizes a highly efficient condensation reaction between 1-(4-pyridyl)-2-acetone and alpha-(N,N-dimethylamine methylene) malononitrile. This strategy fundamentally alters the reaction landscape by replacing volatile and toxic precursors with a stable dimethylamine derivative that facilitates a smoother cyclization process. The reaction proceeds under mild conditions in common alcoholic solvents like ethanol, eliminating the need for cryogenic temperatures or exotic catalysts. Crucially, this new route inherently suppresses the formation of the deep red polymeric impurities that characterize the older ethoxymethylene malononitrile methods, resulting in a crude product that is already off-white and requires minimal purification. The simplicity of the workup, which involves merely adjusting the pH and performing a hot water wash, drastically reduces the operational complexity and solvent consumption. This paradigm shift not only improves the safety profile of the manufacturing plant but also ensures a consistent supply of high-purity material that meets the rigorous specifications required for injectable formulations.
Mechanistic Insights into the Dimethylamine-Mediated Cyclization
The core of this technological advancement lies in the unique reactivity of alpha-(N,N-dimethylamine methylene) malononitrile, which acts as a superior electrophile in the condensation step. Unlike the ethoxy counterpart, the dimethylamine group is a better leaving group under the specific basic conditions employed, facilitating a more rapid and complete nucleophilic attack by the enolate formed from 1-(4-pyridyl)-2-acetone. The reaction mechanism likely proceeds through a Knoevenagel-type condensation followed by an intramolecular cyclization and subsequent elimination of dimethylamine to form the pyridone ring system. This pathway is kinetically favored and thermodynamically stable, ensuring that the reaction drives to completion with minimal side reactions. The use of a strong base, such as sodium hydroxide or sodium ethoxide, to maintain a pH between 12 and 14 is critical for generating the necessary enolate concentration without degrading the sensitive nitrile functionalities. This precise control over the reaction environment prevents the polymerization events that typically lead to tar formation and coloration in other routes, thereby preserving the structural integrity and optical properties of the final molecule.
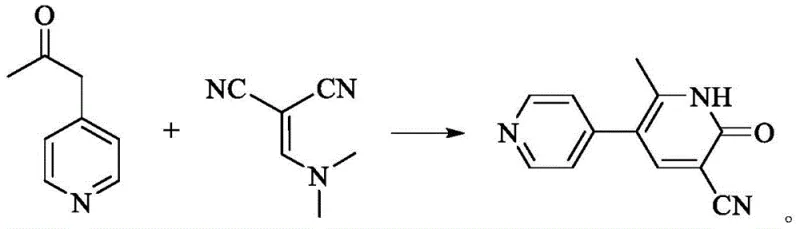
Impurity control is another critical aspect where this mechanism excels, particularly regarding the removal of residual amines and colored byproducts. In traditional syntheses, the persistence of colored impurities often necessitates the use of activated carbon or column chromatography, which are impractical on a multi-ton scale. However, in this new process, the byproduct dimethylamine forms a water-soluble salt that is easily separated during the aqueous workup. The subsequent adjustment of the pH to neutral levels using acetic acid induces the precipitation of the Milrinone product in a highly crystalline form. The final hot water pulping step at 40-50°C serves as a powerful purification tool, dissolving any remaining surface impurities while leaving the pure Milrinone crystals intact. This mechanistic elegance ensures that the final product achieves a purity of 99.97% with an off-white appearance, demonstrating that the choice of the dimethylamine precursor is not merely a substitution but a strategic optimization of the entire synthetic trajectory.
How to Synthesize Milrinone Efficiently
Implementing this synthesis route requires careful attention to stoichiometry and pH control to maximize the benefits of the novel chemistry. The process begins with the dissolution of the ketone and the malononitrile derivative in ethanol, followed by the gradual addition of a base to initiate the reaction under reflux conditions. Once the conversion is complete, the mixture is cooled and acidified to precipitate the product, which is then isolated by filtration. The simplicity of these steps belies the sophistication of the underlying chemistry, which allows for a seamless transition from laboratory bench to commercial reactor. For a detailed breakdown of the specific operating parameters, reagent ratios, and safety protocols required to execute this synthesis, please refer to the standardized guide below.
- Prepare the reaction mixture by adding 1-(4-pyridyl)-2-acetone and alpha-(N,N-dimethylamine methylene) malononitrile into an alcoholic solvent such as ethanol.
- Adjust the pH of the solution to 12-14 using an inorganic or organic base, then heat the mixture to reflux temperature to initiate the cyclization reaction.
- Upon completion, cool the solution, adjust pH to 6-7 with acid to induce crystallization, filter the solid, and wash with hot water to obtain off-white Milrinone.
Commercial Advantages for Procurement and Supply Chain Teams
For procurement managers and supply chain directors, the adoption of this new synthesis route offers transformative benefits that extend far beyond simple yield improvements. The elimination of hazardous organolithium reagents and toxic malononitrile significantly reduces the regulatory burden and insurance costs associated with handling dangerous chemicals. Furthermore, the use of ethanol as a primary solvent aligns with sustainability goals and simplifies solvent recovery systems, leading to substantial operational cost savings. The robustness of the process ensures consistent batch-to-batch quality, reducing the risk of production delays caused by failed batches or out-of-specification results. This reliability is paramount for maintaining continuous supply to downstream formulation partners who depend on timely deliveries of critical API intermediates.
- Cost Reduction in Manufacturing: The economic impact of this process is driven by the drastic simplification of the post-treatment workflow. By removing the need for complex extraction sequences, column chromatography, or multiple recrystallizations, the manufacturing cycle time is significantly shortened, and labor costs are reduced. Additionally, the high yield of over 90% means that less raw material is required to produce the same amount of finished product, directly lowering the cost of goods sold. The avoidance of expensive and specialized reagents like n-butyllithium further contributes to a leaner cost structure, making the final Milrinone product more competitive in the global market.
- Enhanced Supply Chain Reliability: Supply chain resilience is bolstered by the use of readily available and stable starting materials. Unlike reagents that require special storage conditions or have short shelf lives, the precursors used in this method are commodity chemicals that can be sourced from multiple suppliers, mitigating the risk of single-source bottlenecks. The simplified process flow also reduces the dependency on specialized equipment, allowing for greater flexibility in manufacturing site selection. This flexibility ensures that production can be scaled up or shifted between facilities with minimal disruption, guaranteeing a steady flow of high-purity intermediates to meet market demand.
- Scalability and Environmental Compliance: From an environmental perspective, this route offers a greener alternative to traditional methods by minimizing waste generation and solvent usage. The aqueous workup and hot water washing steps generate wastewater that is easier to treat compared to the organic-heavy waste streams of older processes. The high atom economy of the reaction ensures that a greater proportion of the input mass ends up in the final product, reducing the overall environmental footprint. These factors make the process highly scalable and compliant with increasingly stringent environmental regulations, future-proofing the manufacturing operation against potential regulatory changes.
Frequently Asked Questions (FAQ)
The following questions address common technical and commercial inquiries regarding the implementation of this synthesis technology. The answers are derived directly from the experimental data and beneficial effects reported in the patent documentation, providing a clear understanding of the process capabilities. These insights are intended to assist technical teams in evaluating the feasibility of adopting this route for their specific production needs.
Q: What are the primary advantages of this new Milrinone synthesis route compared to conventional methods?
A: The new route described in CN113493411A eliminates the need for harsh reagents like n-butyllithium or toxic malononitrile, resulting in milder reaction conditions, higher yields (up to 92.8%), and superior product purity (99.97%) without the deep red coloration associated with older methods.
Q: Is this synthesis method suitable for large-scale industrial production?
A: Yes, the process is specifically designed for industrial scalability, utilizing common solvents like ethanol and simple unit operations such as filtration and hot water washing, which significantly simplifies the post-treatment process compared to complex extraction methods.
Q: How does this method address impurity control in Milrinone manufacturing?
A: By using alpha-(N,N-dimethylamine methylene) malononitrile instead of ethoxymethylene malononitrile, the reaction avoids the formation of difficult-to-remove colored impurities, allowing the final product to meet strict appearance and purity standards through a simple hot water pulping step.
Partnering with NINGBO INNO PHARMCHEM: Your Reliable Milrinone Supplier
At NINGBO INNO PHARMCHEM, we recognize that the transition to a new synthetic route requires a partner with deep technical expertise and proven scale-up capabilities. Our team of process chemists has extensive experience scaling diverse pathways from 100 kgs to 100 MT/annual commercial production, ensuring that the theoretical benefits of this patent are fully realized in a commercial setting. We operate stringent purity specifications and maintain rigorous QC labs equipped with state-of-the-art analytical instruments to verify that every batch of Milrinone meets the highest international standards. Our commitment to quality and consistency makes us the ideal partner for pharmaceutical companies seeking to secure their supply of this critical cardiac medication.
We invite you to engage with our technical procurement team to discuss how this advanced synthesis method can optimize your supply chain. By requesting a Customized Cost-Saving Analysis, you can gain a detailed understanding of the potential economic benefits specific to your operation. We encourage you to reach out for specific COA data and route feasibility assessments to validate the performance of this technology in your own context. Let us help you engineer a more efficient and reliable supply chain for Milrinone.
Engineering Bottleneck?
Can't scale up this synthesis? Upload your target structure or CAS, and our CDMO team will evaluate the industrial feasibility within 24 hours. Request Evaluation →
